Can Matter change its state Class 9

Lily Fisher
Published May 11, 2026
Matters can be changed from one state to another state. A solid can be changed into liquid and a liquid can be changed into gas. Most of the metals, which are solid, turn into liquid on heating and turn into vapor on further heating.
Can matter can change its state?
The answer is ‘Yes’. It can definitely change its shape, size, and volume. For examples, water turns into ice upon freezing, here the form of water converts from the liquid state into the solid state; the matter itself doesn’t change but it transforms its shape.
Can matter is changed?
Matter can change form through physical and chemical changes, but through any of these changes matter is conserved. The same amount of matter exists before and after the change—none is created or destroyed. This concept is called the Law of Conservation of Mass.
How can we change state of matter?
We can change a solid into a liquid or gas by changing its temperature. This is known as changing its state. Water is a liquid at room temperature, but becomes a solid (called ice) if it is cooled down. The same water turns into a gas (called water vapor) if it is heated up.Can matter change its state if yes explain with example?
The answer is ‘Yes‘. It can definitely change its shape, size, and volume. For examples, water turns into ice upon freezing, here the form of water converts from the liquid state into the solid state; the matter itself doesn’t change but it transforms its shape.
Why matter changes its state?
Changing states of matter occur when matter loses or absorbs energy. When a substance absorbs energy the atoms and molecules move more rapidly and this increased kinetic energy pushes particles far enough, that they change form. This energy is usually heat or thermal energy.
What is an example of matter changing state?
When matter changes from one state to another, it either absorbs energy—as when chocolate melts—or loses energy. For example, if you were to place the melted chocolate in a refrigerator, it would lose energy to the cold air inside the refrigerator. As a result, the liquid chocolate would change to a solid again.
Why do changes in matter occur?
Changes of state are physical changes. They occur when matter absorbs or loses energy. Processes in which matter changes between liquid and solid states are freezing and melting. Processes in which matter changes between liquid and gaseous states are vaporization, evaporation, and condensation.What causes matter to change its state?
Adding or removing energy from matter causes a physical change as matter moves from one state to another. For example, adding thermal energy (heat) to liquid water causes it to become steam or vapor (a gas). And removing energy from liquid water causes it to become ice (a solid).
How does matter change from one state to another state?Matter can change from one state to another when thermal energy is absorbed or released. This change is known as change of state. The graph in Figure 11 shows the changes in temperature as thermal energy is gradually added to a container of ice. heated, it absorbs thermal energy and its temperature rises.
Article first time published onWhat is liquid to gas called?
vaporization, conversion of a substance from the liquid or solid phase into the gaseous (vapour) phase. If conditions allow the formation of vapour bubbles within a liquid, the vaporization process is called boiling. Direct conversion from solid to vapour is called sublimation. … See also evaporation and sublimation.
Can matter be destroyed?
Matter makes up all visible objects in the universe, and it can be neither created nor destroyed.
What are the 3 changes of matter?
Through an instructional video, games, and activities, students explore three types of changes that occur in matter: physical change, in which only the shape of the matter changes; physical phase change, in which matter changes to a different form (solid, liquid, or gas); and chemical change, in which matter is changed …
What is an example of matter changing over time?
Examples of Phase Changes For example, you have probably witnessed freezing, melting, and vaporization just by making ice, melting ice, and boiling water. Condensation often occurs on the outside of cold beverage containers. This is when the humid air changes directly to a liquid on the surface of the container.
What are the states of matter Class 9?
- Solid: Matters which have fixed volume and shape are called solids. …
- Liquid: Matters which have fixed volume but indefinite shape are called liquids. …
- Gas: Matters which have indefinite shape and volume are called gases.
Is there a 5th state of matter?
There are four states of matter common in everyday life — gases, liquids, solids, and plasmas. However, there is also a fifth state of matter — Bose-Einstein condensates (BECs), which scientists first created in the lab 25 years ago.
What is matter in science class 9?
1. Matter- Matter is anything which occupies space and has mass is called matter. Air and water, sugar and sand, hydrogen and oxygen etc. Matter is made up of very small tiny particles. Particles of matter have space between them they attract each other.
Is a change of state of matter a physical or chemical one?
Solution: Change in state of matter is a physical change because of the physical condition and appearance changes but not the chemical composition.
What are the three states of matter?
They are very compressible (particles are widely spaced). There are three states of matter: solid; liquid and gas. They have different properties, which can be explained by looking at the arrangement of their particles.
What is matter made of?
At the most fundamental level, matter is composed of elementary particles known as quarks and leptons (the class of elementary particles that includes electrons). Quarks combine into protons and neutrons and, along with electrons, form atoms of the elements of the periodic table, such as hydrogen, oxygen, and iron.
What are the 7 states of matter?
Matter is any thing that is made from atoms and molecules. ( Studios, 1995) . The seven states of matter that I am investigating are Solids, Liquids, Gases, Ionized Plasma, Quark-Gluon Plasma, Bose-Einstein Condensate and Fermionic Condensate. Solid Definition – Chemistry Glossary Definition of Solid.
What is ice dry?
Dry ice is the solid form of carbon dioxide. … Dry ice sublimates at 194.7 K (−78.5 °C; −109.2 °F) at Earth atmospheric pressure. This extreme cold makes the solid dangerous to handle without protection from frostbite injury.
What form of matter is sweat?
Sweat, or perspiration, is a liquid made by the skin when the body is hot. Sweat is made in sweat glands under the surface of the skin. It comes out of tiny holes in the skin called pores. Sweat is mostly water, but it also contains some salts.
Which is a form of vaporization?
Vaporization is the process by which a liquid is turned into a gas. The two types of vaporization are evaporation and boiling. … Boiling refers to heating up a liquid until it releases vapor, such as heating water on a stove until steam forms.
Are humans matter?
At any rate, yes, human beings are made of “matter”, as is everything else that exists that isn’t energy. Specifically, we are composed of elements, the elements of the periodic table.
Is water a matter?
States of Matter Not only is water the most common substance on earth, but it is also the only substance that commonly appears as a solid, a liquid, and a gas within the normal range of earth’s temperatures. This makes water a good model for discussing the solid, liquid, and gas states of matter.
Is new matter ever created?
Newly created matter is a random assortment of all the fundamental particles it’s possible to create at the given energies. … Most fundamental particles are extremely unstable and decay very rapidly into radiation and the few stable particles: protons, electrons, and their anti-particles.
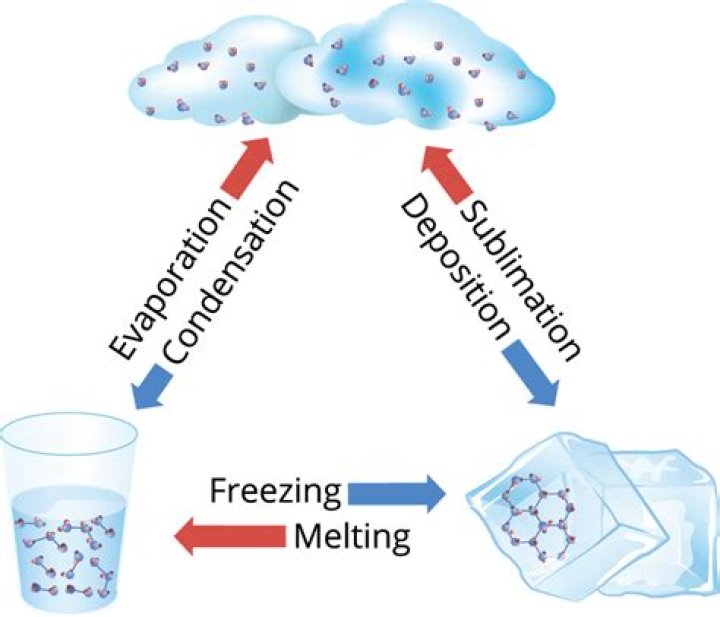
What are four ways that matter can change state?
Changes of state are physical changes in matter. They are reversible changes that do not involve changes in matter’s chemical makeup or chemical properties. Common changes of state include melting, freezing, sublimation, deposition, condensation, and vaporization.



