Does alcohol react with HBr

Nathan Sanders
Published Mar 12, 2026
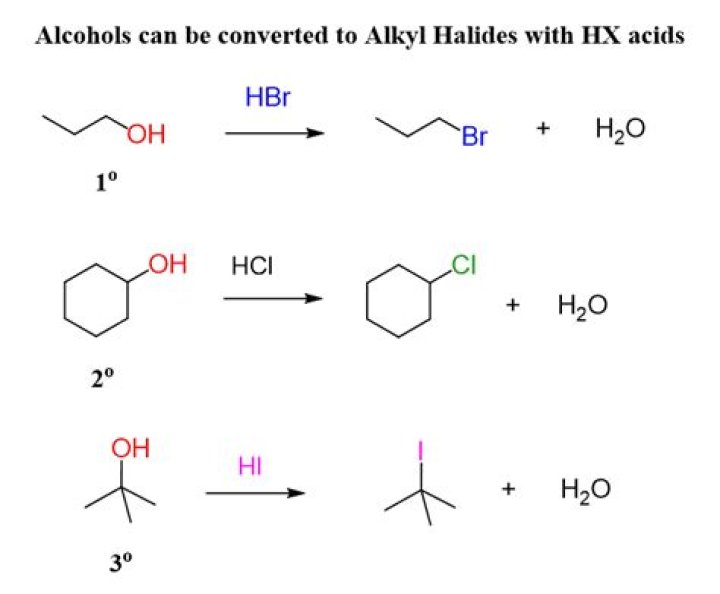
When treated with HBr or HCl alcohols typically undergo a nucleophilic substitution reaction to generate an alkyl halide and water. Alcohol relative reactivity order : 3o > 2o > 1o > methyl. Hydrogen halide reactivity order : HI > HBr > HCl > HF (paralleling acidity order).
What happens when ethanol reacts with HBr?
When treated with HBr or HCl alcohol typically undergo a nucleophilic substitution reaction to generate an alkyl halide and water.
What can alcohol react with?
Alcohols may be oxidized to give ketones, aldehydes, and carboxylic acids. These functional groups are useful for further reactions; for example, ketones and aldehydes can be used in subsequent Grignard reactions, and carboxylic acids can be used for esterification.
Do alcohols react with HCl?
Tertiary alcohols react reasonably rapidly with concentrated hydrochloric acid, but for primary or secondary alcohols the reaction rates are too slow for the reaction to be of much importance. A tertiary alcohol reacts if it is shaken with with concentrated hydrochloric acid at room temperature.Why tertiary alcohol react with HBr faster than secondary alcohol?
so the reactivity is heigher than primary or secondary alcohol. Tertiary alcohols are more reactive because the increased number of alkyl groups increases +I effect. So, the charge density on carbon atom increases and hence around oxygen atom. This negative charge density tryna push the lone pairs on oxygen atom away.
When propene reacts with HBr in presence of peroxide The product formed is?
Reaction of HBr with propene in the presence of peroxide gives n-propyl bromide.
What happens when ethyl alcohol heats PCl3?
The balanced equation shows that the reaction of the reactants, namely ethyl alcohol(C2H5OH) with phosphorus trichloride (PCl3) yields ethyl chloride and phosphorous acid as the two products. 3C2H5OH + PCl3 ——–> 3C2H5Cl + H3PO3.
What is the order of reactivity of HCl HBr and Hi with alcohol?
Order of reactivity of halogen acids towards an alcohol is: HCl > HBr > HI. HBr > HI > HCl.Which alcohol is most reactive with HBr?
2-methyl propane-2-ol is tertiary alcohol and thus, reacts fastest with HBr.
Why HI is more reactive than HBr and HCl?So in this case, H—I has greater bond length than H—Br (due to greater size of I than Br) so it is easy to cleave H—I bond which makes HI more acidic than HBr. HBr and HCl are both polar have similar dipole-dipople forces.
Article first time published onDoes alcohol react with Aluminium?
Anhydrous alcohol is known to react with aluminum at high temperatures, resulting in aluminum alkoxide formation. However the reaction when a mixture of two or more alcohols are used has not been reported. … The reactions were carried out for 72 hours each at 353K, 373K and 393K respectively.
Does alcohol react with metals?
We know that alcohols react with active metals e.g. Na, K, etc. to give corresponding alkoxides. Alcohols contain hydrogen attached to oxygen. Compounds containing hydrogen attached more electronegative elements such as oxygen are acidic. … Sodium metal reacts with alcohol and liberates hydrogen.
Does alcohol react with copper?
The vapours of alcohols react with heated copper at 573 K to form different products depending on whether the alcohol is primary, secondary or tertiary. Reaction of primary alcohols with Cu : Primary alcohols undergo dehydrogenation to form aldehydes.
Which one of the following alcohol reacts readily with a mixture of anhydrous zinc chloride and dilute HCl?
From amongst the following alcohols the one that would react fastest with conc . HCl and anhydrous ZnCl2, is. 1-Butanol.
Which of the following is monohydric alcohol?
An alcohol that has one hydroxyl group is called monohydric; monohydric alcohols include methanol, ethanol, and isopropanol. Glycols have two hydroxyl groups in their molecules and so are dihydric. Glycerol, with three hydroxyl groups, is trihydric.
What is the order of reactivity with HBr?
b > a > c.
How does PCl5 react with C2H5OH?
PCl5 + C2H5OH → POCl3 + C2H5Cl + HCl. Phosphorus(V) chloride react with ethanol to produce phosphoryl chloride, chloroethane and hydrogen chloride.
When a mixture containing PCl3 and PCl5 is heated with?
When a mixture containing PCl_(3) and PCl_(5) is heated with ethyl alcohol, a total of 4 moles of ehtyl chloride is formed.
How are following compounds obtained from alcohols using PCl3?
Step 1: Ethanol reacts with PCl3 to give ethyl Chloride. Step2: Ethyl Chloride reacts with KCN to give Propane nitrile. Step3: Propane nitrile reacts with NaNO2 in the presence of HCl and LiAl4 to give 1-Chloropropane. Step4: 1-Propane reacts with Alcoholic KOH gives Propene.
Which of the following react with HBr in presence of peroxide to give anti-markovnikov product?
(A) Addition of HBr ot alkene in presence of peroxide gives anti-Markownikoff product. (R) This addition follows ionic mechanism .
When propane reacts with HBr in presence of peroxide?
When $HBr$ reacts with propene in the presence of peroxide as catalyst it results in the formation of $n-$ propyl bromide. This is an anti-markovnikov addition reaction. When these free radicals react with hydrogen bromide, it extracts hydrogen from hydrogen bromide and gives bromine radical.
Which of the following alkenes reacts with HBr in the presence of a peroxide to give anti-markovnikov's product?
An alkene C6H12reacts with HBr in the absence as well as in the presence of peroxide to give the same product.
Which of the following alkenes is most reactive towards addition of HBr?
Propyne is more reactive than propene towards electrophilic addition of $$HBr$$
Which alcohol does not give a ketone oxidation?
Primary alcohols are oxidized to form aldehydes. Secondary alcohols are oxidized to form ketones. Tertiary alcohols are not readily oxidized.
What is the order of reactivity of 1 degree 2 degree and 3 degree alcohol with sodium metal?
1o>2o>3o.
What is the order of reactivity of alcohol with halogen acid?
The order of reactivity of alcohols is 3° > 2° > 1° methyl. The order of reactivity of the hydrogen halides is HI > HBr > HCl (HF is generally unreactive).
Which alcohol is more reactive towards halogen acids?
Tertiary alcohols are more reactive towards halogen acids than secondary alcohols.
What is the correct order of reactivity of alcohol in following reaction?
the order of reactivity is tertiary alcohol>secondary alcohol by SN 1 mechanism. With Lucas reagent, tertiary alcohol reacts fastest.
Which alcohol is most reactive towards?
Ethanol because for the dissociation of O-H bond, 10 alcohols are the most reactive.
Why HI is more reactive with alcohol?
Due to lowest bond dissociation energy of HI.
Why HI is most reactive towards alcohol?
Answer: HI has the lowest bond dissociation energy due to longer bond length that’s why it is most reactive.