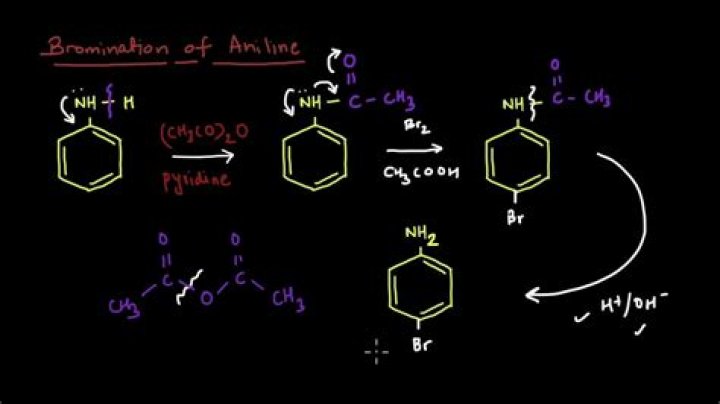
How can bromine behave as an electrophile

Andrew White
Published May 10, 2026
When the Bromine molecule comes close to ethene, it becomes polarised by the negative charge of the double bond. … The positively charged bromine atom acts as an electrophile, reacting with the double carbon bond. A pair of electrons from the carbon double bond move onto the positive bromine atom.
How does bromine become an electrophile?
The electron rich part of the double bond causes a dipole to form meaning that bromine becomes electron deficient and therefore becomes the electrophile (hence electrophillic addition). The double bond attacks the bromine and forms a new carbon – bromine bond, while the other carbon atom becomes electron deficient.
Is Br Br electrophile or nucleophile?
Is it nucleophilic or electrophilic? – Quora. Br2 is an electrophile. When we are taking conditions of free radical like in the presence of sunlight, CCl4, Peroxide. In that case homolytic cleavage will take place and Br free radicals will be produced which are electron deficient and hence electrophile.
Is bromine an electrophile?
Bromine molecule undergo heterolytic cleavage to form Br+ and Br− ions. … Whereas the Br+ ion is very unstable and to attain stability it takes part in chemical reaction. Since Br+ wants to gain electron to attain stability, so it is an. Electrophile (an electron loving specie).Is bromine a gas electrophile?
The electrophilic Br-Br molecule interacts with electron-rich alkene molecule to form a π-complex 1. The alkene is working as an electron donor and bromine as an electrophile. The three-membered bromonium ion 2 consisted of two carbon atoms and a bromine atom forms with a release of Br−.
How does benzene react with bromine?
Benzene reacts with chlorine or bromine in the presence of a catalyst, replacing one of the hydrogen atoms on the ring by a chlorine or bromine atom. … It reacts with some of the chlorine or bromine to form iron(III) chloride, FeCl3, or iron(III) bromide, FeBr3.
How does bromine react with alkenes?
Alkenes react in the cold with pure liquid bromine, or with a solution of bromine in an organic solvent like tetrachloromethane. The double bond breaks, and a bromine atom becomes attached to each carbon. The bromine loses its original red-brown color to give a colorless liquid.
Why is NO2+ described as an electrophile?
An ion is said to be electrophile when it craves for an electron. In NO2+, the nitrogen atom is bonded to one oxygen by the double bond while to the other oxygen atom by the coordinate covalent bond. … The nitrogen in NO2+ does not have an octet around it, hence it is an electrophile.What are the examples of electrophile?
Examples of electrophiles are hydronium ion (H3O+, from Brønsted acids), boron trifluoride (BF3), aluminum chloride (AlCl3), and the halogen molecules fluorine (F2), chlorine (Cl2), bromine (Br2), and iodine (I2). Compare nucleophile.
Why is bromine produced in situ?Since bromine is volatile and highly toxic by inhalation, we will be generating it in situ. When a reagent is formed in situ, this means that instead of adding the reagent directly, it is produced in the reaction mixture.
Article first time published onIs BR a better electrophile than CL?
Although the bromine nucleus is more positively charged than the chlorine nucleus, the increase in the radius and the extra shielding in the bromine atom outweigh this factor, which means that an electron is more easily attracted into the outer shell of a chlorine atom than that of a bromine atom, so chlorine is more …
Is CL an electrophile?
A partial positive charge is gained by the carbon and the chlorine gains a partial negative charge. The electrophile will be the positively charged carbon in this situation. As you can find in halide reactions within organic chemistry, chlorine is also a nucleophile. … Therefore it acts as an electrophile.
Is bromine a good nucleophile?
Under many circumstances, I would agree that bromide is a better nucleophile than water. It’s negatively charged and, due to its size, bromine is more polarizable than oxygen. However, in water, the bromide anion is very stable, reducing its nucleophilicity.
How do alkanes react with bromine?
Alkanes undergo a substitution reaction with halogens in the presence of light. … This reaction is a substitution reaction because one of the hydrogen atoms from the methane is replaced by a bromine atom.
What is a molecule of bromine?
Elemental bromine is a diatomic molecule (Br 2 ). Bromine will combine with most other elements. Reaction with metallic elements leads to salts such as silver bromide (AgBr), in which the bromine atom has a −1 charge and oxidation number.
What does Br2 do in a reaction?
Description: Treatment of alkenes with bromine (Br2) gives vicinal dibromides (1,2-dibromides). Notes: The bromines add to opposite faces of the double bond (“anti addition”). Sometimes the solvent is mentioned in this reaction – a common solvent is carbon tetrachloride (CCl4).
Which type of hydrocarbons will not react with bromine?
Aromatic compounds are not very reactive. They will not react with Br2 or Cl2 under normal conditions. If the aromatic compound has an alkyl substituent, however, the alkyl group can be halogenated in the presence of UV light. The benzene ring will not react under these conditions.
What changes in color occur when bromine reacts with an alkene?
Alkene is an unsaturated compound. Bromine is dark red in color. When bromine is treated with alkene, it loses its dark red color and becomes colorl…
What is reaction mechanism for the bromination of alkene what intermediates are involved?
So in this reaction, the alkene acts as a nucleophile, attacking the electrophilic bromine, giving rise to a 3-membered ring intermediate. This is then attacked from the back side [similar to the backside attack in the SN2] at the carbon best able to stabilize positive charge, to give the trans product.
What is the electrophile in bromination of benzene?
The bromination of benzene is an example of an electrophilic aromatic substitution reaction. In this reaction, the electrophile (bromine) forms a sigma bond to the benzene ring, yielding an intermediate.
Why does bromine not react with benzene?
The six electrons in the π-system above and below the plane of the benzene ring are delocalised over the six carbon atoms, so the electron density is lower. The bromine cannot be polarised sufficiently to react, and the lower electron density does not attract the electrophile so strongly.
What is the electrophile formed by combining br2 and FeBr3?
The electrophile in bromination is the complex of bromine with FeBr3, formed as shown in Eq. 16.4. a resonance-stabilized carbocation intermediate. The electrophile approaches the p-electron cloud of the ring above or below the plane of the molecule.
What are Elecrophiles give example?
Electrophiles are atoms or molecules known to be electron deficient and that carry a partial or fully positive charge and will seek an electron pair to form a covalent bond . An example of an electrophile is a Lewis Acid. Other examples include Br+, Cl+, and CH3+.
Which of the following actors is electrophile?
Electron deficient species or electron acceptors are called electrophiles. Hence, out of the given species, CCl2, has only 6e- with carbon so, it is an electrophile.
What is an electrophile and give an example?
Electrophiles are electron deficient species and can accept an electron pair from electron rich species.Examples include carbocations and carbonyl compounds. A nucleophile is electron rich species and donates electron pairs to electron deficient species. Examples include carbanions, water , ammonia, cyanide ion etc.
Is so3h an electrophile?
Sulphur trioxide is an electrophile because it is a highly polar molecule with a fair amount of positive charge on the sulphur atom. It is this which is attracted to the ring electrons.
Is CH2 CH2 an electrophile?
CH2 act as electrophile as it is short of electrons. … Since CH2 has only 6 electrons that is 2 electron short so show affinity towards electron pair and hence can accept the pair of electrons, so act as an electrophile.
What is name of NO+ electrophile?
The nitrosonium ion is NO+, in which the nitrogen atom is bonded to an oxygen atom with a bond order of 3, and the overall diatomic species bears a positive charge.
What is the main reason to use pyridinium tribromide instead of molecular bromine in this reaction?
Pyridinium tribromide is used in this bromination reaction because it less toxic, more stable, and easier to handle and weigh than elemental bromine.
Why does bromine react with double bonds in alkenes?
This double bond is a region of high electron density. As the bromine molecule approaches the double bond, the identically charged electron regions repel each other, inducing a (temporary) dipole in the bromine molecule (now electron density is unevenly distributed).
Why are bromination reactions important?
Bromination reactions are crucial in today’s chemical industry since the versatility of the formed organobromides makes them suitable building blocks for numerous syntheses. However, the use of the toxic and highly reactive molecular bromine (Br2) makes these brominations very challenging and hazardous.