How can you differentiate the physical states of matter

Lily Fisher
Published May 11, 2026
Solid is the state in which matter maintains a fixed volume and shape; liquid is the state in which matter adapts to the shape of its container but varies only slightly in volume; and gas is the state in which matter expands to occupy the volume and shape of its container.
How do you differentiate phases of matter?
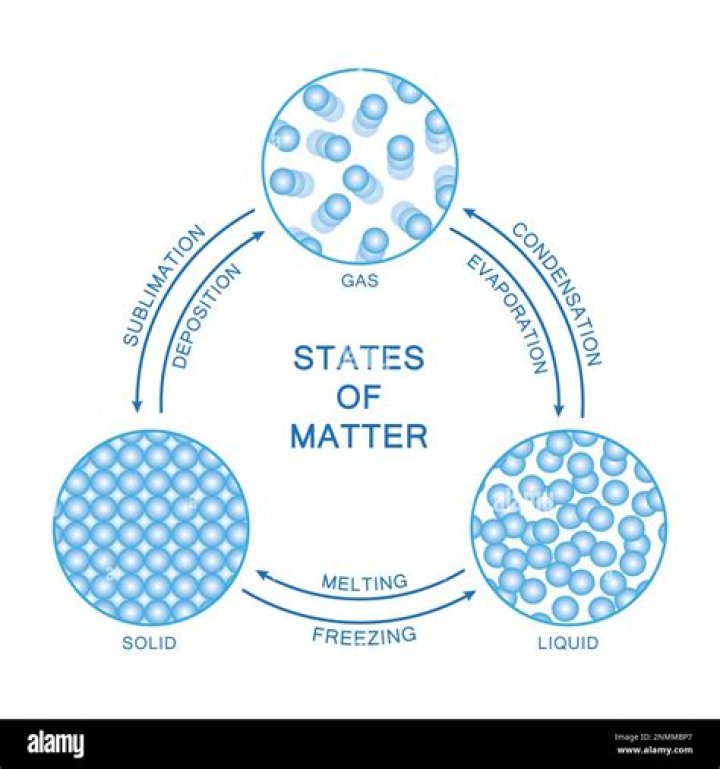
Matter can exist in four phases (or states), solid, liquid, gas, and plasma, plus a few other extreme phases like critical fluids and degenerate gases. Generally, as a solid is heated (or as pressure decreases), it will change to a liquid form, and will eventually become a gas.
Why do the three states of matter differ?
The different states of matter are due to the variation in the characteristics of their constituent particles. The three states of matter, solid, liquid and gas differ in the way their atoms are arranged, their intermolecular distance and the intermolecular force of attraction between their particles.
What are the physical states of matter?
- Solids – relatively rigid, definite volume and shape. In a solid, the atoms and molecules are attached to each other. …
- Liquids – definite volume but able to change shape by flowing. In a liquid, the atoms and molecules are loosely bonded. …
- Gases – no definite volume or shape.
How would you distinguish between a state and a phase of matter is it possible to have items in the same phase but different states or the same state but different phases?
Matter has a mass and a volume that occupy the space. … The main difference between phase of matter and state of matter is that phase of matter explains uniform chemical and physical properties of matter whereas state of matter explains the form of matter at a given temperature and a pressure.
What physical state of matter it has a define shape?
A solid is the state of matter that has a definite shape and volume.
How do you differentiate the arrangement and movement of the particles of matter?
- gas vibrate and move freely at high speeds.
- liquid vibrate, move about, and slide past each other.
- solid vibrate (jiggle) but generally do not move from place to place.
How do you distinguish between solid liquid and gaseous states?
solid: Has a definite shape and volume. liquid: Has a definite volume, but take the shape of the container. gas: Has no definite shape or volume. change of state: When matter is converted from one of the three states (example: solid, liquid, or gas) to another state.How does solid state differ from the other two states?
Answer: Matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place. … Its particles are still close together but move freely. Matter in the gaseous state has both variable volume and shape, adapting both to fit its container.
What determines the state of matter?The amount of energy in molecules of matter determines the state of matter. Matter can exist in one of several different states, including a gas, liquid, or solid state. … A gas is a state of matter in which atoms or molecules have enough energy to move freely.
Article first time published onWhat are the two ways in which the physical state of matter can be changed?
The two main way to change the physical state are by heating and cooling. Explanation: The physical state of matter can be changed from solid to liquid or gas by heating.]
How many different states of matter are there name them?
There are four natural states of matter: Solids, liquids, gases and plasma.
Is there a difference between states of matter and phases of matter?
1. “States of matter” is a more specific and precise term than “phases of matter.” 2. State of matter is the state of a particular compound in a physical system whereas phase is a set of states within such a system.
What is the difference between physical state and phase?
The states of matter (e.g., liquid, solid, gas) are phases, but matter can exist in different phases yet the same state of matter. A phase is a region of space where all physical and chemical properties are uniform. … A state of matter is a form that matter can take. Examples are solid, liquid, gas, plasma.
What is the difference between matter and solid state?
Matter in the solid state maintains a fixed volume and shape, with component particles (atoms, molecules or ions) close together and fixed into place. Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container. Its particles are still close together but move freely.
Why is the state of matter a physical property of matter?
States of matter are examples of physical properties of a substance. … Changes in outward appearances that do not alter the chemical nature of the substance and make no new substance are called physical changes.
How does the arrangement of particles determine the phase of matter?
The looser arrangement of particles is what allows liquids to take on the shape of their container. The intermolecular forces between particles in liquid determine properties like viscosity and surface tension. Particles in a liquid are higher in energy than the solid state, but lower in energy than the gas state.
What is the difference between the movement of particles in solid and gas?
gas vibrate and move freely at high speeds. liquid vibrate, move about, and slide past each other. solid vibrate (jiggle) but generally do not move from place to place.
What is the physical state in which matter has no specific shape but does have a specific volume?
A solid has definite volume and shape, a liquid has a definite volume but no definite shape, and a gas has neither a definite volume nor shape.
What are the characteristics of a solid it has a shape and a definite?
Explanation: A solid has a definite volume and fixed shape. This is in contrast to a liquid, which has a definite volume, but takes the shape of its container and has a flat surface.
What are the differences between solid and liquid?
1. A solid is a state of matter that has a definite shape and volume while a liquid is a state of matter that has volume but no definite shape. 2. A liquid takes the shape of the container that holds it while a solid has a shape of its own.
Why do solids liquids and gases differ in shape and volume class 9?
Solids, liquids, and gases all differ in shape and volumes because each of them have different strenght of interparticle bondings.
Which covalent solid is soft and good conductor?
Graphite is a covalent solid that act as a good conductor of electricity and is soft. The carbon atoms of graphite are prescribed in different layers and are covalently bonded to three of its surrounding atoms in the same layer.
Will you call the arrangement of particles in the solid regular or irregular?
The constituent particles of crystalline solid are arranged in regular pattern which is repeated periodically over the entire crystal. Such type of arrangement is called long range order.
What physical characteristic distinguishes a fluid from a solid?
The physical characteristic that distinguishes a fluid from a solid is that solids have a fixed volume and fixed shape while liquids only have a fixed volume but no fixed shape.
How do you differentiate between solid liquid and gas on the basis of their melting and boiling point?
Solids have high melting and boiling points. Liquids have comparatively low melting and boiling points. Gases have extremely low melting and boiling points.
How can you differentiate between solid liquid and gases on the basis of rigidity?
1. Rigidity: Solid carries Rigidity whereas Liquid and Gas don’t carry Rigidity. … Compressibility: Solids are incompressible, and Liquids are less compressible whereas Gas is highly Compressible.
Which factor mainly influences the physical state of matter explain with examples?
1. Temperature and pressure determine the state of a substance. A change in temperature or pressure will result in the change of state of the substance.
What are the two ways in which the physical state of matter can be changed for class 9?
(a) The two ways by which the physical state of the matter can be changed is either by melting or boiling.
What are the two ways in which the physical state of matter can be changed explain with the help of a ray diagram?
By heating the matter or by cooling it, a solid can be changed to liquid and vice versa. Similarly, by compressing or expanding gas, it can be converted to liquids. Thus, two ways to change the physical states of matter is by changing the temperature and by changing the pressure.
What are some ways that you can change matter?
Now let’s explore these three basic ways that states of matter can be changed: freezing, melting, and boiling.



