How did James Chadwick discovered the neutron

Dylan Hughes
Published Mar 25, 2026
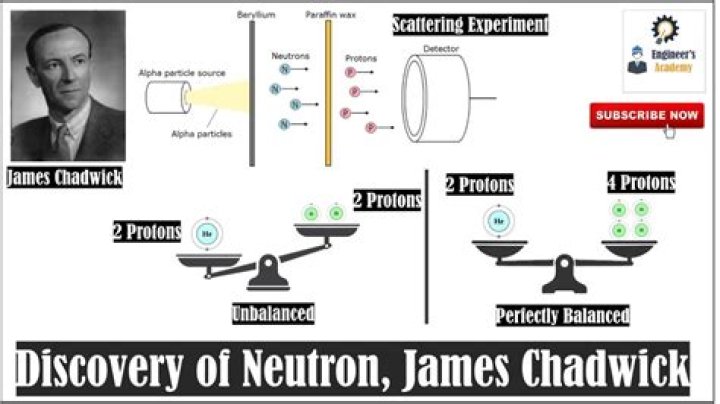
In 1932, the physicist James Chadwick conducted an experiment in which he bombarded Beryllium with alpha particles from the natural radioactive decay of Polonium. The resulting radiation showed high penetration through a lead shield, which could not be explained via the particles known at that time.
How did Chadwick discover his theory?
Chadwick was convinced that alpha particles did not have enough energy to produce such powerful gamma-rays. He performed the beryllium bombardment experiments himself and interpreted that radiation as being composed of particles of mass approximately equal to that of the proton but without electrical charge—neutrons.
Why did James Chadwick make his discovery?
In 1932, Chadwick made a fundamental discovery in the domain of nuclear science: he proved the existence of neutrons – elementary particles devoid of any electrical charge. … Chadwick in this way prepared the way towards the fission of uranium 235 and towards the creation of the atomic bomb.
Where did James Chadwick discover the neutron?
By 1920, physicists knew that most of the mass of the atom was located in a nucleus at its center, and that this central core contained protons. In May 1932 James Chadwick announced that the core also contained a new uncharged particle, which he called the neutron. Chadwick was born in1891 in Manchester, England.What did James Chadwick think the neutron was made of?
He found that the new radiation consisted of not gamma rays, but uncharged particles with about the same mass as the proton. These particles were neutrons. Chadwick won the Nobel Prize in Physics in 1935 for this discovery.
What model did James Chadwick discover?
Chadwick is responsible for discovering the existence of the neutron, the neutral particle that shares the nucleus with the positively charged proton. Chadwick’s discovery forced a revision of the cloud model, and scientists sometimes refer to the revised version as the James Chadwick atomic model.
What was James Chadwick's theory?
Chadwick is best known for his discovery of the neutron in 1932. A neutron is a particle with no electric charge that, along with positively charged protons, makes up an atom’s nucleus. Bombarding elements with neutrons can succeed in penetrating and splitting nuclei, generating an enormous amount of energy.
Did James Chadwick win Nobel Prize?
The Nobel Prize in Physics 1935 was awarded to James Chadwick “for the discovery of the neutron.”Who discovered neutron Class 9?
The British physicist Sir James Chadwick discovered neutrons in the year 1932. He was awarded the Nobel Prize in Physics in the year 1935 for this discovery.
Who disagreed with James Chadwick?He clashed with fellow ACAE member Patrick Blackett, who disagreed with Chadwick’s conviction that Britain needed to acquire its own nuclear weapons; but it was Chadwick’s position that was ultimately adopted.
Article first time published onWhat did James Chadwick think the atom looked like?
Until 1932, the atom was believed to be composed of a positively charged nucleus surrounded by negatively charged electrons. … Chadwick interpreted this radiation as being composed of particles with a neutral electrical charge and the approximate mass of a proton. This particle became known as the neutron.
How did Chadwick prove that the neutron was not charged?
The only good explanation for his result was a neutral particle. To prove that the particle was indeed the neutron, Chadwick measured its mass. He could not weigh it directly. Instead he measured everything else in the collision and used that information to calculate the mass.
Who discovered neutron what is the charge and mass on neutron?
James Chadwick discovered neutron in 1932. Neutron has no charge and mass of a neutron is 1.675×10^(-27).
Who discovered neutron *?
Neutrons are the uncharged, neutral particles present in the nucleus of an atom. The British physicist, James Chadwick discovered neutrons in the year 1932 through the following experiment : James Chadwick fired alpha radiation at a beryllium sheet from a polonium source.
Who discovered proton neutron?
02 Discovery of Electron, Proton and Neutron. Electrons were discovered by J.J. Thomson in 1897. J.
Who discovered cathode rays?
In 1897, British physicist J. J. Thomson showed that cathode rays were composed of a previously unknown negatively charged particle, which was later named the electron.
What did Werner Heisenberg discover?
Werner Heisenberg discovered the uncertainty principle, which states that the position and the momentum of an object cannot both be known exactly.
Did Albert Einstein win a Nobel Prize?
The Nobel Prize in Physics 1921 was awarded to Albert Einstein “for his services to Theoretical Physics, and especially for his discovery of the law of the photoelectric effect.”
Why did James Chadwick use beryllium?
Chadwick felt that the radiation could not be γ rays. The α particles could not provide enough energy to do this. He thought the beryllium rays were neutrons. He bombarded a piece of beryllium in a vacuum chamber with α rays.
How old was Chadwick?
LONDON, July 24—Sir Jamc Chadwick, who won the Nobel Prize for Physics in 1935 fc his discovery three years earlie of the neutron, has died at the age of 82, it was learned the night.
Who gave the quantum model of atom?
Erwin Schrödinger proposed the quantum mechanical model of the atom, which treats electrons as matter waves.
Why does the neutron exist?
Neutrons are required for the stability of nuclei, with the exception of the single-proton hydrogen nucleus. Neutrons are produced copiously in nuclear fission and fusion. They are a primary contributor to the nucleosynthesis of chemical elements within stars through fission, fusion, and neutron capture processes.
What did Niels Bohr discover?
What was Niels Bohr’s most important discovery? Niels Bohr proposed a model of the atom in which the electron was able to occupy only certain orbits around the nucleus. This atomic model was the first to use quantum theory, in that the electrons were limited to specific orbits around the nucleus.
Whose experiments did Chadwick mimic his own after what did he do?
Whos experiments did Chadwick mimic his own after? What did he do? His experiments were similar to those of Jolie-Curies looking at particle radiation. He proved that there was a particle similar in weight to a proton but it didn’t have a charge.
Why was neutron discovery late?
so, it was too easy to find the particles that is charged. … so, these different masses shows , there must be some neutral particles that are also present in the atom , that has the same mass as protons. so from this the neutrons where discovered. and that is the reason why the neutrons were discovered late.
Who discovered neutron Toppr?
James Chadwick concluded the nucleus consists of proton and neutron.
Who discovered neutrons and how class 11?
CBSE NCERT Notes Class 11 Chemistry Structure of Atom. DISCOVERY OF NEUTRON: It was discovered by Chadwick. In which he bombarded some light elements with fast moving alpha particles.