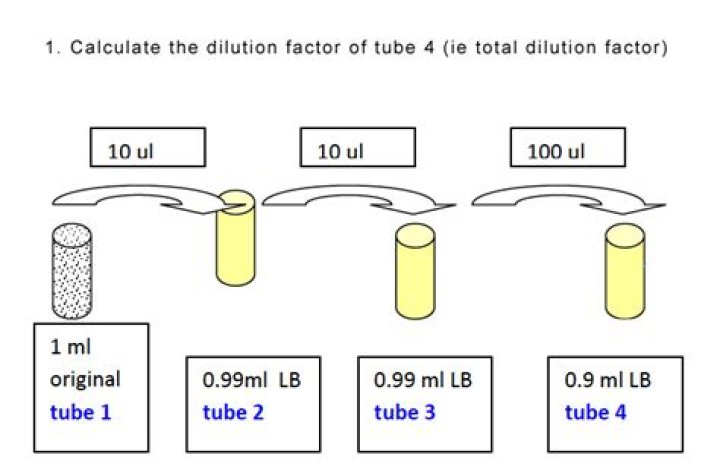
How do you calculate tube dilution

Lily Fisher
Published May 17, 2026
To find a dilution of a single tube, use the formula: sample/(diluent + sample). The sample is the amount you are transferring into the tube, and the diluent is the liquid already in the tube. When you transfer 1 ml into 9 mls, the formula would be: 1/(1+9) = 1/10. This could also be written as 1:10.
What formula do you use to calculate dilution?
Dilution calculations can be performed using the formula M1V1 = M2V2. A serial dilution is a series of stepwise dilutions, where the dilution factor is held constant at each step.
How do you calculate concentration using dilution factor?
The dilution factor is the inverse of the concentration factor. For example, if you take 1 part of a sample and add 9 parts of water (solvent), then you have made a 1:10 dilution; this has a concentration of 1/10th (0.1) of the original and a dilution factor of 10.
How do you calculate dilution in microbiology?
Dilution = amount of specimen transferred divided by the [amount of specimen transferred + amount already in tube]. But after the first tube, each tube is a dilution of the previous dilution tube.How is individual tube dilution factor calculated?
To find a dilution of a single tube, use the formula: sample/(diluent + sample). The sample is the amount you are transferring into the tube, and the diluent is the liquid already in the tube. When you transfer 1 ml into 9 mls, the formula would be: 1/(1+9) = 1/10.
How do I calculate the concentration of a solution?
Divide the mass of the solute by the total volume of the solution. Write out the equation C = m/V, where m is the mass of the solute and V is the total volume of the solution. Plug in the values you found for the mass and volume, and divide them to find the concentration of your solution.
How do you calculate dilution and dilution factor?
For example, a 1:5 dilution (verbalize as “1 to 5” dilution) entails combining 1 unit volume of solute (the material to be diluted) + 4 unit volumes of the solvent medium (hence, 1 + 4 = 5 = dilution factor).
How do you make 500ml of a 1 250 dilution?
Pipet 2.00 mL of your stock solution into a 500 mL volumetric flask. Add diluent to the mark on the flask (you will have added about 498 mL of water). You now have a 1:250 dilution of your original solution.How do you calculate CFU mL?
- To find out the number of CFU/ ml in the original sample, the number of colony forming units on the countable plate is multiplied by 1/FDF. This takes into account all of the dilution of the original sample. …
- 200 CFU x 1/1/4000 = 200 CFU x 4000 = 800000 CFU/ml = 8 x 10.
- CFU/ml in the original sample.
In serial dilutions, you multiply the dilution factors for each step. The dilution factor or the dilution is the initial volume divided by the final volume. For example, if you add a 1 mL sample to 9 mL of diluent to get 10 mL of solution, DF=ViVf = 1mL10mL=110 .
Article first time published onHow do you calculate viable count?
The total number of colonies is referred to as the Total Viable Count (TVC). The unit of measurement is cfu/ml (or colony forming units per milliliter) and relates to the original sample. Calculation of this is a multiple of the counted number of colonies multiplied by the dilution used.
Is CFU mL the same as CFU g?
cfu stands for colony-forming unit. This means that cfu/g is colony-forming unit per gram and cfu/ml is colony-forming unit per millilitre. … They give you the results of the number of colony-forming units, for the number of grams or millilitres of test material that they put on the petri dish.
What is the dilution method?
Dilution is the process of decreasing the concentration of a solute in a solution, usually simply by mixing with more solvent like adding more water to the solution. … If one adds 1 litre of water to this solution, the salt concentration is reduced.
How do you calculate a 1/10 dilution?
For example, to make a 1:10 dilution of a 1M NaCl solution, you would mix one “part” of the 1M solution with nine “parts” of solvent (probably water), for a total of ten “parts.” Therefore, 1:10 dilution means 1 part + 9 parts of water (or other diluent).
How do you calculate the concentration of a dilute solution?
Calculate concentration of solution after dilution: c2 = (c1V1) ÷ V. Calculate the new concentration in mol L-1 (molarity) if enough water is added to 100.00 mL of 0.25 mol L-1 sodium chloride solution to make up 1.5 L.
What are 3 ways to measure the concentration of a solution?
What are three ways to measure the concentration of a solution? Concentration can be expressed as percent by volume, percent by mass, and molarity.
How do you calculate concentration from absorbance and volume?
- Transmission or transmittance (T) = I/I0 …
- Absorbance (A) = log (I0/I) …
- Absorbance (A) = C x L x Ɛ => Concentration (C) = A/(L x Ɛ)
How can you measure the concentration of the solution inside the dropper?
How can you measure the concentration of the solution inside the dropper? Possible Answer: You can drain the water from the tank, add only solution into the tank, and place the concentration sensor into the solution.
How do you calculate CFU g from dilution?
Calculate the number of bacteria (CFU) per milliliter or gram of sample by dividing the number of colonies by the dilution factor The number of colonies per ml reported should reflect the precision of the method and should not include more than two significant figures.
How do you calculate standard plate count?
The standard plate count method consists of diluting a sample with sterile saline or phosphate buffer diluent until the bacteria are dilute enough to count accurately. That is, the final plates in the series should have between 30 and 300 colonies.
How do you do a 20x dilution?
A 20-fold dilution just means the final solution is 20 times less concentrated than the original. An easy way to perform this is to take say 5 mL of your original acid using a pipette, transfer it to a 100 mL volumetric flask and then fill to the mark with distilled or RO water.
How do you do a 1 200 dilution?
200 (1/200 dilution) = the first step in the dilution factor; the second is 1/50, obtained as follows: 1 ml of serum + 199 ml of diluent = 1/200 dilution. 1 ml of 1/200 dilution + 49 ml of diluent = 1/50.
How do you do a 1 1000 dilution?
You could make 1/1,000 by adding 1 microliter of sample to 0.999 ml diluent. Why is that a poor choice? Because you can’t measure 1 microliter (or even 10 microliters) accurately with ordinary pipeters. So, make three serial 1/10 dilutions (0.1 ml [100 microliters] into 0.9 ml): 1/10 x 1/10 x 1/10 = 1/1,000.
How do you calculate concentration from dilution factor and absorbance?
A. take the absorbance of sample (X) minus blank absorbance (Y) then multiply with the dilution factor (DF) and to get the concentration using the calibration curve. B. the absorbance of sample (X) multiplied by the DF then minus blank absorbance to get the concentration using the calibration curve.
What is the total dilution of tube 5 in this serial dilution scheme?
What is the total dilution of tube 5 in this serial dilution scheme? The dilution of 1 ml from tube 4 into 9 ml of tube 5 is 10-1. To calculate the total dilution of tube 5, multiply this dilution by the total dilution of tube 4, which is 10-4. A plate count method using serial diluted sample.
How do you dilute in math?
The dilution factor is the factor by which you will dilute your sample at each step. Mathematically it is equal to the sum of the volume of your stock solution you are adding and the volume of the solvent you are making the dilution in, divided by the volume of your stock solution.
What is a high TVC count in water?
A low TVC level indicates an overall low level of bacteria in the system, whereas a much higher TVC level – for example, 10,000 cfu (colony forming units) per ml of water – indicates that the system may be suffering from biofilm contamination.
How do you calculate CFU bacteria?
Calculate the number of bacteria (CFU) per milliliter or gram of sample by dividing the number of colonies by the dilution factor The number of colonies per ml reported should reflect the precision of the method and should not include more than two significant figures.
What does a dilution factor of 2 mean?
A two-fold dilution reduces the concentration of a solution by a factor of two that is reduces the original concentration by one half. A series of two-fold dilutions is described as two-fold serial dilutions. In this manual, two-fold serial dilutions are carried out in small volumes in microwell plates.
What does 1 CFU mean?
CFU is colony forming unit that may be anything like bacteria, fungus or yeasts. … Suppose if you had exposed 5 plates and each had 1 CFU then the average will be 1 and according to the specification area fails in the viable count. The average of all exposed plates should be less than 1.