How do you rank an element using ionization energy

Sarah Rodriguez
Published Apr 29, 2026
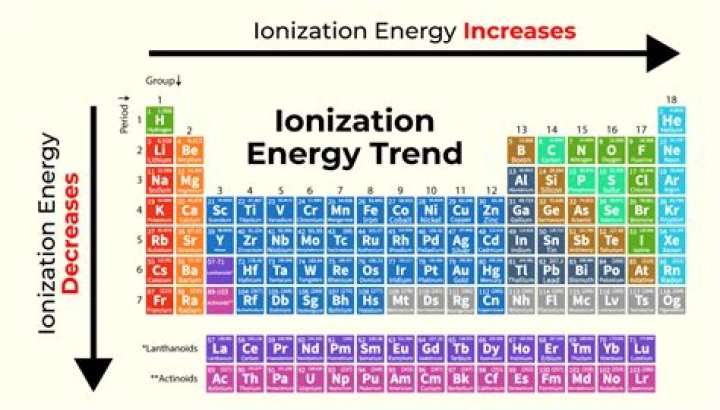
The first ionization energy varies in a predictable way across the periodic table. The ionization energy decreases from top to bottom in groups, and increases from left to right across a period. Thus, helium has the largest first ionization energy, while francium has one of the lowest.
Which element has the highest and lowest ionization energy?
It is because of the shielding effect that the ionization energy decreases from top to bottom within a group. From this trend, Cesium is said to have the lowest ionization energy and Fluorine is said to have the highest ionization energy (with the exception of Helium and Neon).
What elements have the highest ionization energy?
The elements that belong to the noble gases or inert gases or (Group VIII-A) have the highest ionisation energy. If we were to take a single element then Helium is said to have the highest first ionization energy among all the other neutral elements. The ionization potential is said to be 24.5874 eV.
How does the ionization energy of first group elements vary?
Moving left to right within a period or upward within a group, the first ionization energy generally increases. … Ionization energy increases from left to right in a period and decreases from top to bottom in a group.What element has the lowest ionization energy on the periodic table?
Fr (francium) has the lowest ionization energy. its radius is large and there is only in one electron in last energy level. therefore less amount of energy is needed to remove that electron.
How does ionization energy change with atomic number?
Within a group, the ionization energy decreases as the size of the atom gets larger. … In this situation, the first electron removed is farther from the nucleus as the atomic number (number of protons) increases. Being farther away from the positive attraction makes it easier for that electron to be pulled off.
Which group has the highest ionization energy?
Noble Gases have a very stable electron configuration, therefore, they have the highest amount of ionization energy within their periods.
How does the ionization energy vary across a period and down a group?
Ionization energy (IE) is the energy required to remove the highest-energy electron from a neutral atom. In general, ionization energy increases across a period and decreases down a group. … Down a group, the number of energy levels (n) increase and the distance is greater between the nucleus and highest-energy electron.How does ionization vary in a group?
Variation of ionization energy down the group: On moving down the group, the ionization enthalpy decreases. … Screening due to core electrons goes on increasing and the effective nuclear charge decreases down the group. As a result, the removal of the outer electron becomes easier down the group.
How does ionization energy vary for alkali metals?Ionization energy increases from bottom to top within a group, and increases from left to right within a period. The trend within a group can be seen easily by observing how the ionization energies vary for either the alkali metals (Li through Cs) or the noble gases (He through Rn).
Article first time published onWhich of these elements has the highest first ionization energy SN as or S?
The given elements in the choices are Sn, S, Si, and F. Among all the given elements, F has the smallest size due to which the removal of an electron from the outermost shell of the F-atom is difficult. As a result of this fluorine has the highest ionization energy.
Which element has the highest ionization energy in period 3?
ElementSymbolFirst ionisation energy /kJ mol–1phosphorusP1012sulfurS1000chlorineCl1251argonAr1521
Why does he have the highest ionization energy?
Re: Ionization Energies Yes, Helium has the highest ionization energy! This is because the electrons in helium are very close to the nucleus and so the electrostatic attraction is very high. This makes it difficult to remove an electron.
What is the ionization energy of as?
Ionization EnergyName chemical elementSymbol5,1391SodiumNa5,17ActiniumAc5,2117BariumBa5,2784RadiumRa
Why do metals have low ionization energy?
Encyclopædia Britannica, Inc. Metal atoms lose electrons to nonmetal atoms because metals typically have relatively low ionization energies. Metals at the bottom of a group lose electrons more easily than those at the top. That is, ionization energies tend to decrease in going from the top to the bottom of a group.
What is ionization energy trend?
Ionization energy is a periodic trend which increases going from top to bottom and left to right across the periodic table. … So ionization energy increases as successive energy electrons are removed. So you can have a first ionization energy, second, third, fourth and so forth and so on.
Which family of elements has the lowest ionization energy?
Since the nuclear charge is necessarily diminished with respect to the valence shell, the alkali metals display the lowest ionization energies, and these energies (reasonably) decrease down the Group.
Why do Group 1 elements have the lowest first ionization energy?
As the last electron of the group 1 elements is loosely attached to the nucleus, it’s easy to remove the electron from the atom. That is why the I.E. of the group 1 elements are low. … So the nucleus is not heavily able to attract the last electron As a result, the ionization becomes easier and it needs less energy.
How does electron configuration affect ionization energy?
ionization energy increases with increasing numbers of protons in the nucleus, that is, with increasing positive nuclear charge. The distance of the electron from the nucleus: … Repulsion between electrons : ionization energy decreases if 2 negatively charged electrons are in close proximity to each other.
How does ionization energy change as you move from top to bottom in a group explain your answer in terms of electron shells?
They experience a weaker attraction to the positive charge of the nucleus. Ionization energy increases from left to right in a period and decreases from top to bottom in a group.
What are the factors on which ionization energy depends?
- The force of attraction between electrons and the nucleus.
- The force of repulsion between electrons.
What is ionization enthalpy How does it vary in periodic table?
The amount of energy required to remove an electron from an isolated gaseous atom in the ground state is called ionization enthalpy. Ionization enthalpy increases along the period. Reason: This is because along the period atomic size decreases and effective nuclear charge increases.
What is ionization energy name the factors on which it depends how does it vary down the group and across a period?
ln general, the ionisation energy decreases down the group due to increase in atomic size. On the other hand, the ionisation energy increases across the period from left to right, again due to decrease in atomic size from left to right.
How does ionization energy vary on going down in Group 13?
These elements are found in Group 13 (XIII) of the p block in the Periodic Table of Elements. … Ionization Energy decreases going down the group (because the electrons are farther from the core and therefore are easier to remove; Tl does not fit this trend)*
How does ionization energy increase across a period?
On the periodic table, first ionization energy generally increases as you move left to right across a period. This is due to increasing nuclear charge, which results in the outermost electron being more strongly bound to the nucleus.
How does ionization energy change from top to bottom within a group?
The ionization energy decreases from top to bottom in groups, and increases from left to right across a period. … Since the outermost electrons are further away, they are less strongly attracted by the nucleus, and are easier to remove, corresponding to a lower value for the first ionization energy.
How does ionization energy impacts ionic bonding?
In general, elements that have lower ionization energies have a greater chance to form a cation, thereby having a greater tendency to form ionic bonds. Ionization energy decreases down a group in the periodic table due to the fact that the outermost electrons are further away from the nucleus.
Which has a higher ionization energy in or Sn?
Ionization energy is the energy required to remove an electron from an element. The general trend for ionization energy is that it increases as we go up and right in the periodic table. Iodine is in the same row as tin, but it is more to the right.
For which of these elements would the first ionization energy of the atom be higher than that of the diatomic molecule?
Helium is the element that must be having the higher first ionization energy than that of the diatomic molecule.
Why ionization energy of lead is higher than tin?
The first ionization energy of lead is actually higher than that of tin rather than lower. … The 4f electrons screen the nucleus rather inefficiently from 6p electrons causing the effective nuclear charge to be quite high, to the extent that the ionization energy for lead is actually a little higher than that of tin.
Which is the correct order of ionisation energy?
So the order of ionisation energy is F>F− and Cl>Cl−. Since the ionization energy decreases on going down the group, the ionization energy order is F>Cl.



