How do you solve a serial dilution problem

Isabella Wilson
Published Apr 13, 2026
In serial dilutions, you multiply the dilution factors for each step. The dilution factor or the dilution is the initial volume divided by the final volume. For example, if you add a 1 mL sample to 9 mL of diluent to get 10 mL of solution, DF=ViVf = 1mL10mL=110 .
How do you find the concentration of a serial dilution?
A ten-fold serial dilution, which can also be called a 1:10 dilution, or a series with dilution factor of 10. To determine the concentration at each step of the series, you divide the previous concentration by the dilution factor.
What is a serial solution?
A serial dilution is the stepwise dilution of a substance in solution. Usually the dilution factor at each step is constant, resulting in a geometric progression of the concentration in a logarithmic fashion.
How do you calculate the dilution factor of a solution?
Dilution factor is defined as: total volume of solution per aliquot volume. Where total volume of solution is: 10.0 + 240.0 = 250.0 mL (volumetric flask.) Note: For multiple dilutions the dilution factor is the product of the dilution factors for each individual dilution.What is the aim of serial dilution?
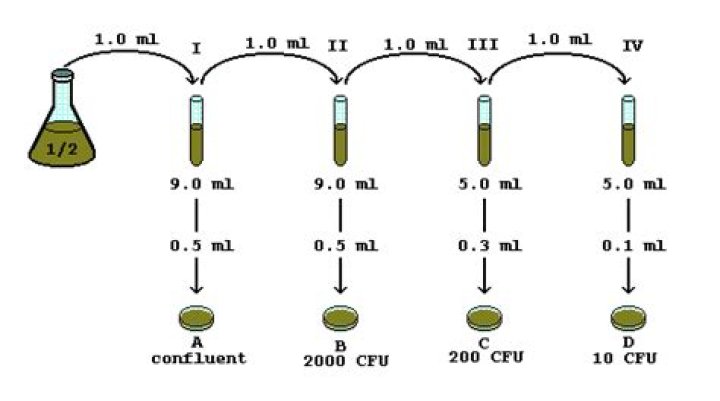
The objective of the serial dilution method is to estimate the concentration (number of colonies, organisms, bacteria, or viruses) of an unknown sample by counting the number of colonies cultured from serial dilutions of the sample, and then back track the measured counts to the unknown concentration.
Why is serial dilution usually performed with a power of 10 dilutions?
Objectives of Serial Dilution Serial dilutions are commonly performed to avoid having to pipette very small volumes (1-10 µl) to make a dilution of a solution.
How do you do a 1/5 serial dilution?
Answer: 1:5 dilution = 1/5 dilution = 1 part sample and 4 parts diluent in a total of 5 parts. If you need 10 ml, final volume, then you need 1/5 of 10 ml = 2 ml sample. To bring this 2 ml sample up to a total volume of 10 ml, you must add 10 ml – 2 ml = 8 ml diluent.
What is serial dilution procedure?
A serial dilution is a series of sequential dilutions used to reduce a dense culture of cells to a more usable concentration. … In this method, exactly 1 ml of each successive dilution is transferred into exactly 9 ml of liquid in a dilution blank, creating a 1/10 dilution.What is a 5 fold serial dilution?
“It was a 5 fold dilution” “It was diluted 1/5” These all mean the same thing, that there is 1 volume part of sample and 4 volume parts of whatever liquid is being used to dilute the sample for a total of 5 volume parts.
Why are serial dilutions more accurate?The more evenly spaced the calibration standards are over this range, makes the results of the analysis more reliable. … Each successive standard uses a small portion of the previous standard, which is diluted by solvent to generate the next calibration standard in the series.
Article first time published onHow do you dilute 10m to 1M?
Take 1 part of your stock solution and add 9 parts of solvent (usually water but sometimes alcohol or other organic solvent). In all cases you are diluting by the same factor. The concentration of the resulting solution is 1M /10 = 0.1M where 10 is the dilution factor.
How do you do a 1/10 dilution?
For example, to make a 1:10 dilution of a 1M NaCl solution, you would mix one “part” of the 1M solution with nine “parts” of solvent (probably water), for a total of ten “parts.” Therefore, 1:10 dilution means 1 part + 9 parts of water (or other diluent).
How do you make a 1 200 dilution?
1 ml of serum + 199 ml of diluent = 1/200 dilution.
Why do we do serial dilution in Elisa?
Dilution is a vital parameter for ELISA experiment which in turn determines the values of detection range for antibody and target antigen concentrations.
What is serial dilution PDF?
It is a method of diluting a stock solution where concentration decreases by the same quantity in each successive step. Materials required: stock solution, test tubes, pipettes, beaker, and distilled water.
What is serial dilution in microbiology?
In microbiology, serial dilutions (log dilutions) are used to decrease a bacterial concentration to a required concentration for a specific test method, or to a concentration which is easier to count when plated to an agar plate.
What does dilution 10 fold mean?
A ten-fold dilution reduces the concentration of a solution or a suspension of virus by a factor of ten that is to one-tenth the original concentration. A series of ten-fold dilutions is described as ten-fold serial dilutions.
How do you do a 1/20 dilution?
For example, a 1:20 dilution converts to a 1/20 dilution factor. Multiply the final desired volume by the dilution factor to determine the needed volume of the stock solution. In our example, 30 mL x 1 ÷ 20 = 1.5 mL of stock solution.
How do you make a serial dilution for Qpcr?
You can start by taking 5 ml of template / DNA extraction and mix with 45 ml sterile water. Mix them thoroughly bu pipetting several times. It’s the 10-1 dilution. Then, take 5 ml of this mixture and add to the second tube containing 45 ml water.
What is the main disadvantage of the serial dilution technique?
The advantage of the serial dilution-agar plat-ing procedure is that the cell count represents only viable cells. The disadvantage of this method is that it requires an incubation period that precludes the ability to obtain immediate results.



