How is alpha used in smoke alarms

Andrew White
Published Mar 21, 2026
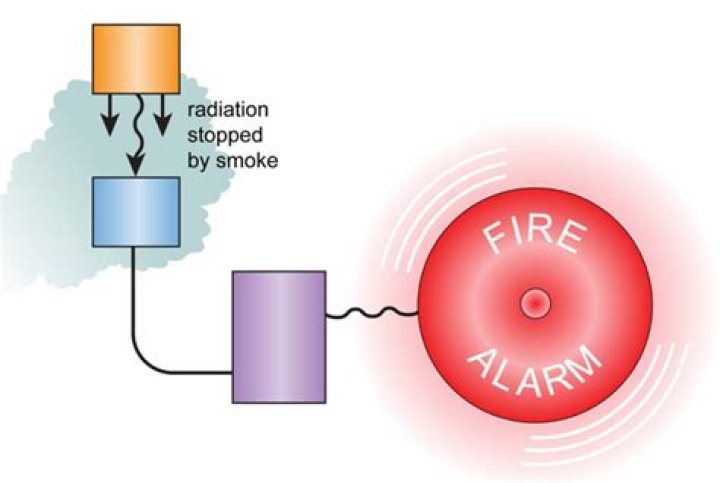
An isotope of americium which emits alpha particles is used in smoke alarms. Alpha radiation ionises the air and this allows a small current to flow between two electrodes. Alpha is weakly penetrating so smoke stops it, the current drops and the alarm goes off.
Why does a smoke detector use alpha radiation rather than beta or gamma?
The main reason that alpha particles are used is that they are strongly ionizing, because they are helium nuclei without any orbiting electrons; they will rip electrons off of any particles that they encounter, whereas gamma rays and beta particles will not.
How is Ionising radiation used in smoke detectors?
Ionization chamber smoke detectors contain a small amount of americium-241, a radioactive material. Smoke particles disrupt the low, steady electrical current produced by radioactive particles and trigger the detector’s alarm. They react quickly to fires that give off little smoke.
How are isotopes used in smoke detectors?
The Am-241 emits alpha particles which ionise the air and allow a current between two electrodes. If smoke enters the detector it absorbs the alpha particles and interrupts the current, setting off the alarm.What are the uses of alpha radiation?
Alpha radiation is used to treat various forms of cancer. This process, called unsealed source radiotherapy, involves inserting tiny amounts of radium-226 into cancerous masses. The alpha particles destroy cancer cells but lack the penetrating ability to damage the surrounding healthy cells.
Why is using an alpha emitter in a smoke alarm safe to use in a house?
An alpha source is used because alpha radiation does not penetrate very far. It is absorbed by a few cm of air. This means that as long as the detector is high up on a wall, or the ceiling, it is safe for humans to be in the same room.
How do smoke detectors detect smoke?
Photoelectric alarms work using a photoelectric sensor and a light source. As smoke enters the chamber and crosses the path of the light beam, light is scattered by the smoke particles, aiming it toward the sensor, which in turn triggers the alarm.
How is nuclear technology used in smoke detectors?
Smoke detectors are the most common consumer product that uses nuclear technology. They work by putting a source of alpha radiation, usually americium-241, near an ion detector. … The electric charge is detectable by the device and, if there is enough smoke in the air, the charge drops, and the detector sounds an alert.Why is americium 242 not used in smoke detectors?
(i) The smoke detector would not work if a radioactive source that emitted only gamma rays was used. (ii) Curium-242 is a radioactive isotope with a half-life of 160 days. It emits alpha particles.
How are isotopes used in climatology and geology?Isotopes in climatology and geology are used to date back rocks, meteorites and archaeological objects, they also use them to discover environmental processes and they can determine the chemical elements in our world. … using the tools able to determine the elements and isotopes.
Article first time published onAre gamma rays used in smoke detectors?
Most smoke detectors use americium-241 as their source. … Am-241 and Ra-226 also emit gamma radiation in an amount so low it cannot be distinguished from natural radiation from space and the earth.
What is a use of beta radiation?
Beta radiation is used for tracers and monitoring the thickness of materials. Doctors may use radioactive chemicals called tracers for medical imaging. … Radiation is used in industry in detectors that monitor and control the thickness of materials such as paper, plastic and aluminium.
What do the alpha particles do to the oxygen and nitrogen atoms in the chamber of a smoke detector?
Radioactive isotopes which emit alpha particles can be used in smoke detectors. … The alpha particles emitted from the Americium-241 collide with the oxygen and nitrogen molecules in the air causing them to ionise. To ionise means to knock off electron/electrons from an atom.
Which type of radiation is used in smoke alarms?
Ionization smoke detectors use americium as a source of alpha particles. Alpha particles from the americium source ionize air molecules. This makes some particles positively charged and some negatively charged.
How does alpha radiation work?
An alpha particle is produced by the alpha decay of a radioactive nucleus. Because the nucleus is unstable a piece of it is ejected, allowing the nucleus to reach a more stable state. … An interesting thing about alpha particles is that they do not penetrate far through matter (unlike beta or gamma radiation).
What are alpha beta and gamma rays used for?
The radioactive decay products we will discuss here are alpha, beta, and gamma, ordered by their ability to penetrate matter. Alpha denotes the largest particle, and it penetrates the least. Alpha particles carry a positive charge, beta particles carry a negative charge, and gamma rays are neutral.
What are the 3 types of smoke detectors?
There are three types of smoke alarms, ionization, photoelectric and a combination of the two which is commonly called a “dual” detector.
Do smoke detectors detect smoke or heat?
A smoke detector will only detect heat if that is an included function of the device. There is no rule that a smoke detector must also detect high temperatures. … A monitored smoke is a smoke detector that is connected with an alarm system in some way.
What is better photoelectric or ionization?
Ionization smoke alarms tend to respond faster to the smoke produced by flaming fires than photoelectric smoke alarms. Photoelectric smoke alarms tend to respond faster to the smoke produced by smoldering fires than ionization smoke alarms.
Why are ionization smoke detectors banned?
Whilst perfectly safe to residents, ionisation fire alarms are banned in several countries as they contain small quantities of radioactive material (Americium 241) meaning there are storage and disposal issues.
What is the daughter isotope?
In nuclear physics, a decay product (also known as a daughter product, daughter isotope, radio-daughter, or daughter nuclide) is the remaining nuclide left over from radioactive decay.
Is there uranium in smoke detectors?
The isotope of americium used in smoke detectors is americium-241, which decays by α emission to neptunium-237 with a half-life of 432.2 years. Plutonium-239 is itself the primary fissile isotope of plutonium (used in nuclear weapons), and is in turn made from uranium. …
How many isotopes does americium have?
About 19 isotopes and 8 nuclear isomers are known for americium. There are two long-lived alpha-emitters, 241Am and 243Am with half-lives of 432.2 and 7,370 years, respectively, and the nuclear isomer 242Am has a half-life of 141 years.
Are all smoke detectors radioactive?
Almost all smoke-detectors will use a radioactive substance called americium-241. If you live in an older house, your detector might be equipped with radium-226 or perhaps nickel-63.
What type of radioactive decay is used in smoke detectors apex?
Americium-241 is used in a number of smoke detectors; as this radioisotope decays it causes particles to ionize, conduct an electric current and thereby cause the alarm to buzz.
How are isotopes used to make nuclear power?
In a nuclear reactor the uranium fuel is assembled in such a way that a controlled fission chain reaction can be achieved. The heat created by splitting the U-235 atoms is then used to make steam which spins a turbine to drive a generator, producing electricity.
How are isotopes used in environment?
Radioisotopes are used to determine the age of water, whilst stable isotopes can be used to determine the source’s history, rainfall conditions, mixing/interaction characteristics of related water bodies, pollution processes, and evaporation processes.
How do climatologist use isotopes?
The isotopes found in the core of glaciers and ice caps are used to determine factors such as as temperature, precipitation rate, wind speed, and greenhouse gas concentration over extremely long periods.
How are isotopes used in geology?
Denver Radiogenic Isotope Lab Radiogenic isotopes are daughter nuclides derived from natural decay of radioactive parent nuclides. They are useful for determining radiometric ages of rocks, minerals, and soils, and can be used as natural tracers of lithologic, hydrologic, and biologic materials.
Why is beta not used in smoke alarms?
the _______________ number. (c) An atom of iodine-131 decays into an atom of xenon (Xe) by emitting a beta particle.
What type of electromagnetic waves are used in security alarms?
Infrared (IR) light is used by electrical heaters, cookers for cooking food, short-range communications like remote controls, optical fibres, security systems and thermal imaging cameras which detect people in the dark.