How is an anion formed from a neutral atom

Olivia Owen
Published Mar 28, 2026
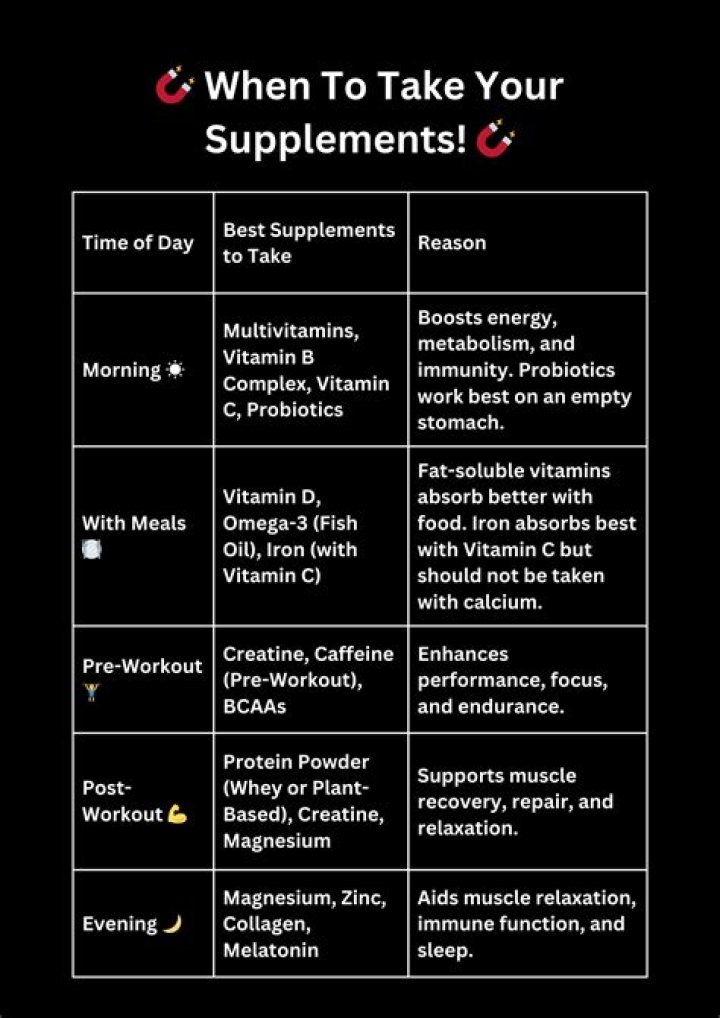
Anions. Anions are the negative ions formed from the gain of one or more electrons. When nonmetal atoms gain elections, they often do so until their outermost principal energy level achieves an octet.
How does a neutral atom become an anion?
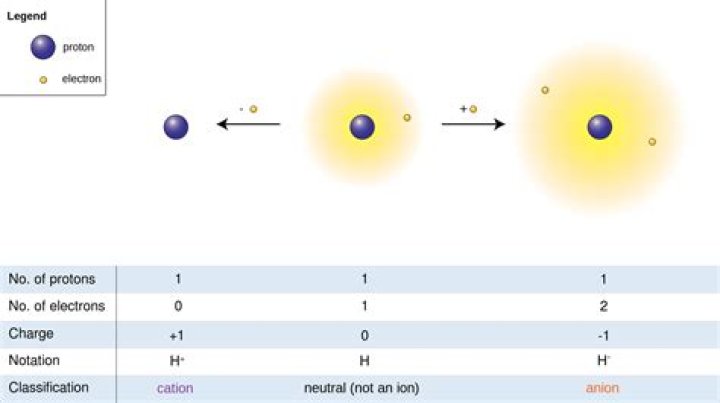
Cations and anions When a neutral atom gains one or more electrons, the number of electrons increases while the number of protons in the nucleus remains the same. The result is that the atom becomes an anion—an ion with a net negative charge.
How is a cation formed from a neutral atom?
A cation (a positive ion) forms when a neutral atom loses one or more electrons from its valence shell, and an anion (a negative ion) forms when a neutral atom gains one or more electrons in its valence shell.
How are anions formed?
An anion has more electrons than protons, consequently giving it a net negative charge. For an anion to form, one or more electrons must be gained, typically pulled away from other atoms with a weaker affinity for them.What are ions and how are they formed?
Ions are formed by the addition of electrons to, or the removal of electrons from, neutral atoms or molecules or other ions; by combination of ions with other particles; or by rupture of a covalent bond between two atoms in such a way that both of the electrons of the bond are left in association with one of the …
What happens when an anion is formed?
An anion is an ion with negative charge, meaning it has more electrons than protons. Anions are formed when an atom gains one or more electrons: the gain of the negatively-charged electron(s) results in an overall negative charge.
How an ion is different from an atom?
Explanation: An atom is neutral when it has equal numbers of protons and electrons. An ion is an atom that is no longer neutral because it has either lost or gained one or more electrons. When an atom loses one or more electrons, it becomes a positively charged ion, called a cation .
How are ionic compounds formed?
An ionic bond is formed by the complete transfer of some electrons from one atom to another. The atom losing one or more electrons becomes a cation—a positively charged ion. The atom gaining one or more electron becomes an anion—a negatively charged ion.What is anion in chemistry?
anion, atom or group of atoms carrying a negative electric charge. See ion.
How are ions formed quizlet?Ions are formed when atoms lose or gain electrons in order to fulfill the octet rule and have full outer valence electron shells.
Article first time published onHow are ions formed in the environment?
Formation of monatomic ions Monatomic ions are formed by the gain or loss of electrons to the valence shell (the outer-most electron shell) in an atom.
What is anion example?
Anions are negatively charged ions. They are formed when non-metal gains the electrons. They gain one or more than one electron and do not lose any protons. Therefore, they possess a net negative charge. Some examples of anions are Iodide (I–), chlorine (Cl–), hydroxide (OH–).
What distinguishes a neutral atom from an ion?
What distinguishes a neutral atom from an ion? The charge of the atom. A neutral atom has no charge because there is a balance between the positively charged protons and negatively charged electrons.
What is neutral atom?
When an atom has an equal number of electrons and protons, it has an equal number of negative electric charges (the electrons) and positive electric charges (the protons). The total electric charge of the atom is therefore zero and the atom is said to be neutral.
What atom forms a ion?
Ions. Atoms are neutral; they contain the same number of protons as electrons. By definition, an ion is an electrically charged particle produced by either removing electrons from a neutral atom to give a positive ion or adding electrons to a neutral atom to give a negative ion.
When an anion formed from an element it usually has the same?
This is produced when an atom gains one or more valence electrons. When one of these is formed from a nonmetallic element, the ending of the element’s name is typically changed to -ide. Nonmetals gain electrons and their anions have the same amount of negative charge as the number of electrons the nonmetal gained.
How are anions used?
Anions are negatively charged ions, and are formed from atoms or molecules that have more electrons than protons. Anions often combine with cations to make salts, which are important in the human body. These particles play a role in many vital biological processes, from hormone production to DNA formation.
How is anion formed in short?
Anions are the negative ions formed from the gain of one or more electrons. When nonmetal atoms gain electrons, they often do so until their outermost principal energy level achieves an octet. … Under typical conditions, three electrons is the maximum that will be gained in the formation of anions.
What is anion in biology?
A ion attracted to the anode of an electrolytic cell, which being positively charged, attracts anions, which are, therefore, negatively charged. Similarly, a cation, which is attracted to the negatively charged cathode, is positively charged.
Are metals cation or anion?
Alkali metals and alkaline earth metals always form cations. Halogens always form anions. Most other nonmetals typically form anions (e.g. oxygen, nitrogen, sulfur), while most metals form cations (e.g. iron, gold, mercury).
How are ionic compounds formed 10?
When a metal reacts with a non-metal, then they form ionic bond and the compound is called the ionic compound. As a result of reaction between metal and non-metal, they are bonded with electrostatic force of attraction with each other; such bonds are called chemical bonds.
Which ions forms the strongest ionic bond with a given anion?
A cation with a 2+ charge will make a stronger ionic bond than a cation with a 1+ charge. A larger ion makes a weaker ionic bond because of the greater distance between its electrons and the nucleus of the oppositely charged ion.
Why are electrons transferred in ionic bonds?
It is the power of an atom in a molecule to attract electrons to itself. When the electronegativity difference between the two atoms is greater than 2.0, the bond is considered ionic; i.e. there is complete transfer (between the two atoms) of the electrons in the bond.
Which of the following can change into a anion?
Among the given elements, the element that can change to an anion is iodine. Explanation: Metals have loosely bound electrons. They can lose one or more electrons to change into positive ions called cations.
Why do anions form quizlet?
How do anions form? The gain of negatively charged electrons by a neutral atom produces an anion.
How do ions form ionic bonds quizlet?
An ionic bond is the force of attraction that holds together oppositely charged ions. It forms when atoms of a metal transfer electrons to atoms of a nonmetal. When this happens, the atoms become oppositely charged ions. … As a result, ionic compounds are brittle solids with high melting and boiling points.
What changes when an ion is formed from an atom quizlet?
An atom becomes an ion when its number of protons change. Some ions are positively charged, and some ions have no charge. The formation of an ion results in a full outermost energy level. Ions usually form when electrons are transferred from one atom to another.
Why do atoms form ions?
When ions form atoms gain or lose electrons until an outer energy level without full. Ions are formed when atoms lose or gain electrons in pack to fulfill the octet rule some have any outer valence electron shells When they lose. … Ion Definition Chemistry Examples & Facts Britannica.
What type of ion forms when an atom loses an electron?
If atoms lose electrons, they become positive ions, or cations. If atoms gain electrons, they become negative ions, or anions.
What is the difference between cation and anion how are they formed?
A positively charged ion is known as cation. A cation is formed by the loss of one or more electrons by an atom. … An anion is formed by the gain of one or more electrons by an atom. For example: A chlorine atom gains (accepts) 1 electron to form a chloride ion, Cl – , which is an anion.
What is anion explanation?
An anion is an ionic species with a negative charge. This is a type of atom that has gained electrons. Anions are one of the two types of ions. The other type is called a cation. Anions are attracted to the anode, while cations are attracted to the cathode.