How is latent heat measured

Nathan Sanders
Published Mar 23, 2026
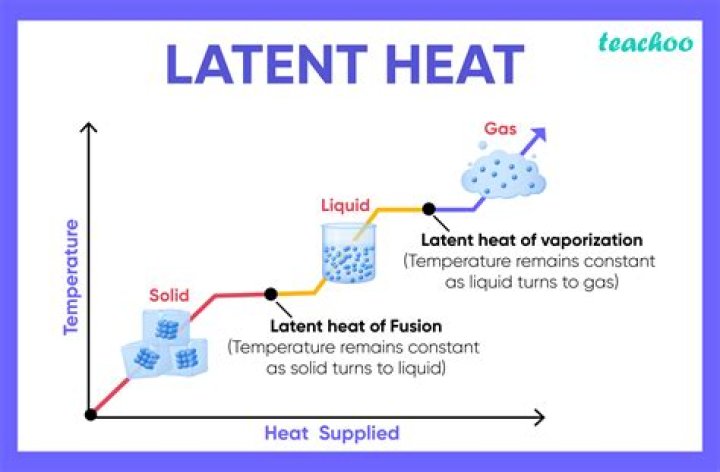
The latent heat is normally expressed as the amount of heat (in units of joules or calories) per mole or unit mass of the substance undergoing a change of state. … Similarly, while ice melts, it remains at 0 °C (32 °F), and the liquid water that is formed with the latent heat of fusion is also at 0 °C.
What unit is latent heat measured in?
Hence, the unit that is used to measure latent heat of a substance is Joule per kilogram (Jg−1).
How do you measure latent heat of ice?
Find the latent heat of fusion, Lf, according to Lf = q ÷ m by dividing the heat, q, absorbed by the ice, as determined in step 3, by the mass of ice, m, determined in step 4. In this case, Lf = q / m = 2293 J ÷ 7.0 g = 328 J/g. Compare your experimental result to the accepted value of 333.5 J/g.
Is latent heat measurable?
It can be said that heat which is measurable is sensible heat and one which is not measurable is latent heat. Although water evaporate and becomes vapor, the vapor becomes water when it is cooled.What is latent heat in HVAC?
Latent heat is the heat the results from an increase or decrease in the amount of moisture held by the air. Specifically, it’s the amount of energy needed to cause a phase change (for our purposes, liquid-to-gas or gas-to-liquid) without changing the actual temperature of a substance.
What is the unit of latent heat of vaporization?
The S.I. unit of specific latent heat of vaporization, Iv of a substance is Joules per kilogram (Jkg-1). Q = Amount of thermal energy immersed or released.
Why is latent heat called so?
Latent heat, also called heat of transformation, is the heat given up or absorbed by a unit mass of a substance as it changes from a solid to a liquid, from a liquid to a gas, or the reverse of either of these changes. … It is called latent because it is not associated with a change in temperature.
How do you calculate latent heat of evaporation?
Latent heat calculation The specific latent heat is different for solid to liquid transition and liquid to gas transition. For example, if we want to turn 20 g of ice into water we need Q = 20 g * 334 kJ/kg = 6680 J of energy. To turn the same amount of water into vapor we need Q = 45294 J .How do you calculate latent heat of steam?
8 Factor of evaporation. H = Total heat in 1 kg of steam at working pressure above 0°C taken from steam tables in kJ/kg. T = Heat in feedwater (kJ/kg). 2256 = the latent heat of steam at atmospheric conditions.
How do you measure latent heat of fusion?The formula of specific latent heat of fusion is given by: Q = mL, which is the required specific latent heat of fusion equation.
Article first time published onIs latent heat measured in Btu?
(also known as moisture tons or wet tons), cooling load required to remove latent heat. … quantity of heat required to cause a change of state of a substance from a saturated liquid to a saturated vapor with no change in temperature, measured in Btu/lbm (J/kg).
What is latent load in HVAC?
Regarding HVAC, latent energy essentially refers to the relative humidity or moisture in the air. A high latent load would mean that the relative humidity was high. … Moreover, the air temperature determines how much moisture the air can hold. The warmer air is, the more moisture it can hold.
What is measurable heat?
Heat is a measurable form of energy that can be transferred from one body to another; it is not a substance.
What are the two types of latent heat?
Two common forms of latent heat are latent heat of fusion (melting) and latent heat of vaporization (boiling). These names describe the direction of energy flow when changing from one phase to the next: from solid to liquid, and liquid to gas.
What is the absolute zero temperature?
absolute zero, temperature at which a thermodynamic system has the lowest energy. It corresponds to −273.15 °C on the Celsius temperature scale and to −459.67 °F on the Fahrenheit temperature scale.
Why is sand hot in the day and cold at night?
At day time sands absorb heat from the sun and become very heated. … Desert is hot throughout the day because of lack of water and desert are cold throughout the night because sand cannot hold heat, so for the period of night when the sun is not shining the sand loses all its heat and compose desert cold.
Which unit is dyne second?
dyneUnit ofForceSymboldynConversions1 dyn in …… is equal to …
What is SI unit of latent heat of fusion?
Notes: The latent heat for a solid liquid state change is called the latent heat of fusion The S.I. unit of the latent heat of fusion is J kg-1.
What is latent heat Short answer?
latent heat, energy absorbed or released by a substance during a change in its physical state (phase) that occurs without changing its temperature. … The latent heat is normally expressed as the amount of heat (in units of joules or calories) per mole or unit mass of the substance undergoing a change of state.
What is VF and VG in steam table?
vf = Specific volume of saturated water (liquid). vg = Specific volume of saturated steam (gas).
What is the delta H vaporization of water?
SubstanceFormulaΔH(vaporization) / kJ mol-1Carbon tetrachlorideCCl430.00Water*H2O40.657 at 100 °C, 45.051 at 0 °C, 46.567 at -33 °Cn-NonaneC9H2040.5MercuryHg58.6
How do you calculate latent heat of sublimation?
Calculate the heat of sublimation of the substance by dividing the heat absorbed by the substance, as calculated in step 2, by the mass of substance in grams. For example, if 47.5 g of substance was placed in the calorimeter, then the heat of sublimation would be 27,100 / 47.5 = 571 J/g.
How do you calculate BTU from CFM?
Sensible heating Btu is measured by the ageless formula CFM x Delta-T x 1.08. To measure heating Btu, multiply the measure supply airflow (in cfm) by the temperature change from the average supply register temperature to the average return grille temperature, and multiply this total by the formula constant of 1.08.
How do you calculate CFM for heating?
- CFM = (Input BTU x thermal efficiency) / (1.08 x DT)
- DT is the temperature rise across the heat exchanger in degrees Fahrenheit.
- CFM = (Volts x Amps x 3.41) / (1.08 x DT)
- CFM = ((Btu/gal oil) x (Nozzle size GPH) x (combustion/thermal efficiency)) / (1.08 x DT)
How much energy does it take to melt 5g of ice?
How much energy is needed to melt 5g of ice? How much energy is needed to melt 5g of ice? Answer: The needed energy to melt of ice is 1670 J.
How many MBtu are in a BTU?
MBtu (IT)/hour [MBtu/h]Btu (IT)/hour [Btu/h]1 MBtu/h1000000 Btu/h2 MBtu/h2000000 Btu/h3 MBtu/h3000000 Btu/h5 MBtu/h5000000 Btu/h
Is there a difference between a BTU and a BTUH?
One BTU is the amount of heat energy we need to raise the temperature of one pound of water by one degree Fahrenheit. … A BTUH is also defined as the number of BTU’s lost (if we’re talking about heat loss or air conditioning), or provided (if we’re talking about providing heat for a building) in one hour.
Is BTU the same as BTU H?
A British Thermal Unit (Btu) is a measurement of heat energy. One Btu is the amount of heat required to raise one pound of water by one degree Fahrenheit. Btus per hour (Btuh) is the benchmark used to estimate the capacity of heating systems, such as gas furnaces.