How magnesium bromide MgBr2 would be formed from atoms of magnesium and bromine

Sarah Rodriguez
Published Feb 17, 2026
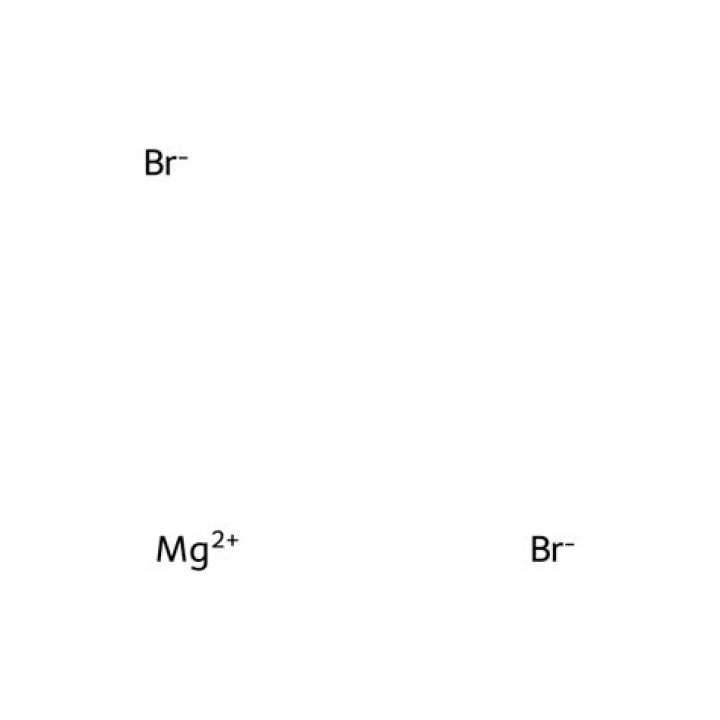
Therefore, for the resulting compound to be neutral, two bromine anions must combine with one magnesium cation to form magnesium bromide (MgBr2).
How would magnesium bromide would be formed from atoms of magnesium and bromine?
Therefore, for the resulting compound to be neutral, two bromine anions must combine with one magnesium cation to form magnesium bromide (MgBr2).
What happens when magnesium reacts with bromine?
Reaction of magnesium with the halogens Magnesium is very reactive towards the halogens such as chlorine, Cl2 or bromine, Br2, and burns to form the dihalides magnesium(II) chloride, MgCl2 and magnesium(II) bromide, MgBr2, respectively.
How would magnesium bromide be formed?
Magnesium bromide can be synthesized by reacting hydrobromic acid with magnesium oxide and crystallizing the product. It can also be made by reacting magnesium carbonate and hydrobromic acids, and collecting the solid left after evaporation.What ions are formed when magnesium bromide dissolves in water?
5. if you dissolve the salt magnesium bromide (MgBr2) in water, the magnesium atoms and the bromine atoms dissociate from each other as positively-charged magnesium ions (Mg2+) and negatively charged bromide ions (Br1-).
Does magnesium and bromine form an ionic bond?
For example, magnesium and bromine form an ionic bond. Magnesium has an oxidation number of +2 and bromine has an oxidation number of -1.
What is the name of the compound formed from magnesium and bromine?
Magnesium bromide (MgBr2)
Will a reaction occur between magnesium bromide and chlorine?
This is a single replacement (double replacement) reaction involving halogens instead of metals. Chlorine and bromine are both halogens in group 17/VIIA. … Chlorine is above bromine, therefore it is more reactive and will replace bromine in MgBr2 .What type of reaction is magnesium bromide and chlorine?
It’s a displacement reaction magnesium bromide react with chlorine and from bromine gas. Because Cl is more reactive then Br.
What is the formula of an ionic compound formed between magnesium and bromide ions?Magnesium bromide, MgBr2 , is an ionic compound containing the metal magnesium (Mg) and the nonmetal bromine (Br). MgBr2 is the formula unit for the compound, which means that the subscripts give the lowest whole number ratio of magnesium and bromide ions in the compound, which is 1Mg:2Br .
Article first time published onWhat happens when magnesium bromide reacts with water?
Grignard reagents react with water to produce alkanes. The reaction of ethyl magnesium bromide with water will produce ethane.
What are the ions present in magnesium bromide?
The magnesium ion is represented as, Mg2+, while the bromide ion is represented as, Br−, this is because magnesium has 2 valence electrons so it has a charge of positive of 2 while bromine needs only 1 electron in valence shell to complete full configuration, and is more electronegative, so it has a negative charge of …
What type of bond will form between Mg and O?
Another example of ionic bonding takes place between magnesium (Mg) and oxygen (O2) to form magnesium oxide (MgO). Magnesium has two valence electrons and an electronegativity of 1,31, while oxygen has six valence electrons and an electronegativity of 3,44.
Is CH4 ionic or covalent?
Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds. We draw this covalent bonding as a Lewis structure (see diagram). The lines, or sticks, as we say, represent the covalent bonds. There are four bonds from a central carbon (C) linking or bonding it to four hydrogen atoms (H).
What is the chemical formula for the compound formed between magnesium and selenium?
Magnesium selenide | MgSe – PubChem.
What is the chemical formula for the compound formed between magnesium and sulfur?
Magnesium Sulfide has a formula of MgS . In order to bond ionically the charges must be equal and opposite. It will take one -2 sulfide ion to balance one +2 magnesium ion forming a magnesium sulfide molecule of MgS . I hope this was helpful.
What is the formula for the simplest stable compound formed between magnesium and nitrogen?
Magnesium and nitrogen combine to form magnesium nitride, Mg3N2 .
How many bromine atoms does magnesium bromide?
One formula unit of MgBr2 consists of one atom of magnesium (Mg) and two atoms of bromine (Br). The atomic mass of Mg is 24.3 amu.
What is the percent composition of magnesium and bromine in MgBr2?
The percent composition of bromine is 86.7% , and the percent composition of magnesium is 13.3% .
What are ions and how are they formed?
Ions are formed by the addition of electrons to, or the removal of electrons from, neutral atoms or molecules or other ions; by combination of ions with other particles; or by rupture of a covalent bond between two atoms in such a way that both of the electrons of the bond are left in association with one of the …
When writing a chemical equation magnesium bromide would be written as MgBr2 because the compound is ionic?
Magnesium bromide is an ionic compound with the chemical formula MgBr2. What does the “2” tell you? There are two bromide ions for every magnesium.
Does magnesium and sulfur form an ionic compound?
Magnesium, Mg , is located in period 3, group 2 of the periodic table, and has two electrons on its outermost shell, i.e. two valence electrons**. … The electrostatic force of attraction will then bring the magnesium cations and the sulfur anions together → an ionic bond is formed.
Is magnesium an ionic compound?
NameFormulasulfite ionSO32−
How are cations and anions formed?
Ions are charged substances that have formed through the gain or loss of electrons. Cations form from the loss of electrons and have a positive charge while anions form through the gain of electrons and have a negative charge.
What does a balanced chemical equation allows one to determine?
A balanced chemical equation gives the identity of the reactants and the products as well as the accurate number of molecules or moles of each that are consumed or produced. … A stoichiometric quantity is the amount of product or reactant specified by the coefficients in a balanced chemical equation.
What is the balanced equation for aluminum and oxygen?
Finally we have a balanced equation: 4Al + 3O2 –> 2Al2O3 . There 4 aluminums on each side and 6 oxygens on each side!
What is the word equation for magnesium and chlorine?
Under the correct conditions:- Mg + Cl2 ——-> MgCl2 . I.e. it forms the ionic compound magnesium chloride which is made up of Mg2+ ions and Cl- ions in an ionic lattice.
What is the chemical reaction of magnesium and hydrochloric acid?
The reaction between magnesium and hydrochloric acid combine to form a salt of magnesium chloride and release hydrogen gas. This reaction is an example of a single replacement. This single replacement reaction is a classic example of a metal reacting in acid to release hydrogen gas.