How many neutrons does beryllium have with 4

Lily Fisher
Published Feb 16, 2026
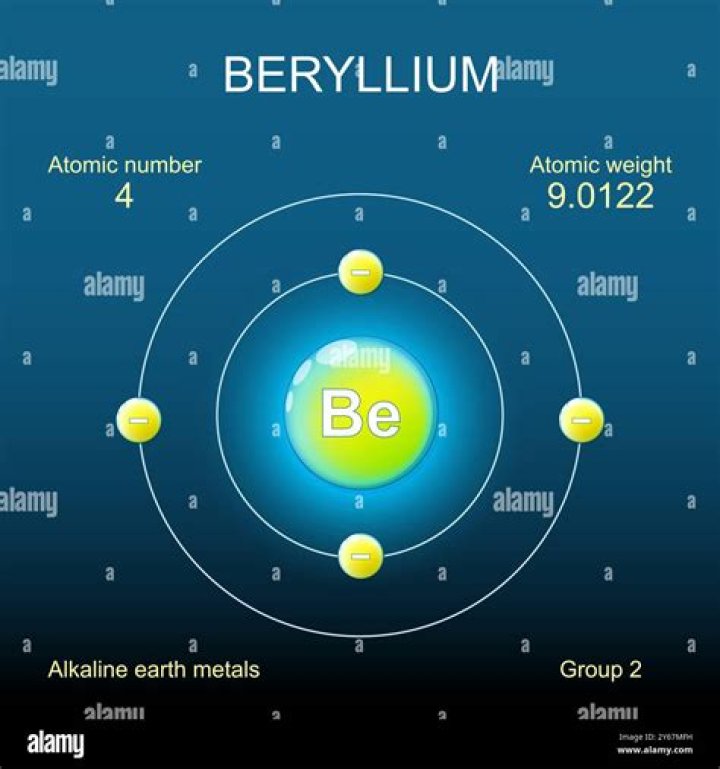
NameBerylliumNeutrons5Electrons4Atomic Number (Z)49
Does beryllium have 4 electrons?
So… for the element of BERYLLIUM, you already know that the atomic number tells you the number of electrons. That means there are 4 electrons in a beryllium atom.
Does a beryllium atom have 4 protons?
Each atom contains an equal number of protons and electrons, thus providing it with a neutral charge. The mass of an atom is contributed by masses of protons and neutrons, electrons are considered comparatively massless. Here, the atomic number is 4, thus beryllium contains 4 electrons and 4 protons.
What atom has 4 protons and 4 neutrons?
The element with atomic number 4 is beryllium, which means each atom of beryllium has 4 protons. A stable atom would have 4 neutrons and 4 electrons. Varying the number of neutrons changes the isotope of beryllium, while varying the number of electrons can make beryllium ions.What has 4 protons 4 neutrons and 6 electrons?
NameProtonsNeutronsLithium34Beryllium45Boron56Carbon66
What element has 4 electrons 4 protons and 5 neutrons?
4 protons, 5 neutrons, and 4 electrons are there in an atom of beryllium.
How do u find neutrons?
The atomic mass equals the number of protons plus the number of neutrons, so you find the number of neutrons by subtracting the number of protons (i.e. the atomic number) from the atomic mass (in atomic mass units).
Which atom has an atomic number of 4?
Atomic NumberSymbolName4BeBeryllium5BBoron6CCarbon7NNitrogenWhat is the atomic number of an atom with 4 electrons 4 protons and 5 neutrons?
So, for beryllium, there are 4 protons, 5 neutrons and 4 electrons but to calculate atomic mass number, four electrons won’t be counted. Hence the atomic mass number of beryllium will be the sum of 4 protons and 5 neutrons and that will be equal to 9.
What element has 4 protons and neutrons?NameBerylliumSymbolBeAtomic Number4Protons4Neutrons5
Article first time published onHow many neutrons does beryllium have?
NameBerylliumNeutrons5Electrons4Atomic Number (Z)49
What is Element 4 on the periodic table?
Beryllium is a silvery-white metal.
What is the VEC of atomic number 4?
Atomic Mass9.012183uElectron Configuration[He]2s2Oxidation States+2Year Discovered1798
What has 4 protons and 3 neutrons?
Notice that because the lithium atom always has 3 protons, the atomic number for lithium is always 3. The mass number, however, is 6 in the isotope with 3 neutrons, and 7 in the isotope with 4 neutrons.
What element has an atomic mass of 4?
Atomic MassName chemical elementnumber6.941Lithium39.0122Beryllium410.811Boron512.0107Carbon6
How many neutrons does beryllium 14 have?
D. All beryllium atoms have four protons but could have between one and ten neutrons. There are ten known isotopes of beryllium, ranging from Be-5 to Be-14. Many beryllium isotopes have multiple decay paths depending on the overall energy of the nucleus and its total angular momentum quantum number.
How many neutrons does each element have?
Elementary ParticleChargeMassNeutron01Electron−1~0
How a neutron is formed?
Neutrons are produced copiously in nuclear fission and fusion. They are a primary contributor to the nucleosynthesis of chemical elements within stars through fission, fusion, and neutron capture processes. The neutron is essential to the production of nuclear power.
How do you find neutrons and electrons?
The number of electrons in a neutral atom is equal to the number of protons. The mass number of the atom (M) is equal to the sum of the number of protons and neutrons in the nucleus. The number of neutrons is equal to the difference between the mass number of the atom (M) and the atomic number (Z).
What is the charge of an atom with 4 protons and 4 electrons?
The atomic mass is expressed by the number of massive particles, protons, and neutrons, and since we have 5 neutrons, the isotope is 9Be . Finally we can assign the charge. There are 4 protonic charges, and 4 electronic charges: the isotope is neutral.
What is the likely atomic mass of an atom that has 4 protons?
The Bohr Model for Beryllium (Be) has 4 protons in the nucleus due to the atomic number of Be being 4. The Mass number is 9 which means Beryllium needs 5 neutrons in the nucleus. (Mass number = protons + neutrons, 9 = 4 + n). Beryllium has four electrons to balance the four protons.
What is the element in Period 4 and Group 8?
Iron. Iron (Fe) is an element in group 8.
What has 12 protons and 12 neutrons?
So you might account for this isotope problem by saying that about 79% of all Magnesium atoms have 12 neutrons, 12 protons, and 12 electrons.
How many neutrons does bromine have?
a) How many neutrons does the atom of bromine have? The mass number = protons + neutrons. Bromine has a mass number of 80 and 35 protons so 80-35 = 45 neutrons.
What is the mass number of a boron atom with 4 neutrons?
We’re being asked to determine the mass number of a boron atom with 4 neutrons. Therefore, the mass number of a boron atom with 4 neutrons is 9.
How is beryllium made?
These days, beryllium is typically obtained from the minerals beryl and bertrandite in a chemical process or through the electrolysis of a mixture of molten beryllium chloride and sodium chloride, the Jefferson Lab reports.
Why does Period 4 have 18 elements?
The third period is similar to the second, filling the 3s and 3p sublevels. Notice that the 3 d sublevel does not actually fill until after the 4s sublevel. This results in the fourth period containing 18 elements due to the additional 10 electrons that are contributed by the d sublevel.



