How many protons electrons and neutrons does nickel have

Dylan Hughes
Published Feb 15, 2026
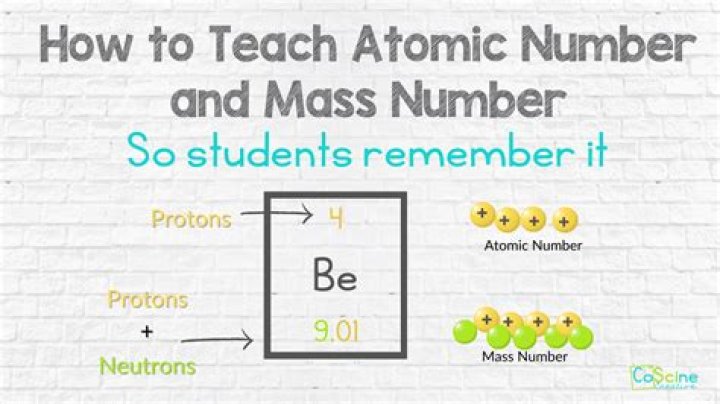
A typical nickel atom has 28 electrons (negative charges) surrounding a nucleus of 28 protons (positively charged nucleons) and 30 neutrons (neutral nucleons).
How many protons neutrons and electrons does nickel 58 have?
Ni-58 has an atomic number of 28 and a mass number of 58. Therefore, Ni-58 will have 28 protons, 28 electrons, and 58-28, or 30, neutrons.In Ni-60 2+ species, the number of protons is same as in neutral Ni-58.
Does nickel have 28 electrons?
Nickel atoms have 28 electrons and 28 protons with 30 neutrons in the most abundant isotope. Under standard conditions nickel is a silvery-white metal that is fairly hard, but malleable.
How many electron does nickel have?
The element nickel (Ni) has 28 electrons. The number of electrons can be determined from the atomic number, which gives the number of protons in an…How many protons and neutrons does nickel 64 have?
Properties of Nickel-64 Isotope:NICKEL-64Relative Isotopic Mass63.927966Neutron Number (N)36Atomic Number (Z)28Mass Number (A)64
How many protons does nickel have?
A typical nickel atom has 28 electrons (negative charges) surrounding a nucleus of 28 protons (positively charged nucleons) and 30 neutrons (neutral nucleons).
How many protons neutrons and electrons are there if nickel has an atomic number of 28 a mass number of 58 and a charge of 2?
Ni-58 has an atomic number of 28 and a mass number of 58. Therefore, Ni-58 will have 28 protons, 28 electrons, and 58-28, or 30, neutrons.In Ni-60 2+ species, the number of protons is same as in neutral Ni-58.
Which of the following electron configuration is correct for nickel?
Answer and Explanation: The answer is e. [Ar]4s23d8 [ A r ] 4 s 2 3 d 8 . The correct electron configuration for Nickel is [Ar]4s23d8 [ A r ] 4 s 2 3 d 8 .Does nickel have 10 valence electrons?
Nickel have 10 valence electrons because transition metals have electrons in both the s shell and the d shell. there are eight electrons in the 3d shell, and two in the 4s shell. Nickel have 10 valence electrons because transition metals have electrons in both the s shell and the d shell.
How many orbitals are in nickel?Nickel is in the 3d sub shell therefore it contains 5 orbitals that can hold up to 10 electrons.
Article first time published onHow do you find the electrons?
- The number of protons in the nucleus of the atom is equal to the atomic number (Z).
- The number of electrons in a neutral atom is equal to the number of protons.
What element contains 28 protons and 30 neutrons?
An atom with 28 protons and 30 neutrons has an atomic number of 28 and a mass number of (30+28) = 58. The atom with atomic number 28 is Ni.
How many neutrons does nickel 60 have?
Properties of Nickel-60 Isotope:NICKEL-60Neutron Number (N)32Atomic Number (Z)28Mass Number (A)60Nucleon Number (A)60
How many neutrons does nickel 63?
NuclideZNExcitation energy61 Ni283362 Ni283463 Ni2835
How many protons does nickel 59?
In the nickel nucleus represented above, the atomic number 28 indicates that the nucleus contains 28 protons, and therefore, it must contain 31 neutrons in order to have a mass number of 59.
How many electrons does K 40 have?
Potassium-40 is composed of 19 protons, 21 neutrons, and 19 electrons. Traces of K-40 are found in all potassium, and it is the most common radioisotope in the human body.
What atom has 19 protons 21 neutrons and 19 electrons?
This atom has atomic number Z = 19 and mass number A = 40, so go to periodic table to find it. This atom has 19 protons and 21 neutrons in the nucleus. The number of electrons is equal to the number of protons. Therefore there are 19 electrons in a potassium atom.
How many protons neutrons and electrons does H+ have?
Explanation: And this is the CATION of the hydrogen atom, 1H , and which necessarily possesses ONE proton, and NO neutrons, and NO ELECTRONS, and hence is positively charged…
How many neutrons are in CA?
Johnson Z. In calcium-40, there are 20 neutrons.
How do you figure out neutrons?
To calculate the number of neutrons in the nucleus of an atom is simple. You take the atomic, or proton number of the element, and you subtract it from the element’s mass number. For example, take the isotope of copper, Cu-63. It contains 29 protons and has a mass number of 63, as suggested in the name.
Does nickel have 3 valence electrons?
Therefore, we can say that nickel has two valence electrons.
How many valence electrons does nickel +2 have?
The electron configuration of nickel ions shows that nickel ion(Ni2+) has three shells and the last shell has sixteen electrons(3s2 3p6 3d8). For this, nickel(Ni) ions have a total of sixteen valence electrons.
How many total electrons does element 15 have?
How many total electrons does element 15 have? How many total and valence electrons are in a neutral phosphorus atom? A neutral phosphorus atom has 15 total electrons. Two electrons can go into first shell, eight in the second shell, and it has five more in the third shell.
Which of the following is the correct configuration of nickel with atomic number 28?
Answer: 2-8-16-2.
Which of the following is the correct electronic configuration of nickel with an atom number 28?
Explanation: Nickel is in the 4th energy level, d block, 7th column, this means that the electron configuration will end 3d8 with the d orbital being one level lower than the energy level it is on.
What is the electronic configuration of nickel 2?
Explanation: Electron configuration for Ni is 1s22s22p64s23d8 .
What is the group number of Nickel?
Group101455°C, 2651°F, 1728 KAtomic number2858.693State at 20°CSolid58NiElectron configuration[Ar] 3d84s27440-02-0ChemSpider ID910ChemSpider is a free chemical structure database
How many protons and electrons are in vanadium?
Vanadium atoms have 23 electrons, 28 neutrons and 23 protons.
How do you find protons neutrons and electrons?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
How many protons and electrons does in have?
ElementHydrogenSymbolHAtomic No.1Number of Electrons in Each ShellFirst1
What are protons neutrons and electrons?
Protons and neutrons are in the center of the atom, making up the nucleus. Electrons surround the nucleus. Protons have a positive charge. Electrons have a negative charge. The charge on the proton and electron are exactly the same size but opposite.