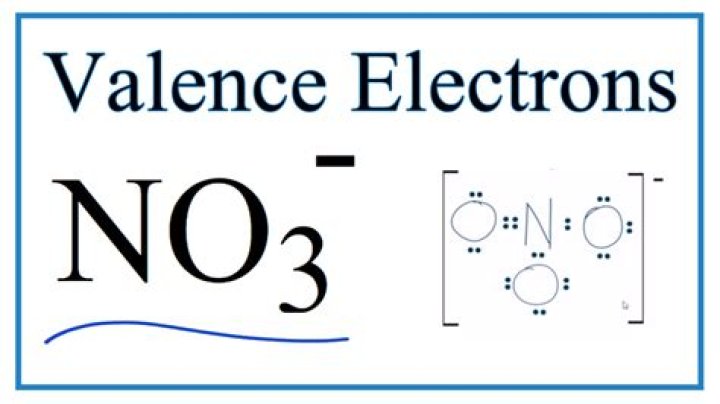
How many valence electrons are available in the ion no3

Andrew White
Published Apr 10, 2026
There are 24 valence electrons available for the Lewis structure for NO3-. It is helpful if you: Try to draw the NO3- Lewis structure before watching the video.
How many electrons are there in no3 ion?
Answer: 32 electrons. Explanation: In N atom, there are 7 electrons.
What is the total number of valence electrons present in a NO2 ion?
After determining how many valence electrons there are in NO2-, place them around the central atom to complete the octets. There are a total of 18 valence electrons for the Lewis structure for NO2-.
What is the valence of no3?
Nitrate ion (NO3) has one Nitrogen and three Oxygen atoms. Oxygen lies in group 16 and thus has a valency of -2. Nitrogen lies in the group 15 and has a charge of +5 forming the bond with three oxygen atoms. The valency of Nitrate is -1.How do you determine valence electrons?
Valence electrons can be found by determining the electronic configurations of elements. Thereafter the number of electrons in the outermost shell gives the total number of valence electrons in that element.
What ion is NO3?
Nitrate is a polyatomic ion with the chemical formula NO−3. Salts containing this ion are called nitrates.
How many valence electrons are there in a carbonate ion co32?
Transcript: Let’s do the CO3 2- Lewis structure: the carbonate ion. Carbon has 4 valence electrons; Oxygen has six, we have 3 Oxygens, and this negative 2 means we have an extra two valence electrons. Add that all up: 4 plus 18 plus 2: 24 valence electrons.
How many valence electrons are in clf2+?
Alternate method: We know that the central atom will be chlorine and it holds 7 valence electrons and also holds an extra electron, hence 8 electrons. We also know that the 2 fluorine atoms bond by occupying an electron each, thus leaving you with 2 bonded and 3 lone pairs on the central atom.How many valence electrons are in the nitrate ion quizlet?
How many valence electrons are in the nitrate ion? N has 5 valence electrons.
What is the valence NO2?The NO2 Lewis structure has a total of 17 valence electrons. It’s not common to have an odd number of valence electrons in a Lewis structure. Because of this we’ll try to get as close to an octet as we can on the central Nitrogen (N) atom.
Article first time published onWhat is the total number of valence electrons in nh3?
Ammonia (NH3) is a commonly tested Lewis structure due to it’s widespread use in agriculture as a fertilizer. It also is a good example of a molecule with a trigonal prymidal molecular geometry. There are 8 valence electrons available for the Lewis structure for NH3.
How do you find valence electrons in Class 9?
In order to find out the number of valence electrons in an atom of the element, we should write down the electronic configuration of the element by using its atomic number. The outermost shell will be the valence shell and the number of electrons present in it will give us the number of valence electrons.
How do you find number of electrons?
To calculate the numbers of subatomic particles in an atom, use its atomic number and mass number: number of protons = atomic number. number of electrons = atomic number.
How many electrons are in co32?
Thirty-two electrons as required.
How many valence electrons are there in ch4?
Example #1 – Methane CH. 4 1. This compound is covalent. This means there are 8 valence electrons, making 4 pairs, available.
How many electron groups are in co32?
With three bonding groups around the central atom, the structure is designated as AX 3. 4. We see from Figure 10.3. 3 that the molecular geometry of CO 3 2− is trigonal planar with bond angles of 120°.
What does the 3 in NO3 mean?
nitrate group. trioxonitrate(V) trioxonitrate(1-) trioxidonitrate(1-) Nitrates, inorganic, n.o.s.
How many valence does nitrogen have?
Nitrogen is found to have either 3 or 5 valence electrons and lies at the top of Group 15 on the periodic table. It can have either 3 or 5 valence electrons because it can bond in the outer 2p and 2s orbitals.
Why is NO3 an ion?
But these three oxygen atoms want 6 electrons. So there’s still a strong need for one last electron. And so when, for example, sodium forms the compound sodium nitrate, NaNO3, the single outer-shell sodium electron transfers over to the NO3, making it a nitrate ion, giving it a -1 charge.
What family has 7 valence electrons?
Any element in the halogen group will have seven valence electrons. These elements include fluorine, chlorine, bromine, iodine, and astatine.
How many valence electrons does chlorine?
The atomic number of chlorine is 17. Hence it has got 7 electrons in its outermost shell. There are 7 valence electrons in the chlorine atom.
How many valence electrons are in a chlorine atom quizlet?
Terms in this set (30) An atom of chlorine has 7 valence electrons. So, according to the Octet Rule, it wants to gain an electron to become stable.
What is the shape of CH3+?
The shape of CH3^+ is (planar/bent).
How many bonding pairs does ClF2+ have?
The central atom has two lone pairs and two bonding pairs (four electron domains). According to VSEPR theory, this predicts a tetrahedral electron geometry.
What is the shape of ClF2+?
The shape of the molecule ClF2+ is “BENT”.
How many ions do you need of each to make sodium nitrate?
Sodium nitrate features an ionic bond between one Na+ ion and one NO3– ion. The structure of a NaNO3 molecule is illustrated below. The nitrate anion has a trigonal planar structure in which 3 oxygen atoms are bonded to a central nitrogen atom.
What is the bond order of no3?
For example, for NO3-, you have three bonds: One double bond (2 electron pairs) and two single bonds (1 + 1= 2 electron pairs). The bond order is therefore 4/3 = 1.33.
Why is nitrate formula no3?
When a laboratory reports directly the concentration of nitrate, it is referring specifically to the nitrate ion, which is designated chemically as NO3-. … Nitrate is one part nitrogen plus three parts oxygen so nitrogen only makes up about 22.6 percent on the nitrate ion.
Is no3 ionic or covalent?
NO3 is covalent in nature because nitrogen and oxygen, both are non-metals and hence, an ionic bond cannot be formed between them.
What is the charge of N in NO3 1?
From the structure itself, we find a positive charge on Nitrogen, hence it carries a formal charge of +1.
How many nitrate ions are needed to balance the charge of the calcium ion?
Calcium ions have a charge of 2+, while nitrate ions have a charge of 1−. We will need two nitrate ions to balance the charge on each calcium ion.