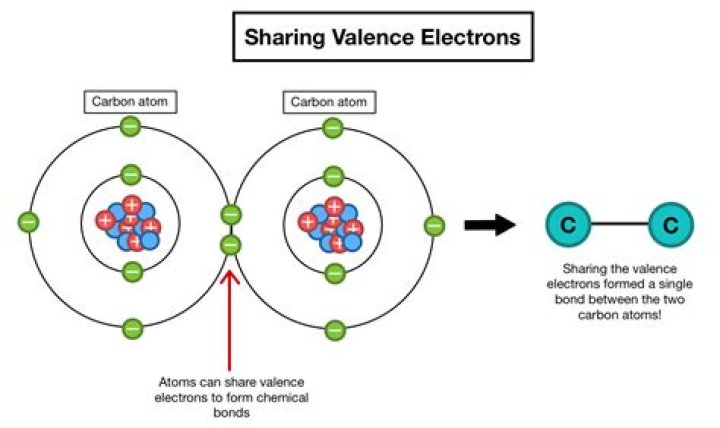
How many valence electrons do insulators have

Andrew Campbell
Published Apr 07, 2026
Normally, a conductor has three or less valence electrons, an insulator has five or more valence electrons, and semiconductors usually have four valence electrons.
Can a conductor have 2 valence electrons?
Most commonly used electrical conducting material is aluminium and it has three valence electrons, and another metallic conductor is magnesium which has two valence electrons.
What determines if an atom is a conductor or an insulator?
The distribution of electrons in the orbital rings around an atom’s nucleus determines the element’s electrical properties. The stability of the electrons in the outer ring determines whether a material made from this element is a conductor, insulator, or semiconductor.
Do insulators have many free electrons?
Materials with high electron mobility (many free electrons) are called conductors, while materials with low electron mobility (few or no free electrons) are called insulators.How many valence electrons do have?
Iodine has seven valence electrons.
How do you find the valence electrons of an atom?
For neutral atoms, the number of valence electrons is equal to the atom’s main group number. The main group number for an element can be found from its column on the periodic table. For example, carbon is in group 4 and has 4 valence electrons. Oxygen is in group 6 and has 6 valence electrons.
How many electrons are in an insulator?
Remember that a good conductor has 1 valence electrons and an insulator has eight valence electrons.
How many electrons are in the valence ring of an atom which is a semiconductor?
Semiconductors: Semiconductors are materials that have a conductivity between conductors (generally metals) and non-conductors or insulators. It has 4 valance electrons in the outermost shell.Do insulators have electrons?
Insulators are materials whose atoms have tightly bound electrons. These electrons are not free to roam around and be shared by neighboring atoms. Some common insulator materials are glass, plastic, rubber, air, and wood.
Which of the following is an insulator answer?– Graphite is a non-metal. It is an allotrope of carbon. Due to the movement of non-bonded electrons in between its adjacent layers, graphite does conduct electricity and therefore, graphite is a conductor. – Therefore, sulphur is an insulator.
Article first time published onWhat makes something a good insulator?
Insulators have strong bonds that hold their particles rigidly in place. … This prevents particles from gaining energy and increasing the temperature. Wool, dry air, plastics, and polystyrene foam are all examples of good insulators. Materials that do not insulate well are called conductors.
What makes an atom A insulator?
“Conductor” implies that the outer electrons of the atoms are loosely bound and free to move through the material. Most atoms hold on to their electrons tightly and are insulators.
How do particles move in insulators?
Other substances, such as glass, do not allow charges to move through them. These are called insulators. Electrons and ions in insulators are bound in the structure and cannot move easily—as much as 1023 times more slowly than in conductors.
How many valence electron does neon have?
Neon, with its configuration ending in s2p6, has eight valence electrons.
How many electron does I have?
Iodine, I , is located in period 5, group 17 of the periodic table, and has an atomic number equal to 53 . That means that a neutral iodine atom will have a total of 53 electrons surrounding its nucleus.
Which group has two valence electrons?
Periodic table groupValence ElectronsGroup 2 (II) (alkaline earth metals)2Groups 3-12 (transition metals)2* (The 4s shell is complete and cannot hold any more electrons)Group 13 (III) (boron group)3Group 14 (IV) (carbon group)4
What is the valence of conductor?
Atoms with 1 valence electron in their outer ring are considered to be the best conductors. Materials with atoms containing 2 or 3 valence electrons are still considered conductors.
What has the least number of valence electrons?
Like charges ____ each other.RepelSicle current flowing throughElectrical current flowing through a conductor produces a ______.Magnetic fieldA _____ has the fewest number of valence electronsConductorWhich of the voltages listed is more likely to be delivered to a commercial building480v
What is an example of a valence electron?
Valence electrons are the electrons in the outermost shell, or energy level, of an atom. For example, oxygen has six valence electrons, two in the 2s subshell and four in the 2p subshell.
How many valence does nitrogen have?
Nitrogen is found to have either 3 or 5 valence electrons and lies at the top of Group 15 on the periodic table. It can have either 3 or 5 valence electrons because it can bond in the outer 2p and 2s orbitals.
How many valence electrons does the element indium have?
Atomic Number49Number of Neutrons66Shell structure (Electrons per energy level)[2, 8, 18, 18, 3]Electron Configuration[Kr] 4d10 5s2 5p1Valence Electrons5s2 5p1
How are insulators charged?
Insulators can be charged by FRICTION only. because due to FRICTION,surface charges get transferred easily. But always remember theSe transferred electrons are not movable inside the insulators. By transferring of electron an insulator can also be charged..
What is an insulator in physics?
insulator, any of various substances that block or retard the flow of electrical or thermal currents. … Different insulating and conducting materials are compared with each other in this regard by means of a material constant known as resistivity.
How do electrons flow?
Electron flow is what we think of as electrical current. … Each atom has electrons in it. If you put new electrons in a conductor, they will join atoms, and each atom will spit out an electron to the next atom. This next atom takes in the electron and spits out another one on the other side.
What are semiconductor chips?
A semiconductor chip is an electric circuit with many components such as transistors and wiring formed on a semiconductor wafer. An electronic device comprising numerous these components is called “integrated circuit (IC)”.
What are the rings around an atom?
The rings, also known as electron shells, can hold a variable amount of electrons depending on its shell number. For example, the first shell can hold only two electrons. If the atom has more than two electrons, then that atom must have more than one ring.
Are metalloids semiconductors?
A number of the metalloids are semiconductors as they have intermediate conductivity which is temperature dependent. The ability of the metalloids to conduct electricity and heat is far better than the nonmetals, for example, diamond , which are insulators.
Which of the following is a set of only insulators?
The Correct Answer is plastics, ebonite, paper, cotton, Bakelite, dry air, rubber. An insulator is a material that does not conduct electrical current. The vacuum is also an insulator but is not actually material. Most electrical conductors are covered by insulation.
Which of the following is an insulator of heat?
Any material that keeps energy such as electricity, heat, or cold from easily transferring through is an insulator. Wood, plastic, rubber, and glass are good insulators.
Which group among the following is insulator?
Which group among the following is insulator? Explanation: Glass, paper, and cotton are good quality insulators. The rest options contain one or more conducting materials.
How do you determine the best insulator?
The formula is R = l/Λ. The smaller the thermal conductivity and the thicker the insulation, the bigger the R value. Materials with a large R value are good insulators.