Is CPT code 81002 CLIA waived

Ava Hall
Published Mar 16, 2026
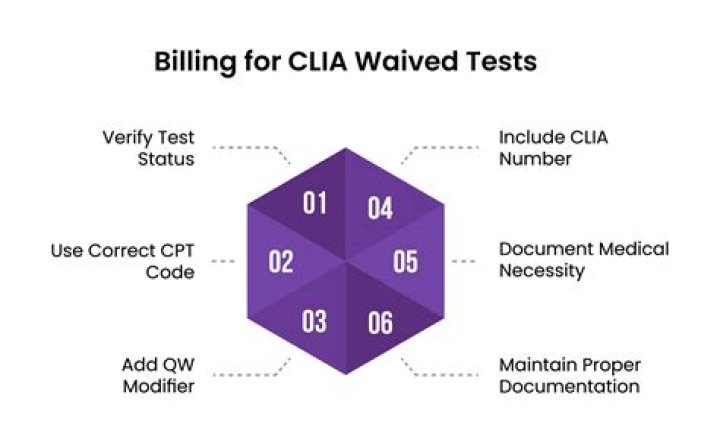
Certain codes describe only CLIA-waived tests and therefore are exempt from the requirement to add the QW modifier. The CPT codes for the tests currently exempt from the requirement are 81002, 81025, 82270, 82272, 82962, 83026, 84830, 85013, and 85651.
Does 81002 require a CLIA number?
QW is Key to CLIA Waived Test Codes The modifier QW CLIA waived test must be appended to all but a handful of CPT codes to be recognized as a waived test. Codes not requiring the QW are 81002, 82270, 82272, 82962, 83026, 84830, 85013, and 85651 .
What are 4 examples of CLIA waived tests?
- CPT 81002: URINALYSIS NONAUTO W/O SCOPE.
- CPT 81025: URINE PREGNANCY TEST.
- CPT 82270: OCCULT BLOOD FECES.
- CPT 82272: OCCULT BLD FECES 1-3 TESTS.
- CPT 82962: GLUCOSE BLOOD TEST.
- CPT 83026: HEMOGLOBIN COPPER SULFATE.
- CPT 84830: OVULATION TESTS.
- CPT 85013: SPUN MICROHEMATOCRIT.
Does Medicare cover CPT code 81002?
All services billed to Medicare must be documented as billed and be medically necessary. CPT codes 81002, 81025, 82270, 82272, 82962, 83026, 84830, 85013, and 85651 do not require a QW modifier to be recognized as a waived test.What are the 3 types of CLIA waivers?
CLIA regulatory requirements classify testing into three categories: high complexity, moderate complexity and waived.
Is 87428 CLIA waived?
The use of code 87428QW for claims submitted by facilities with a valid, current CLIA certificate of waiver is permitted with dates of service on or after November 10, 2020.
What does it mean CLIA waived?
Waived Testing. As defined by CLIA, waived tests are simple tests with a low risk for an incorrect result. They include: Certain tests listed in the CLIA regulations. Tests cleared by the FDA for home use.
What is the difference between 81002 and 81003?
Usually, the automated (81001, 81003) method leads to a print-out from the machine used. In contrast, the non-automated (81000, 81002) method leads to documentation by the tester.Does 81002 need a modifier?
Exception: Because it is the simplest urine dipstick (manual, without microscopy), 81002 is one of the original CLIAwaived tests and does not require modifier QW.
Can you bill for urine collection?CPT code 36415 code is used to report routine venipunctures (and for Medicare only, the collection of urine by catheter) Medicare pays a flat rate of $3.00 for HCPCS code 36415 and does not cover CPT capillary blood collection (CPT code 36416).
Article first time published onWhat is considered waived testing?
Waived testing is laboratory testing that employs specific test methods designated under the Clinical Laboratory Improvement Amendments (CLIA) of the Food and Drug Administration (FDA) as “waived.” Waived testing is designated by CLIA as simple tests that carry a low risk for an incorrect result.
Which of the following is a CLIA-waived test?
CLIA-waived testing, such as Point of Care Testing, are low complexity tests (including Hemoglobin and Hematocrit) that may be performed by other personnel with proper training.
Is CBC CLIA-waived?
The Food and Drug Administration (FDA) has granted premarket clearance and a CLIA waiver to Sysmex America for its complete blood cell count (CBC) test, the XW-100 Automated Hematology analyzer.
How do you get CLIA waived?
- Apply for a CLIA Certificate using Form CMS-116, available through the Centers for Medicare and Medicaid Services. …
- Review your qualifications for a waiver. …
- Apply for a certificate of waiver. …
- Develop a quality assurance plan.
What is the difference between a CLIA certificate and a CLIA waiver?
Certificate of Waiver – This certificate is issued to a laboratory to perform only waived tests. … Certificate of Compliance – This certificate is issued to a laboratory after an inspection that finds the laboratory to be in compliance with all applicable CLIA requirements.
What is the first step for CLIA waived tests?
CLIA Certificate of Waiver (COW) Requirements To qualify for a certificate of waiver, your lab must only perform tests that have received the CLIA waived status. If you meet this requirement, simply fill out the application form CMS-116 at the CMS website or from your state health. The COW is valid for two years.
What is CLIA in medical billing?
Get Online Payment Info (PDF) The Centers for Medicare & Medicaid Services (CMS) regulates all laboratory testing (except research) performed on humans in the U.S. through the Clinical Laboratory Improvement Amendments (CLIA).
Which POS used for emergency department?
Place of Service Code(s)Place of Service Name23Emergency Room – Hospital24Ambulatory Surgical Center25Birthing Center26Military Treatment Facility
What CPT code is used for Covid testing?
March 13, 2020: Accepted addition of code 87635 to report infectious agent detection by nucleic acid (DNA or RNA); severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) (coronavirus disease [COVID-19]), amplified probe technique. The new code was effective March 13, 2020.
What is CPT code 96372 used for?
Subcutaneous and Intramuscular Injection Non-Chemotherapy Instead, the administration of the following drugs in their subcutaneous or intramuscular forms should be billed using CPT code 96372, (therapeutic, prophylactic, or diagnostic injection (specify substance or drug); subcutaneous or intramuscular).
What three components are considered when relative value units are established?
ANS: A Rationale: Per CMS – Relative value units (RVUs) Ð RVUs capture the three following components of patient care: Physician work RVU, Practice Expense RVU, and Malpractice RVUs.
What is the correct code for urinalysis?
Urinalysis, Complete With Microscopic Examination With Reflex to Urine Culture, Comprehensive. CPT: 81001.
Does CPT code 81003 need a QW modifier?
Providers possessing a Clinical Laboratory Improvement Amendments (CLIA) Certificate of Wavier or Provider – Performed Microscopy Procedures (PPMP) must utilize a test kit and bill the program utilizing a QW modifier with for the following codes: 80061, 80101, 81003, 81007,82010, 82044, 82055, 82120, 82273, 82274, …
Does 81003 require a modifier?
CPT codes 81002 and 81003 will not be separately reimbursed unless Modifier 25 is appended to the E/M service indicating that a diagnostic, non-screening, urinalysis was performed.
What is the CPT code for CMP?
Test Name:COMPREHENSIVE METABOLIC PANEL (CMP) with A/G RATIO and BUN/CREAT RATIOTest Code:2128989Alias:CMP LAB9653CPT Code(s):80053
Who can Bill 99000?
Handling and Conveyance of Specimens When a specimen is obtained and sent to an outside laboratory, the provider may add CPT code 99000 to the bill to describe the handling/conveyance of the specimen. The carrier shall reimburse $5.00 for this service in addition to the E/M service.
Is modifier 25 needed for urinalysis?
Modifier 25 is not needed. What they payer wants to know, is if your office meets the criteria for Clia Waved Labs and has a Clia Certificate on file. If your office has Clia Certificate, you would bill the UA with QW modifier.
What is procedure code 81001?
CPT® Code 81001 in section: Urinalysis, by dip stick or tablet reagent for bilirubin, glucose, hemoglobin, ketones, leukocytes, nitrite, pH, protein, specific gravity, urobilinogen, any number of these constituents.
Do waived tests require validation?
Each of these categories has different regulatory requirements for personnel, quality control, quality assurance, proficiency testing, etc. Waived tests. The minimum requirement for waived testing is to follow the manufacturers’ directions. There are no recommendations for method validation.
Is urinalysis a CLIA waived test?
Urinalysis dipsticks fall into the CLIA-waived category and are generally very reliable, simple to use, and easy to interpret.
Is a urine pregnancy test a CLIA test?
By the CLIA law, waived tests are those tests that are determined by CDC or FDA to be so simple that there is little risk of error. Some testing methods for glucose and cholesterol are waived along with pregnancy tests, fecal occult blood tests, some urine tests, etc.