Is HCN polar or nonpolar

Lily Fisher
Published Feb 13, 2026
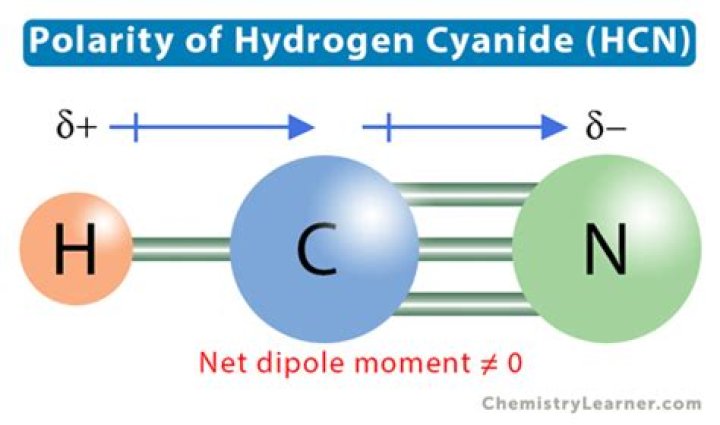
HCN, or hydrogen cyanide, is a polar molecule because there is a large electronegative difference between the N and H across the linear molecule. It consists of two polar bonds whose polarities line up in the same direction.
What bond type is HCN?
In HCN, Carbon is bonded to Nitrogen with a triple covalent bond consisting of one sigma bond and two pi bonds. The sigma bond is formed by overlapping hybridized orbitals, with the two remaining unhybridized orbitals overlapping to form the two pi bonds.
Is HCN bent or linear?
Hydrogen cyanide is a linear molecule. A Lewis formulation counts 1 electron from the hydrogen, 4 electron from the carbon, and 5 electron from the nitrogen, so 5 electron pairs to distribute.
Does HCN have a net dipole moment?
(d) HCN is a linear molecule; it does have a permanent dipole moment; it does contain N, however the nitrogen is not directly bonded to a hydrogen. Therefore dispersion forces and dipole-dipole forces act between pairs of HCN molecules.Is CHN polar or nonpolar?
HCN is an overall polar molecule with a slightly negative pole on the nitrogen atom and a slightly positive pole on the hydrogen. Looking at the bonds, there are no ionic bonds in the molecule. The C-N bond is a slightly polar covalent bond due to the difference in electronegativity between the two atoms.
What is the electron geometry of HCN?
Central atom:CTotal VSEP:41 x triple bond:− 2 pairsRevised Total:2Geometry:Linear
What is the electronegativity for HCN?
HCN is a linear molecule with a single bond between the H and the C and a triple bond between the C and the N. Looking at the electronegativities H<C<N (2.1<2.5<3.0). Carbon will pull the shared electrons in the H-C bond toward C and nitrogen will pull the shared electrons in the C-N triple bond toward N.
Is CH3COOH polar or nonpolar?
Acetic acid(CH3COOH) is a polar molecule because it contains double-bonded oxygen which is more electronegative than a carbon atom, so, the difference of electronegativity in carbon and oxygen atom, generates a dipole moment in the C-O bond because of inducing a positive and negative charge on them.Is HCN soluble in water?
Hydrogen cyanide has a faint, bitter almond odor and a bitter, burning taste. It is soluble in water and is often used as a 96% aqueous solution.
Is HCN a trigonal planar?HCN only has two electron-dense areas around the central atom; therefore, it cannot be trigonal planar in shape.
Article first time published onIs n20 linear?
Nitrous oxide is linear.
How many double bonds are in HCN?
If we draw the Lewis structure for hydrogen cyanide, we will see that there are no double bonds present in HCN. HCN has a total of ten (10) valence…
How many bonding electrons are in HCN?
The carbon atom has (or shares) 3 electrons from the triple bond, and a lone pair of electrons, which it owns. With 2 inner core electrons, this makes 7 electrons with which it is associated.
Why is HCN a linear shape?
Hydrogen Cyanide: The carbon and nitrogen are bonded through a triple bond which counts as “one electron pair”. Hence the molecule has two electron pairs and is linear.
Is HCN soluble in a nonpolar solvent?
HCN is soluble in water due to the following reasons. It is polar in nature which means it has some value of dipole moment.
Is HCN a strong base?
AcidConjugate BaseHCN (hydrocyanic acid) (weakest)CN− (cyanide ion) (strongest)
Is HCN an acid or base?
HCN is an acid with a pKa of 9.2. When hydrogen cyanide is mixed in an aqueous solution, it gives away one proton(H+) and one CN–. The solution containing one hydrogen and one cyanide anion(CN–) is called hydrocyanic acid.
Is hexane non-polar?
Hexane is a non-polar solvent with a boiling point of 68°C, and is therefore the solvent of choice for oil extraction from rice bran to yield rice bran oil (RBO).
Is 2propanol polar?
2–propanol or isopropyl alcohol is polar. The structure (CH3)2-CH-OH is composed of a more negative hydroxyl group and a more positive carboxylate isopropyl group.
Is CH3CH2OH polar or nonpolar?
The structure of ethanol is given below. Hence, the molecule CH3CH2OH C H 3 C H 2 O H is polar.
Is HBr most polar?
The answer is d. HF. Polar bonds are formed when the two atoms involved in the bond have a large difference in their electronegativity values….
What shape is HBr?
NamesStructureMolecular shapeLinearDipole moment820 mDThermochemistry
Is HBr bent shape?
The HBr molecule has a linear or tetrahedral geometry shape because it contains one hydrogen atom in the tetrahedral and three corner with three lone pairs of electrons.
Is HCN planar or nonplanar?
HCN is a planar molecule. In HCN, central atom C is attached to H atom and N atom through two sigma bonds and between C and N there are two π bonds.
What is the geometry of the sf6 molecule its Lewis structure is?
The molecular geometry of SF6 is octahedral with a symmetric charge distribution around the central atom. Therefore this molecule is nonpolar.
How many valence electrons does HCN have?
The total number of valence electrons in HCN is equal to the sum of the number of valence electrons in Hydrogen plus the number of valence electrons in Carbon Nitrogen has ten valence electrons, so Hydrogen Cyanide, HCN, has ten valence electrons.
Is n20 trigonal planar?
Nitrogen Dioxide: The nitrogen and and one oxygen are bonded through a double bond which counts as “one electron pair”. Hence the molecule has three electron pairs and is trigonal planar for electron pair geometry.
Is N2O a geometry?
N2O molecular geometry is a linear shape with a bond angle of 180 degrees. Nitrogen(N) and Oxygen(O) atoms are covalently linked in the N2O molecular geometry. N and O have five and six valence electrons, respectively. N2O contains a total of 16 valence electrons.
What type of intermolecular force is N2O?
NO2 is a polar molecule (due to it having a bent shape) so the most significant intermolecular forces would be dipole-dipole forces.
How many bonds are in HCN molecule?
So, the HCN molecule has 2 sigma (σ) bonds and 2 pi (π) bonds.
Does HCN have a delocalized pi bond?
Among the given molecules, The O3 ,and CO2−3 C O 3 2 − contain the π bonds between the atoms which are delocalized on the oxygen atoms. HCN H C N also contains the π bond between carbon and nitrogen but the bond is localized as its hydrogen atom cannot accommodate the double or triple bond.