Is if5 a polar molecule

Nathan Sanders
Published Feb 12, 2026
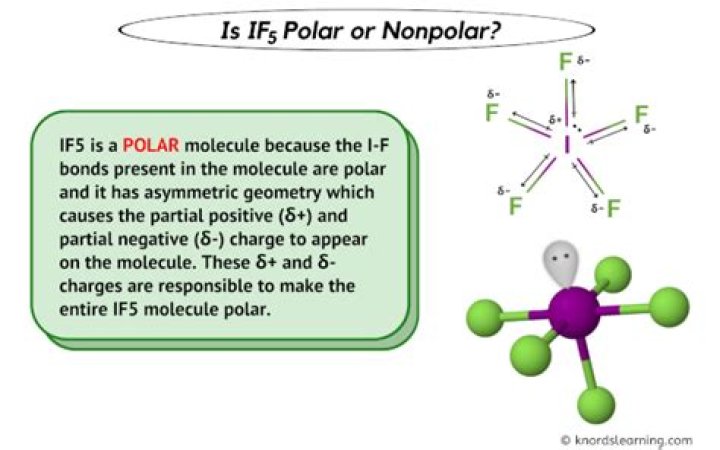
Therefore, IF5 is definitely POLAR. The iodine-fluorine bond will have a dipole moment.
Why is IF5 polar?
IF5 Polarity IF5 has a bent square Pyramidal shape because of lone pair and bond pair repulsion. Notice that the 4 bonds of IF5 cancel each other out, but there is still one bond remaining. Due to Fluorine being more electronegative than I, a dipole moment is induced thus making IF5 a polar molecule.
Which is a polar molecule?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. … A dipole is any molecule with a positive end and a negative end, resulting from unequal distribution of electron density throughout the molecule.
Is sf5 a polar molecule?
The ion has polar bonds and the ion is polar.What determines if a molecule is polar or nonpolar?
A molecule is polar if there’s a significant difference in the electronegativity charges between elements. The bonds don’t cancel each other out and are asymmetrical. A nonpolar molecule has no separation of electric charges or difference in electronegativity.
Is CO2 polar?
Both CO2 and H2O have two polar bonds. However the dipoles in the linear CO2 molecule cancel each other out, meaning that the CO2 molecule is non-polar. The polar bonds in the bent H2O molecule result in a net dipole moment, so H2O is polar.
Is SF4 polar?
The Sulfur tetrafluoride is a polar molecule because Fluorine is more electronegative than Sulfur. With this, the distribution of the charge is not equal, making the SF4 polar molecules. The molecular geometry of SF4 is in a seesaw molecular shape that can be seen when you draw the Lewis Structure.
What substances are non polar?
- Any of the noble gasses: He, Ne, Ar, Kr, Xe (These are atoms, not technically molecules.)
- Any of the homonuclear diatomic elements: H2, N2, O2, Cl2 (These are truly nonpolar molecules.)
- Carbon dioxide – CO. …
- Benzene – C6H. …
- Carbon tetrachloride – CCl. …
- Methane – CH. …
- Ethylene – C2H.
How do you know if a molecule is polar?
- If the arrangement is symmetrical and the arrows are of equal length, the molecule is nonpolar.
- If the arrows are of different lengths, and if they do not balance each other, the molecule is polar.
- If the arrangement is asymmetrical, the molecule is polar.
- Water – H2O.
- Ammonia – NH. …
- Sulfur dioxide – SO. …
- Hydrogen sulfide – H2S.
- Carbon monoxide – CO.
- Ozone – O. …
- Hydrofluoric acid – HF (and other molecules with a single H)
- Ethanol – C2H6O (and other alcohols with an OH at one end)
Is scl2 polar?
The S−Cl bond is polar because the difference in electronegativity between the two atoms is >0.5 , but the resulting dipole moments will not cancel each other out because the molecule is not symmetrical.
Is H2S polar?
H2S is a slightly polar molecule because of the small difference in electronegativity values of Hydrogen (2.2) and Sulfur (2.58) atoms.
Which molecule is most polar?
Water is the most polar molecule because a bond between oxygen and hydrogen has the most difference out of the atoms listed.
What are examples of polar and nonpolar molecules?
CharacteristicsPolar MoleculesNonpolar MoleculesExamplesSome examples of polar molecules are H2O, CHF3, NH3, etc.Some examples of nonpolar molecules are CO2, H2, benzene, etc.
What is the shape of sef6?
The molecular geometry of SeF6 is octahedral with symmetric charge distribution on the central atom.
What is SF4 molecular geometry?
SF4 molecular geometry is see-saw with one pair of valence electrons. The nature of the molecule is polar. These atoms form a trigonal bipyramidal shape. The equatorial fluorine atoms have 102° bond angles instead of the actual 120o angle.
What best describes the polarity of IF5?
Which choice best describes the polarity of IF5? The molecule is polar and has polar bonds.
Why is SF2 and SF4 polar?
In VSEPR theory, the lone pair forces the molecular geometry of SF4 into a see-saw shape. Two of the S-F bonds are pointing away from each other, and their bond dipoles cancel. But the other two S-F dipoles are pointing “down”. Their bond dipoles do not cancel, so the molecule is polar.
Are BH3 bonds polar?
Each B-H bond in BH3 is polar / forms a dipole because the B and H atoms have different electronegativities. The shape of the molecule is trigonal planar which is symmetrical, so the dipoles / bond polarities cancel. The resultingBH3 molecule is non-polar.
Is KrF2 polar?
Molecules with lone pairs around the central atom are generally nonsymmetric, meaning that the individual bond dipoles do not usually cancel each other out giving a polar molecule. Exceptions: KrF2 (linear shape) and XeF4 (square planar shape) type compounds are nonpolar even though the central atom has lone pairs.
Why is ICl3 polar?
The iodine-chlorine and three iodine-chlorine bonds in the iodine trichloride(ICl3), for example, are polarised toward the more electronegative value chlorine atoms, and because all three (I-Cl) bonds have the same size and polarity, their sum is nonzero due to the ICl3 molecule’s bond dipole moment due to pulling the …
Why SF6 is non polar?
SF6 is a nonpolar compound in nature because as per VSEPR theory six fluorine atoms are arranged symmetrically with the sulfur atom such that dipole moment of S-F bond gets canceled out making the SF6 a nonpolar compound.
Why is bf4 nonpolar?
Dear Student, BF3 has triagonal planar shape while BF4- has a tetrahedral shape. BF3 being symmetrical in shape and all the bonds are identical, so it will have zero dipole moment i.e. it will be non polar.
Is no2 polar?
no2 is polar molecules because this molecules has different Electronegativity. Polar molar occur when molecules has different Electronegativity. If i m talking about nitrogen dioxide molecules then, oxygen has more Electronegativity compare with nitrogen.
What is the polarity of NH3?
NH3 is polar because it has 3 dipoles that do not cancel out. Each N-H bond is polar because N is more electronegative than H. NH3 is overall asymmetrical in its VSEPR shape, so the dipoles don’t cancel out and it is therefore polar.
Is vitamin A polar?
Nonpolar Vitamins Vitamin A is nonpolar and can dissolve in fat in your body because fat is also a nonpolar substance.
What causes a molecule to be polar?
Polarity results from the uneven partial charge distribution between various atoms in a compound. Atoms, such as nitrogen, oxygen, and halogens, that are more electronegative have a tendency to have partial negative charges. … A polar molecule results when a molecule contains polar bonds in an unsymmetrical arrangement.
What makes a molecule non polar?
A nonpolar molecule is one whose charge distribution is spherically symmetric when averaged over time; since the charges oscillate, a temporary dipole moment exists at any given instant in a so-called nonpolar molecule.
Which one is non polar?
Difference between Polar and NonpolarPOLARNONPOLARH bonds occur in polar bondsVan der waal interactions between nonpolar bondsAt Least one polar covalent is present in all polar moleculesNonpolar covalent is not present in all nonpolar moleculesCharge separationNo charge separation
Is milk polar or non polar?
Milk is a mix of polar and non-polar substances. Most of the chemical components of milk are polar. … Since Milk is mostly water it is largely a polar substance.
Is NaCl polar?
Sodium Chloride (NaCl) which is an ionic compound acts as a polar molecule. Usually, the large difference in electronegativities in sodium and chlorine makes their bond polar.