Is LiOH an ionic compound

Rachel Hunter
Published Mar 07, 2026
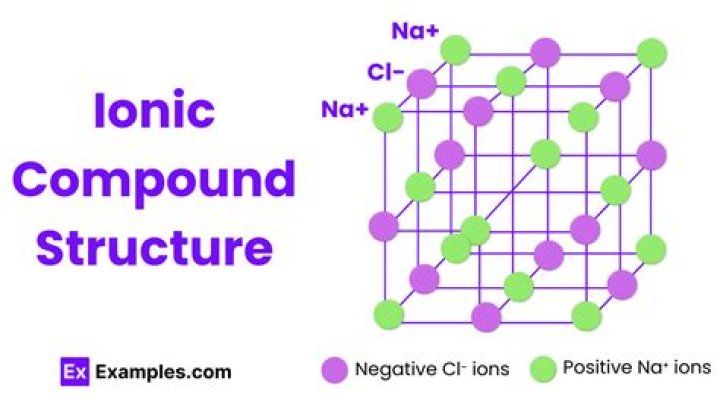
LiOH is the compound Lithium Hydroxide. OH− is a polyatomic anion. Lithium is a Li−1 anion. Because the charges are already equal and opposite these bond together ionically in a one:to:one ratio.
Is LiOH an ionic or covalent?
It is reasonably polar ( ENH=2.2 , ENLi=0.98 ), which is why it is an ionic compound. Mike J. While LiH is a network solid with alternating Li and H atoms, the bonds between atoms have significant covalent character (only about 30% ionic character based on the electronegativity difference).
What is the name of the ionic compound LiOH?
PubChem CID3939Molecular FormulaHLiO or LiOHSynonymsLithium hydroxide 1310-65-2 Lithium hydrate Lithium hydroxide (Li(OH)) Lithium hydroxide anhydrous More…Molecular Weight24.0Component CompoundsCID 962 (Water) CID 3028194 (Lithium)
What type of compound is LiOH *?
Lithium hydroxide is an inorganic compound with the formula LiOH(H2O)n.Is lithium ionic compound?
If we look at the ionic compound consisting of lithium ions and bromide ions, we see that the lithium ion has a 1+ charge and the bromide ion has a 1− charge. Only one ion of each is needed to balance these charges. The formula for lithium bromide is LiBr. … Therefore, the proper formula for this ionic compound is MgO.
Is Li and S ionic or covalent?
Ionic bonds are formed when positively charge cation (Li+ ) is attracted towards negatively charged anion (S2− ). So, when lithium and sulfur combine, it forms an ionic bond. The compound formed is Lithium Sulfide and the chemical formula is Li2S L i 2 S . Thus, lithium and sulfur form an ionic bond.
Is Li and O ionic or covalent?
Lithium oxide is an ionic compound formed between a metal (Li) and a non- metal (O) by the complete transfer of electrons from Li to O to give Li+ cations and O2– anions.
Is LiOH an electrolyte?
Strong bases are also strong electrolytes. Compounds that are formed with the hydroxide ion, OH-, are typically strong bases. Examples include LiOH, NaOH, KOH, Ca(OH)2 and Ba(OH)2. … If the compound is either one of these, then it is definitely a strong electrolyte.Is Li a compound or element?
lithium (Li), chemical element of Group 1 (Ia) in the periodic table, the alkali metal group, lightest of the solid elements. The metal itself—which is soft, white, and lustrous—and several of its alloys and compounds are produced on an industrial scale.
Is LiOH an acid or base?Is LiOH an acid or base? Lithium hydroxide (LiOH) is the base since it releases OH– ions on dissolving in an aqueous solution. LiOH is the strong base because it completely dissociates in an aqueous solution and produces a large number of OH– ions.
Article first time published onIs LiCN a molecular compound?
IdentifiersChemical formulaLiCNMolar mass32.959 g/molAppearanceWhite PowderDensity1.073 g/cm3 (18 °C)
What type of compound is HC2H3O2?
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula CH3COOH (also written as CH3CO2H, C2H4O2, or HC2H3O2).
Is Li+ cation or anion?
Lithium(1+) is a monovalent inorganic cation, a monoatomic monocation and an alkali metal cation.
Is BrCl an ionic compound?
Rubidium is a metal while both chlorine and bromine are non-metals. … Whereas, in $BrCl$ , both bromine and chlorine are halogens and tend to gain electrons. So they share an electron and form a covalent bond. Thus, we can say that $RbCl$ is the more ionic compound.
Why is lithium not ionic?
Lithium belongs to the family of Alkali metals whereas Beryllium belongs to the family of Alkali Earth Metals. In order to form ionic bonds an atom must lose electron easily to become positively charged ion or Cation. … The lesser the ionization enthalpy the easier it is to form a cation and thus ionic bond.
What kind of element is lithium?
A highly reactive and inflammable element, lithium (Li) is the first alkali metal in the periodic table which consists of elements such as sodium (Na). Present all over the world, lithium does not exist in its pure state in nature but can be extracted in small quantities from rock, clay and brine.
How is lithium fluoride an ionic compound?
LiF is lithium fluoride. This is an example of a binary ionic compound, which consists of two elements, a cation and anion. Since lithium, the metal has a plus one charge, and fluoride, a nonmetal, has a negative charge, these two ions are held together through an ionic bond. … In this case, Li is written as lithium.
Is NH4NO3 ionic or covalent?
Ionic bond. NH4NO3 is a nitrate salt of the ammonium cation. Since ammonium is a cation and bonds with the anion nitrate, hence the compound is bonded by an ionic bond.
Is lialh4 ionic?
Lithium aluminum hydride, LiAlH4, contains both ionic and covalent bonds.
Is lithium a mineral?
Lithium was first discovered in the mineral petalite. Lepidolite and spodumene are other common minerals which contain lithium. Commercial quantities of these three minerals are in a special igneous rock deposit that geologists call pegmatite. … Lithium resources are detailed in the USGS Mineral Commodities Summaries.
Is lithium a common element?
At 20 mg lithium per kg of Earth’s crust, lithium is the 25th most abundant element. According to the Handbook of Lithium and Natural Calcium, “Lithium is a comparatively rare element, although it is found in many rocks and some brines, but always in very low concentrations.
Is lithium an isotope?
Naturally occurring lithium (3Li) is composed of two stable isotopes, lithium-6 and lithium-7, with the latter being far more abundant on Earth. Both of the natural isotopes have an unexpectedly low nuclear binding energy per nucleon (5332. 3312±0.0003 MeV for lithium-6 and 5606.
What type of electrolyte is LiOH?
Strong Electrolytesstrong acidsHCl, HBr, HI, HNO3, HClO3, HClO4, and H2SO4strong basesNaOH, KOH, LiOH, Ba(OH)2, and Ca(OH)2saltsNaCl, KBr, MgCl2, and many, many moreWeak Electrolytesweak acidsHF, HC2H3O2 (acetic acid), H2CO3 (carbonic acid), H3PO4 (phosphoric acid), and many more
Is LiOH a base in aqueous solution?
LiOH is one of the few strong bases. In aqueous solution, it dissociates completly into Li+ and OH− . Although water molecules can react with OH− , hydroxide ions will still form as the water molecule will donate its hydrogen ion to hydroxide ion.
How LiOH qualifies as a base?
Chemist Johannes Bronsted and scientist Thomas Lowry proposed that an acid should be defined as a substance that can donate a proton, while a base is any substance that can accept protons.
How does LiOH qualify as a base?
Bases are defined as a compound or element that releases hydroxide (OH-) ions into the solution. In this reaction lithium hydroxide (LiOH) dissociates into lithium (Li+) and hydroxide (OH−) ions when dissolved in water.
What is the compound name for LiCN?
PubChem CID75478Molecular FormulaLiCN or CLiNSynonymsLithium cyanide 2408-36-8 lithium;cyanide LiCN Lithium cyanide (Li(CN)) More…Molecular Weight33.0Component CompoundsCID 3028194 (Lithium) CID 768 (Hydrogen cyanide)
Is LiCN an acid or base?
Lithium cyanide, LiCN, is a salt formed from by the reaction of the weak acid hydrocyanic acid, HCN, and the strong base lithium hydroxide.
What kind of compound is H3PO4?
A phosphorus oxoacid that consists of one oxo and three hydroxy groups joined covalently to a central phosphorus atom. Phosphoric acid, also known as orthophosphoric acid or phosphoric(V) acid, is a weak acid with the chemical formula H3PO4. The pure compound is a colorless solid.
What is the ionic charge of H3PO4?
The formula for phosphoric acid is H3PO4, what you have listed is HPO4 with a charge of -2 and is an ion. This ion is named hydrogen phosphate.
Is H3PO4 metal or nonmetal?
For example, one molecule of water which has a formula of ${H_2}O$ is formed by 2 atoms of hydrogen and 1 atom of oxygen whereas in case 2 atoms of hydrogen combine with 2 atoms of oxygen, we get ${H_2}{O_2}$ which is a different compound.