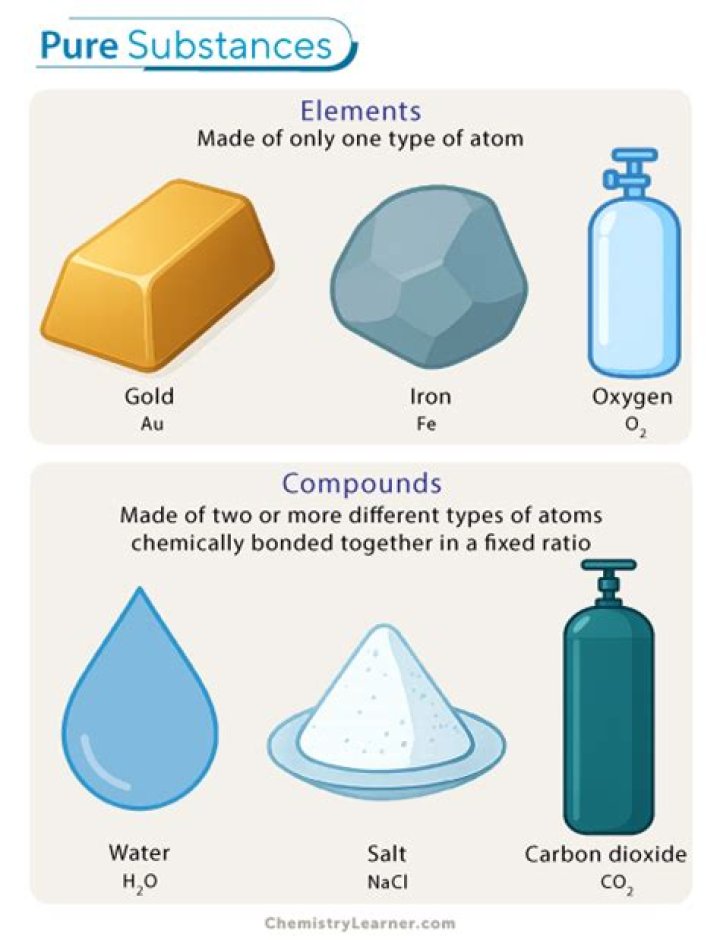
Is oxygen a pure substance or a mixture

Rachel Hunter
Published Feb 14, 2026
Pure SubstanceElement or Compound?Consists of:Ammonia (NH3)compoundammonia molecules
Is oxygen a pure substance yes or no?
Yes, elemental oxygen and oxygen gas are both considered a pure substance. The reason is that there is only one kind of atom in the elemental oxygen and in the resulting molecule that is oxygen gas.
Is air a pure substance or a mixture?
Air is not a pure substance, as it’s a mixture of various elements in just the right proportion to sustain human life on Earth. A pure substance is one which has same chemical composition in all its phases. ir is a mixture, mostly of nitrogen (78%) and oxygen (20%).
Why is oxygen is not a mixture?
The hydrogen and oxygen are not joined together. The hydrogen and oxygen have joined together to form the new substance water. Keeps the properties of the substances involved. This mixture is in the gas state.Is oxygen homogeneous mixture?
Mixtures with a consistent composition throughout are called homogeneous mixtures (or solutions). … Air, a mixture of mainly nitrogen and oxygen, is a gaseous solution.
What is a pure substance answer?
A pure substance is a substance which has only one type of atoms or molecules thereby having a constant composition and structure. Complete answer: Substances which are made up of only one kind of atoms or molecules and have a definite composition throughout and have a fixed structure are known as pure substances.
What mixture is oxygen?
Oxygen mixture generally refers to gas blends containing between 50% and 95% oxygen. Oxygen – enriched air, nitrox , and enriched-air nitrox (EAN) are generally used synonymously in the diving industry since all refer to pressurized diving gas mixtures containing various oxygen concentrations greater than standard air.
Is O2 a compound or element?
The oxygen molecule O2 is considered a molecule but not a compound. This is because O2 is made of two atoms…Which of these is a pure substance?
Examples of pure substances include tin, sulfur, diamond, water, pure sugar (sucrose), table salt (sodium chloride) and baking soda (sodium bicarbonate). Crystals, in general, are pure substances. Tin, sulfur, and diamond are examples of pure substances that are chemical elements. All elements are pure substances.
Why air is classified as a solution and oxygen is classified as a pure substance?Air is not a pure substance because it is a homogeneous mixture of different substances.
Article first time published onWhy is air a pure substance?
Air isn’t considered a pure substance because there is no one single chemical element of compound that forms air. Air is a mixture of various pure chemical substances, most of which exist in the gaseous state. There are physical processes through which we can separate the various components of air.
Is the air a solution?
Air is a solution made up of many gases. … There is more nitrogen than any other gas in air, so it is considered the solvent in an air solution.
Is oxygen a product or reactant?
They indicate the number of each chemical species that reacts or is formed. Methane and oxygen (oxygen is a diatomic — two-atom — element) are the reactants, while carbon dioxide and water are the products.
Why is oxygen a homogeneous mixture?
It’s ‘homogeneous’ because the substances are distributed evenly throughout the mixture.
Is oxygen an example of a solution?
A solution may exist in any phase. A solution consists of a solute and a solvent. … Using air as an example, oxygen and carbon dioxide gases are solutes, while nitrogen gas is the solvent.
Is oxygen a element?
oxygen (O), nonmetallic chemical element of Group 16 (VIa, or the oxygen group) of the periodic table.
What is pure oxygen?
Pure oxygen or oxygen-enriched air is used in many industrial applications. … The US CGA (Compressed Gas Association) and other standard-setting bodies define oxygen-enriched mixtures or atmospheres as any mixture or atmosphere containing greater than 23% oxygen.
What is pure oxygen made of?
Dry air is a mixture of 21% oxygen, 78% nitrogen and 1% argon, with a few other trace gases such as carbon dioxide. At the moment, most of the world’s pure oxygen is made by the liquefaction and subsequent distillation of air, to separate it into its components.
What are examples of pure substances and mixtures?
Pure SubstancesMixturesCan be categorised as elements and compounds.Can be categorized as only homogeneous and heterogeneous.Examples include Pure water, Hydrogen gas, Gold.Examples include a mixture of Sand and Sugar, Oil and Water etc.
What are examples of pure substances?
And examples of pure substances include all the elements, water, methane gas, sugar, table salt.
What are 10 examples of pure substances?
- Hydrogen gas.
- Gold metal.
- Sugar (sucrose)
- Baking soda (sodium bicarbonate)
- Ammonia.
- Diamond.
- Copper wire.
- Silicon chip.
Is an egg a pure substance?
An egg is neither a pure substance nor a mixture. If you crack open an egg, you see eggshells, egg whites, and egg yolks. This means that even macroscopically, an egg is not a mixture, it is an unmixed combination of egg shell, egg white, and egg yolk.
What are types of mixture?
There are two types of mixtures: heterogeneous and homogeneous. Heterogeneous mixtures have visually distinguishable components, while homogeneous mixtures appear uniform throughout. The most common type of homogenous mixture is a solution, which can be a solid, liquid, or gas.
Which of the following is not an example of a pure substance?
Answer: Aerated water is a homogeneous mixture of carbon dioxide and water. It is not a pure substance. Carbon dioxide is a compound while oxygen and zinc are elements which are a type of pure substance.
Why is O2 a pure element?
O2 represents a molecule of oxygen which is made up of two atoms of oxygen ;compounds are composed of different types of elements for example HO. … Thus,O is an element.
Is O2 diamagnetic or paramagnetic?
Because the O2 molecule has two unpaired electrons, it is paramagnetic.
Is O2 a molecule or atom?
Two oxygen atoms strongly bind together with a covalent double bond to form dioxygen or O2. Oxygen is normally found as a molecule. It is called dioxygen.
Why air is a mixture?
Air is a mixture and not a compound because: Air does not have a formula like a mixture, while compounds have a fixed formula. When air is formed by gases, there is no energy change. Properties of air are variable and subjective to time and place.
What is the mixture of pure air?
So, Air is a pure substance with chemical composition of 78% of nitrogen, 21% of oxygen, 0.93% of argon, 0.04% of carbon dioxide and 0.01% other gases. Liquid air is a pure substance with chemical composition of 78% of nitrogen, 21% of oxygen, 1% of argon.
Is water pure substance or mixture?
Water, H2O, is a pure substance, a compound made of hydrogen and oxygen. Although water is the most abundant substance on earth, it is rarely found naturally in its pure form. Most of the time, pure water has to be created. Pure water is called distilled water or deionized water.
Is oxygen in air a solution?
Gas-Gas Solutions Our air is a homogeneous mixture of many different gases and therefore qualifies as a solution. … The next major constituent is oxygen (about 21%), followed by the inert gas argon (0.9%), carbon dioxide (0.03%) and trace amounts of neon, methane, helium, and other gases.