Is SDS PAGE same as Western blot

Sarah Rodriguez
Published Mar 12, 2026
SDS-PAGE is an electrophoresis method that separates proteins by mass. Western blot is an analytical technique to identify the presence of a specific protein within a complex mixture of proteins, where gel electrophoresis is usually used as the first step in procedure to separate the protein of interest.
Is Western Blot more specific than SDS-PAGE?
The key difference between SDS Page and western blot is that SDS Page allows the separation of proteins in a mixture while western blot allows detection and quantification of a specific protein from a mixture. Both are useful in protein analysis studies.
Which blotting technique is called SDS?
The principle and method of polyacrylamide gel electrophoresis (SDS-PAGE) SDS-PAGE is an analytical technique to separate proteins based on their molecular weight.
Why is SDS-PAGE preferred for western blotting?
Western blot is preferred with SDS-PAGE instead of native PAGE for a few reasons as following: The role of SDS in SDS-PAGE is to coat the hydrophobic region of the protein with its negative charge and overcome the overall positive charge of the protein so that the protein can migrate towards the positive electrode.Is Western blot same as gel electrophoresis?
The western blot method is composed of a gel electrophoresis to separate native proteins by 3-D structure or denatured proteins by the length of the polypeptide, followed by an electrophoretic transfer onto a membrane (mostly PVDF or nitrocellulose) and an immunostaining procedure to visualize a certain protein on the …
Does western blotting use SDS?
In Western blotting (immunoblotting) the protein mixture is applied to a gel electrophoresis in a carrier matrix (SDS-PAGE, native PAGE, isoelectric focusing, 2D gel electrophoresis, etc.) to sort the proteins by size, charge, or other differences in individual protein bands. … This process is called blotting.
Is SDS-PAGE necessary for western blotting?
SDS-PAGE (1D) separates protein based on molecular weight, while western blotting is done to detect the protein of interest using specific antibodies.
Why is Western blot called Western?
W. … Burnette definitely gave the technique the name “Western blotting” as a nod to Southern blotting and because their lab was on the west coast. He developed his technique independently, including the electrophoretic transfer step, but became aware of Stark’s and Towbin’s publications before he submitted his in 1979.What is SDS-PAGE?
SDS-PAGE is an electrophoresis method that allows protein separation by mass. The medium (also referred to as ′matrix′) is a polyacrylamide-based discontinuous gel.
How does SDS-PAGE work Pubmed?The SDS-PAGE method involves the denaturation of proteins with the detergent sodium dodecyl sulfate (SDS) and the use of an electric current to pull them through a polyacrylamide gel, a process termed polyacrylamide gel electrophoresis (PAGE).
Article first time published onWhat is the difference between PAGE and SDS-PAGE?
The major difference between native PAGE and SDS-PAGE is that in native PAGE, the protein migration rate is dependent on both the mass and structure, whereas in SDS-PAGE, the migration rate is determined only by protein’s mass. In native PAGE, protein samples are prepared in a non-denaturing and non-reducing buffer.
Is SDS-PAGE the same as gel electrophoresis?
SDS-PAGE is a non-selective method of gel electrophoresis used in fields such as: biochemistry, forensics, biology, and genetics to detach protein from their electrophoretic mobility while gel electrophoresis is usually used for separation of biological macromolecules such as DNA, ribonucleic acid (RNA), and protein.
Where is SDS-PAGE used?
Sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) is commonly used to obtain high resolution separation of complex mixtures of proteins. The method initially denatures the proteins that will undergo electrophoresis.
How is Western blot different from Elisa?
The key difference between Elisa and western blot is that Elisa or enzyme-linked immunoassay is a diagnostic tool that detects whether the patient has been exposed to a particular type of virus or another infectious agent while western blot is a technique which detects a specific protein from a protein sample.
Is immunoblot the same as Western blot?
There is no significant difference between immunoblot and western blot. However, immunoblot is the more correct name for the technique due to its usage of antibodies for the detection of proteins in the sample.
What SDS-PAGE gel should I use?
Commonly used are 4-20% gradient gels that can cover a vast range of molecular weight sizes. Proteins ≥ 200 kDa will resolve better in 4-8% gels.
Does SDS-PAGE use antibodies?
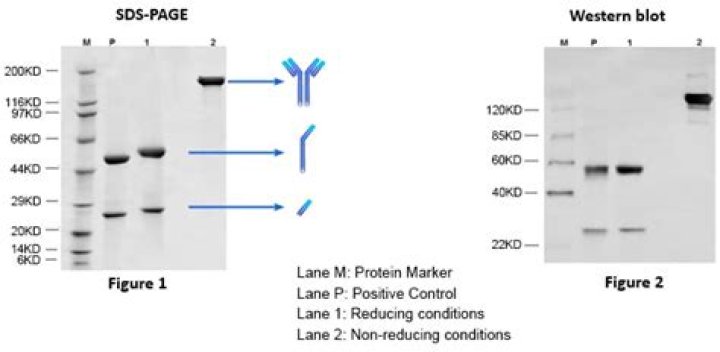
SDS-PAGE is one of the simplest, least expensive, and most commonly used techniques to analyze antibodies for purity. Reduced samples of the IgG class of antibodies give rise to glycosylated heavy chains of approximately 50 kDa and light chains of approximately 25 kDa on SDS-PAGE.
What is the difference between Western blot and PCR?
It actually depends on what you intend to show: WB is about protein and PCR is about nucleic acids (DNA, mRNA). Both may be used as quantitave techniques. If you want to prove the existence on the protein level, western blotting is of course your method of choice.
What information can Immunolabeling provide about proteins that SDS-PAGE and Western blotting Cannot?
Immunoblotting can be used to determine a number of important characteristics of protein antigens-the presence and quantity of an antigen, the relative molecular weight of the polypeptide chain, and the efficiency of extraction of the antigen.
What is Page technique?
Polyacrylamide gel electrophoresis (PAGE) is a technique widely used in biochemistry, forensic chemistry, genetics, molecular biology and biotechnology to separate biological macromolecules, usually proteins or nucleic acids, according to their electrophoretic mobility.
What is SDS used for in SDS-PAGE?
SDS (sodium dodecyl sulfate) is an anionic detergent that unfolds and denatures proteins, coating proteins in negative charge. It is added in excess to the proteins, so that the proteins’ intrinsic charge is covered, and a similar charge-to-mass ratio is obtained for all proteins.
How accurate is SDS-PAGE?
The accuracy of MW estimation by SDS-PAGE is in the range of 5–10%. Polypeptides like glyco- and lipoproteins are usually not fully coated with SDS and will not behave as expected in SDS-PAGE, leading to inaccurate molecular weight estimations.
What does SDS-PAGE separate by?
SDS-PAGE separates proteins primarily by mass because the ionic detergent SDS denatures and binds to proteins to make them uniformly negatively charged. Thus, when a current is applied, all SDS-bound proteins in a sample will migrate through the gel toward the positively charged electrode.
Is Western blotting capitalized?
After all, it was named after its inventor, Edwin Southern, and therefore the use of the capital is justified. Western (and Northern) blots however, were not named after inventors but were given these names in honor of Southern. … Many science editing sites online also agree with not capitalizing western blot.
What do the bands mean on Western blot?
Multiple bands that are scientifically relevant can be observed during Western blot analysis. Higher molecular weight bands are seen when proteins become bound to modifiers whereas lower molecular weight bands are observed when proteins are spliced.
How do you read a Western blot?
To know how to analyze western blot data, Look for the sizes of the bands. These will be represented by a number, either followed by “kDa” or preceded by “p.” This is the size of the protein which has been detected and is the scale on which the proteins are separated in a Western blot.
What are the advantages of SDS-PAGE?
SDS polyacrylamide gel electrophoresis (SDS-PAGE) has the advantages of simple operation and good reproducibility in the determination of protein molecular weight, detection of specific proteins, and identification of strain species.
Does SDS PAGE separate subunits MCAT?
SDS PAGE is different from Native PAGE because it denatures the protein (so subunits released and more bands) and applies a uniform negative charge. The purpose of applying a uniform negative charge is to eliminate the effect of charge on migration.
What is the difference between SDS PAGE and PAGE electrophoresis?
The main difference between gel electrophoresis and SDS PAGE is that gel electrophoresis is a technique used to separate DNA, RNA, and proteins whereas SDS PAGE is a type of gel electrophoresis used mainly to separate proteins.
Does SDS PAGE break disulfide bonds?
Sodium dodecyl sulfate (SDS) is an anionic detergent used to denature proteins prior to gel electrophoresis. … However, SDS does not break down any of the disulfide bonds that participate in many tertiary structures; treatment with DTT, described below, is often necessary to break down disulfide bonds.
What is the difference between SDS PAGE and agarose gel?
Agarose gels are used mainly for nucleic acid separation; when higher resolution is required, polyacrylamide gels are used. SDS Page is a type of gel electrophoresis commonly used to separate complex mixtures of proteins. It is considered as a high-resolution protein separation technique.