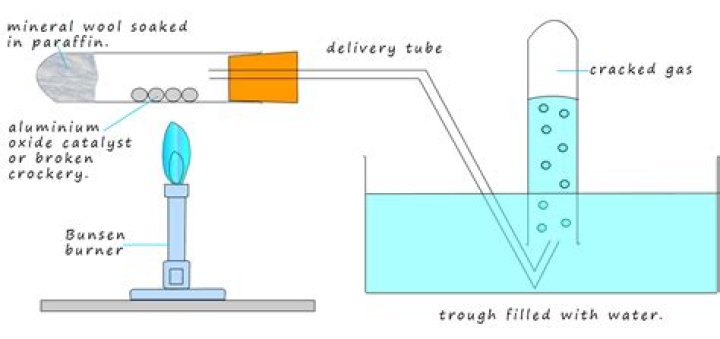
What are the conditions for cracking

Lily Fisher
Published Apr 11, 2026
Catalytic cracking uses a temperature of approximately 550°C and a catalyst known as a zeolite which contains aluminium oxide and silicon oxide. Steam cracking uses a higher temperature of over 800°C and no catalyst.
What are the conditions for thermal cracking?
In thermal cracking, high temperatures (typically in the range of 450°C to 750°C) and pressures (up to about 70 atmospheres) are used to break the large hydrocarbons into smaller ones. Thermal cracking gives mixtures of products containing high proportions of hydrocarbons with double bonds – alkenes.
What is cracking GCSE?
Cracking is a reaction in which larger hydrocarbon molecules are broken down into smaller, more useful hydrocarbon molecules, some of which are unsaturated: the original starting hydrocarbons are alkanes.
What are the methods of cracking?
Cracking is primarily of two types – thermal cracking and catalytic cracking. Thermal cracking is further categorised into modern thermal cracking and steam cracking. On the other hand, sub-classifications of catalytic methods of cracking are hydrocracking and fluid catalytic cracking.What are the products for cracking?
The cracking products, such as ethene, propene, buta-1,3-diene and C4 alkenes, are used to make many important chemicals. Others such as branched and cyclic alkanes are added to the gasoline fraction obtained from the distillation of crude oil to enhance the octane rating.
Why is cracking in air absent?
In the absence of air, steam and raw material are mixed and heated to temperatures around 800oC inside a furnace. … Carbon dioxide and hydrogen sulfide are unwanted byproducts of thermal cracking. These gases must be removed before they escape into the atmosphere by reaction with sodium hydroxide solution.
What do catalysts do in cracking?
The catalytic cracking process involves the presence of solid acid catalysts, usually silica-alumina and zeolites. The catalysts promote the formation of carbocations, which undergo processes of rearrangement and scission of C-C bonds.
What is cracking explain about it?
Cracking is defined as a process, wherein complex organic molecules namely long chain hydrocarbons or kerogens are broken down into smaller molecules namely light hydrocarbons. It is caused by the breaking of carbon-carbon bonds.Why is cracking important?
Cracking is important for two main reasons: It helps to match the supply of fractions with the demand for them. … Since cracking converts larger hydrocarbons into smaller hydrocarbons, the supply of fuels is improved. This helps to match supply with demand.
What is cracking reforming?Catalytic cracking and reforming processes were developed to produce high-octane gasoline. They involve breaking an alkane chain to produce an α-olefin. This occurs with the attachment at the second carbon atom of the α-olefin, resulting in branching.
Article first time published onWhy is ethene an alkene?
The second subset of hydrocarbons is called the alkenes. Their names all end in –ene for example ethene. Alkenes all contain a carbon to carbon double bond which makes them more reactive than the alkanes.
How is cracking done in chemistry?
cracking, in petroleum refining, the process by which heavy hydrocarbon molecules are broken up into lighter molecules by means of heat and usually pressure and sometimes catalysts. Cracking is the most important process for the commercial production of gasoline and diesel fuel.
What is cracking in chemistry BBC Bitesize?
Cracking is a reaction in which larger saturated hydrocarbon molecules are broken down into smaller, more useful hydrocarbon molecules, some of which are unsaturated: the original starting hydrocarbons are alkanes. the products of cracking include alkanes and alkenes, members of a different homologous series.
Why cracking is a thermal decomposition?
These processes break covalent bonds in the molecules, causing thermal decomposition reactions. Cracking produces smaller alkanes and alkenes .
How is cracking an example of thermal decomposition?
Thermal cracking is a type of chemical reaction that uses heat to break down long chain molecules into smaller, more reactive, and therefore potentially more useful, molecules. … The solution is to crack these large alkane molecules to produce the smaller molecules of the fractions that are under produced.
What is naphtha cracker?
The plant with the facilities for cracking naphtha at a high temperature in excess of 800 degrees Celsius to produce petrochemical feedstocks like ethylene, propylene, mixed-C4 and pyrolysis gasoline (PG), is called the naphtha cracking center or naphtha cracking plant.
What is cracked gas?
Cracked gas is gas from a refining process, which is often compressed afterwards. Ethane is cracked in furnaces and then the cracked gas is compressed by the compressor string for finish processing and separation of products.
What is cracking of kerosene?
Kerosene is activated by pre-combustion cracking, which produces a large amount of hydrogen, ethylene, carbon monoxide and acetylene. It can effectively improve kerosene activity and help to realize kerosene-air detonation.
What catalysts are used in cracking?
Catalytic cracking uses a temperature of approximately 550°C and a catalyst known as a zeolite which contains aluminium oxide and silicon oxide.
Does cracking produce carbon dioxide?
Each ton of the extra bituminous coal (60% carbon) for cracking produces 2.2 tons more CO2. Each of those additional tons requires 0.72 tons of coal, and so on, adding rather than subtracting emissions.
How can the temperature required for cracking be reduced?
The most effective way to manage thermal cracking in mass concrete applications is to use concrete mix designs which have a reduced temperature rise (i.e. a lower heat of hydration). This usually involves replacing some of the portland cement in the mix with class F fly ash, slag cement, or a combination of both.
Is Cracking legal?
The distribution of cracked copies is illegal in most countries. There have been lawsuits over cracking software. It might be legal to use cracked software in certain circumstances. Educational resources for reverse engineering and software cracking are, however, legal and available in the form of Crackme programs.
What are the disadvantages of cracking?
- Lung damage.
- Respiratory problems.
- Increased blood pressure.
- Tachycardia or racing heart beat.
- Onset of psychotic symptoms.
Why is cracking knuckles bad?
Knuckle “cracking” has not been shown to be harmful or beneficial. More specifically, knuckle cracking does not cause arthritis. Joint “cracking” can result from a negative pressure pulling nitrogen gas temporarily into the joint, such as when knuckles are “cracked.” This is not harmful.
What is a crack code?
crack the code, to To solve a difficult problem or mystery. The term is derived from deciphering coded intelligence during wartime and became current during World War II, when cryptography, used by armies at least since Napoleon’s day, reached new heights of sophistication.
What is cracking Class 11?
Define cracking. The thermal decomposition of higher hydrocarbons into lower hydrocarbons in the presence or absence of a catalyst is called cracking.
Do alkanes burn in oxygen?
However, these alkanes burn very rapidly. The combination of alkanes with oxygen-generating heat is known as combustion. More precisely, combustion is defined as “a chemical reaction with oxygen in which alkane is converted into carbon dioxide and water with the release of heat energy”.
How do you make heptane?
Preparation. The linear n-heptane can be obtained from Jeffrey pine oil. The six branched isomers without a quaternary carbon can be prepared by creating a suitable secondary or tertiary alcohol by the Grignard reaction, converting it to an alkene by dehydration, and hydrogenating the latter.
Are alkanes flammable?
In general, alkanes show a relatively low reactivity. However, protonation, oxygenation, pyrolysis, radiolysis, and photolysis are possible under extreme reaction conditions. … Lower alkanes in particular are highly flammable and form explosive mixtures (methane, benzene) with air (oxygen).
Does cracking produce hydrogen?
The hydrogen produced in methane cracking can be used as an energy source in fuel cell vehicles as well as for generating electricity and heat. … In addition to hydrogen, very pure powdery carbon is created as a by-product during cracking, the importance of which is constantly increasing as an industrial raw material.
What is the effect of pressure on cracking?
Increased pressure decreased the cracking of the unstable molecular classes of the LAF and also secondary cracking of some newly formed heavier compounds (C15–C20 and C20+ polyaromatics).



