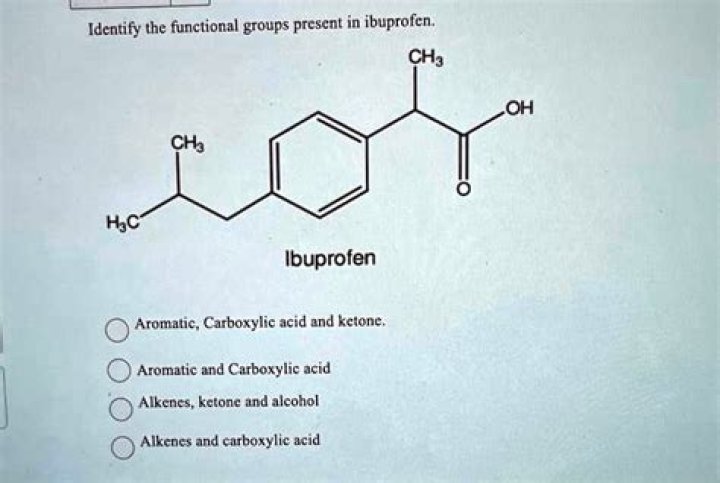
What are the functional groups of ibuprofen

Olivia Owen
Published Mar 30, 2026
Question 2 What functional groups does ibuprofen have? A carboxylic acid and an aromatic ring.
Which functional groups are present in ibuprofen?
- carboxyl group (COOH)
- aromatic group (benzene ring)
What is the functional group for Tylenol?
The functional groups in acetaminophen are hydroxyl, aromatic ring, and amide.
What is the function of ibuprofen?
Nonprescription ibuprofen is used to reduce fever and to relieve minor aches and pain from headaches, muscle aches, arthritis, menstrual periods, the common cold, toothaches, and backaches. Ibuprofen is in a class of medications called NSAIDs.What functional groups are in aspirin?
There are three different functional groups in aspirin, which contribute to its properties, a weak acid soluble in basic solutions. These functional groups include carboxylic acid, ester, and an aromatic group.
Is methyl a functional group?
The methyl group is the only nonpolar functional group in our class list above. The methyl group consists of a carbon atom bound to three hydrogen atoms. … This means that methyl groups are unable to form hydrogen bonds and will not interact with polar compounds such as water.
Is ibuprofen ionic or covalent?
Example 1: Computing Molecular Mass for a Covalent Compound Ibuprofen, C13H18O2, is a covalent compound and the active ingredient in several popular nonprescription pain medications, such as Advil and Motrin.
What is the content of ibuprofen?
Ibuprofen tablets contain the active ingredient Ibuprofen, which is (±) – 2 – (p – isobutylphenyl) propionic acid. Ibuprofen is a white powder with a melting point of 74-77°C and is very slightly soluble in water (<1 mg/mL) and readily soluble in organic solvents such as ethanol and acetone.What is the mechanism of action of ibuprofen?
Mechanism of action Ibuprofen is a non-selective inhibitor of an enzyme called cyclooxygenase (COX), which is required for the synthesis of prostaglandins via the arachidonic acid pathway. COX is needed to convert arachidonic acid to prostaglandin H2 (PGH2) in the body. PGH2 is then converted to prostaglandins.
What is ibuprofen 400mg used for?It can be used to relieve headaches, rheumatic and muscular pain, pain from non-serious arthritic conditions, backache, migraine, period pain, dental pain and neuralgia. It can also be used to reduce fever and relieve the symptoms of colds and flu.
Article first time published onHow many functional groups are there in paracetamol?
Paracetamol contains three functional groups: the hydroxyl group (OH), the amide group (HN-CO-R), and the aromatic group (benzene ring), as shown in Fig. …
What functional groups are in ketoprofen?
Ketoprofen is an oxo monocarboxylic acid that consists of propionic acid substituted by a 3-benzoylphenyl group at position 2. It has a role as a non-steroidal anti-inflammatory drug, an antipyretic, an EC 1.14.
What is aromatic functional group?
Aromatics. The additional functional group that contains only carbon and hydrogen is an aromatic ring which is a six-carbon ring with alternative double bonds. The aromatic ring is also shown as a ring with a circle in the middle representing the double bonds. … 2: Aromatic rings contain alternating double bonds.
What are the functional groups in naproxen?
The chemical structures of naproxen, ibuprofen and oxybenzone are shown in Fig. 1. The three PPCPs have various functional groups such as free carboxylic acid, phenol, ketone and ether groups that can interact effectively with adsorbents such as functionalized MOFs.
Is benzene ring a functional group?
Benzene ring: An aromatic functional group characterized by a ring of six carbon atoms, bonded by alternating single and double bonds. A benzene ring with a single substituent is called a phenyl group (Ph).
What is salicylic acid functional group?
Structure of Salicylic Acid Its IUPAC name is 2-hydroxybenzoic acid. It contains a hydroxyl group (–OH group) attached at the ortho position with respect to the carboxylic acid functional group(–COOH group) present on the benzene ring. The molecular weight (or molar mass) of Salicylic Acid is 138.12 g/mol.
Is ibuprofen a carboxylic acid?
Ibuprofen is a monocarboxylic acid that is propionic acid in which one of the hydrogens at position 2 is substituted by a 4-(2-methylpropyl)phenyl group.
What are the 7 functional groups?
Functional groups include: hydroxyl, methyl, carbonyl, carboxyl, amino, phosphate, and sulfhydryl.
Is ethyl a functional group?
Chemical classEsterGroupEsterFormulaRCO2R’Prefix/ExampleEthyl acetate (Ethyl ethanoate)
Is alkene a functional group?
NumberPrefix7hepta
What are the pharmacodynamics of ibuprofen?
Pharmacodynamics of Ibuprofen Ibuprofen acts through stopping the creation of certain prostaglandins, which are lipids that deal with injury or illness and are active in the pathways that control inflammation, pain, and fever.
How does ibuprofen inhibit nociception?
Ibuprofen may activate anti-nociceptive axis through binding to the cannabinoid receptors and through inhibition of fatty acid amide hydrolase (FAAH) that metabolizes endocannabinoid anandamide.
Is Ibuprofen a vasodilator or vasoconstrictor?
Compared to aspirin, ibuprofen has a milder and shorter-lived antiplatelet effect. It has also been shown that it can have a vasodilator effect, dilating mainly coronary arteries but also others can be effected.
What are the physical properties of ibuprofen?
Clinical dataDensity1.03 g/cm3Melting point75 to 78 °C (167 to 172 °F)Boiling point157 °C (315 °F) at 4 mmHgSolubility in water0.021 mg/mL (20 °C)
Is ibuprofen acidic or basic?
Ibuprofen is a relatively weak acid (pka 4.4) and its solubility in water or at acid pHis very low. This results in a relatively long residence time in the acid environment of the stomach, which slows down absorption of the substance.
Why is ibuprofen so bad for you?
Ibuprofen changes your body’s production of prostaglandins. This change can lead to an imbalance in your body fluid pressure, which can decrease your kidney function and increase your blood pressure. Symptoms of decreased kidney function include: increased blood pressure.
What is 800mg of ibuprofen used for?
IBUPROFEN (eye BYOO proe fen) is a non-steroidal anti-inflammatory drug (NSAID). It is used for dental pain, fever, headaches or migraines, osteoarthritis, rheumatoid arthritis, or painful monthly periods.
Can I take two 800 mg ibuprofen at once?
Use exactly as directed on the label, or as prescribed by your doctor. Use the lowest dose that is effective in treating your condition. An ibuprofen overdose can damage your stomach or intestines. The maximum amount of ibuprofen for adults is 800 milligrams per dose or 3200 mg per day (4 maximum doses).
Is phenol a functional group?
Phenyl is a functional group with an aromatic ring bonded to another group. And, phenol is a molecule that is just a phenyl bonded to a hydroxyl group. However, some sources consider phenol itself a functional group.
How many functional groups does caffeine have?
So caffeine contains two amine and two amide functional groups.
What is the functional group of aldehyde?
Aldehydes and ketones are organic compounds which incorporate a carbonyl functional group, C=O. The carbon atom of this group has two remaining bonds that may be occupied by hydrogen or alkyl or aryl substituents.