What are the properties of Group 6 elements

Andrew Campbell
Published Mar 07, 2026
O, S, Se, Te and Po are the elements of group VI-A.They have six electrons in their outermost shell.Their oxidation number is (–2).They have high values of electronegativity.Oxygen and sulphur are non-metals, Se and Te are metalloids, but Po is a metal.Elements of group VI-A show allotropy.
What is Group 6 of the periodic table?
A Group 6 element is one in the series of elements in group 6 (IUPAC style) in the periodic table, which consists of the transition metals chromium (Cr), molybdenum (Mo), tungsten (W), and seaborgium (Sg).
What are the 6 properties of metals?
- high melting points.
- good conductors of electricity.
- good conductors of heat.
- high density.
- malleable.
- ductile.
What are the properties of elements in a group?
- Atomic Size: From top to bottom in a group, the size of atom increases as a new shell to the atom.
- Valence Electrons(V.E) : Number of valence electrons in a group are same.
- Valency: All elements in a group generally have the same valency.
What are some properties of halogens?
- They have very high electronegativities.
- They have seven valence electrons (one short of a stable octet).
- They are highly reactive, especially with alkali metals and alkaline earths. …
- Because they are so reactive, elemental halogens are toxic and potentially lethal.
Which element in Group 6 is the least reactive?
Chemical Properties of Noble Gases Noble gases are the least reactive of all known elements. Their outer energy levels are full because they each have eight valence electrons. The only exception is helium, which has just two electrons.
What is Group 6 Period 6 on the periodic table?
The period 6 transition metals are lanthanum (La), hafnium (Hf), tantalum (Ta), tungsten (W), rhenium (Re), osmium (Os), iridium (Ir), platinum (Pt), gold (Au), and mercury (Hg).
What are the properties of group 8 elements?
- They are inert gases located on the right of the periodic table.
- They have a full-set of valence electrons, so they’re stable.
- They are colorless, odorless and tasteless.
- They have low melting and low boiling points.
What are properties of elements?
All elements have properties. Those properties include, but are not limited to, conductivity, magnetism, melting point, boiling point, color, state of matter, and others. Elements with similar properties are grouped together in different areas of the periodic table of elements.
What element is in group 10 Period 6?Group10Period428 Ni546 Pd678 Pt
Article first time published onWhat six elements have the appearance and properties of metal?
A series of six elements called the metalloids separate the metals from the nonmetals in the periodic table. The metalloids are boron, silicon, germanium, arsenic, antimony, and tellurium. These elements look metallic; however, they do not conduct electricity as well as metals so they are semiconductors.
What are the 5 properties of metal?
Metals are lustrous, malleable, ductile, good conductors of heat and electricity.
What are the 10 properties of metals?
- Metals can be hammered into thin sheets. …
- Metals are ductile. …
- Metals are a good conductor of heat and electricity.
- Metals are lustrous which means they have a shiny appearance.
- Metals have high tensile strength. …
- Metals are sonorous. …
- Metals are hard.
What are the chemical and physical properties of halogens?
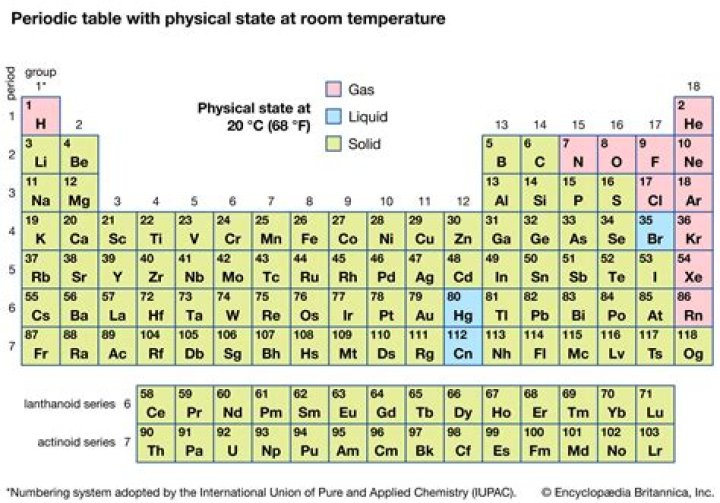
Halogens display physical and chemical properties typical of nonmetals. They have relatively low melting and boiling points that increase steadily down the group. Near room temperature, the halogens span all of the physical states: Fluorine and chlorine are gases, bromine is a liquid, and iodine is a solid.
What are the properties of group 7?
- At room temperature (20 °C), the physical state of the halogens changes as you go down the group. Fluorine and chlorine are gases, bromine is a liquid and iodine is crumbly solid.
- The colours of the halogens also change as you descend the group – they become darker.
What are the general properties of metalloids?
Properties. Metalloids usually look like metals but behave largely like nonmetals. Physically, they are shiny, brittle solids with intermediate to relatively good electrical conductivity and the electronic band structure of a semimetal or semiconductor.
What is Group 6 called?
Group 6, numbered by IUPAC style, is a group of elements in the periodic table. Its members are chromium (Cr), molybdenum (Mo), tungsten (W), and seaborgium (Sg). These are all transition metals and chromium, molybdenum and tungsten are refractory metals. … That group is now called group 16.
What is the most reactive element in Group 6?
Cesium is second from the bottom of this group, with 6 electron shells, so it fits all the characteristics of a reactive atom, therefore making it the most reactive element.
Which is the only element of Group 6 forms hydride?
Only one element of group 6 i.e., Cr forms hydride.
What are the properties of a noble gas?
Noble gases are colorless, odorless, tasteless, and nonflammable gases under standard conditions. In the periodic table, the noble gases are arranged according to their boiling point. Noble gases are widely used in different fields, from incandescent lighting to excimer lasers.
Why are noble gases gases?
They are all gases because in their natural state they have a complete octet of outer electrons and don’t need another atom to be complete. They are the only atoms which do not combine with themselves like oxygen is O2 and fluorine is F2.
What makes a noble gas?
Noble gases, most often found as monatomic gases, have completely filled outer electron shells, so have no inclination to react with other elements, thus very rarely forming compounds with other elements.
What are the two properties of elements?
These properties include color, density, melting point, boiling point, and thermal and electrical conductivity. While some of these properties are due chiefly to the electronic structure of the element, others are more closely related to properties of the nucleus, e.g., mass number.
What are the 5 properties of the periodic table?
Major periodic trends include electronegativity, ionization energy, electron affinity, atomic radii, ionic radius, metallic character, and chemical reactivity.
What are the properties of group 3 elements?
All the group 3 elements are rather soft, silvery-white metals, although their hardness increases with atomic number. They quickly tarnish in air and react with water, though their reactivity is masked by the formation of an oxide layer.
What are the properties of Group 1?
Group one elements share common characteristics. They are all soft, silver metals. Due to their low ionization energy, these metals have low melting points and are highly reactive. The reactivity of this family increases as you move down the table.
What are the properties of Group 0?
Group 0 Elements the outer shell, apart from Helium which has 2 – stable full outer shell. Unreactive. They are very unreactive. This means they don’t form molecules easily, so elements are found as single atoms.
What are Group 7 elements called?
Group 7A (or VIIA) of the periodic table are the halogens: fluorine (F), chlorine (Cl), bromine (Br), iodine (I), and astatine (At). The name “halogen” means “salt former”, derived from the Greek words halo- (“salt”) and -gen (“formation”).
What is the properties of transition metals?
- they are good conductors of heat and electricity.
- they can be hammered or bent into shape easily.
- they have high melting points (but mercury is a liquid at room temperature)
- they are usually hard and tough.
- they have high densities.
How many elements are there in sixth period?
The sixth period contains 32 elements, tied for the most with period 7, beginning with caesium and ending with radon.
What element is in group 15 Period 6?
BismuthGroupgroup 15 (pnictogens)Periodperiod 6Blockp-blockElectron configuration[Xe] 4f14 5d10 6s2 6p3



