What are the reactions of benzene

Olivia Owen
Published Mar 15, 2026
Reaction TypeTypical EquationNitration:C6H6+ HNO3 & heat H2SO4 catalystSulfonation:C6H6+ H2SO4 + SO3 & heatAlkylation: Friedel-CraftsC6H6+ R-Cl & heat AlCl3 catalystAcylation: Friedel-CraftsC6H6+ RCOCl & heat AlCl3 catalyst
What is the most common reaction of benzene?
The most characteristic reaction of benzene and many of its derivatives is electrophilic aro- matic substitution. In an electrophilic aromatic substitution reaction, a hydrogen of an aro- matic ring is substituted by an electrophile—that is, by a Lewis acid.
Which type of reactions are not given by benzene?
It does not undergo the addition, oxidation, and reduction reactions characteristic of alkenes. For example, benzene does not react with bromine, hydrogen chloride, or other reagents that usually add to carbon–carbon double bonds.
What type reaction does benzene undergo?
Benzene is a planar molecule having delocalized electrons above and below the plane of the ring. Hence, it is electron-rich. As a result, it is highly attractive to electron-deficient species i.e., electrophiles. Therefore, it undergoes electrophilic substitution reactions very easily.What are aromatic reactions?
Aromatic compounds or arenes undergo substitution reactions, in which the aromatic hydrogen is replaced with an electrophile, hence their reactions proceed via electrophilic substitution.
What are the most characteristic reactions of benzene give two examples?
- Halogenation of Benzene. By the means of electrophilic aromatic substitution reaction, one hydrogen atom of the arene is substituted by one halogen atom. …
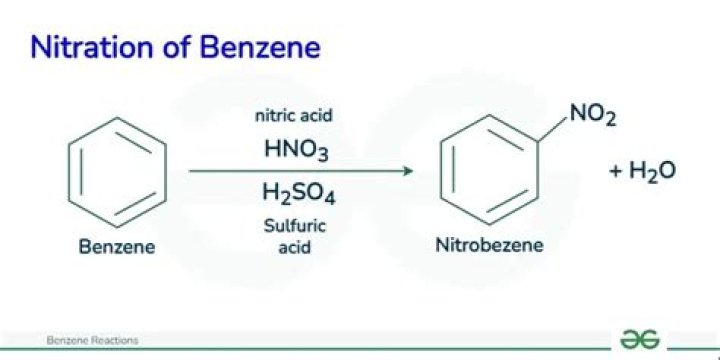
- Nitration of Benzene. …
- Sulfonation of Benzene. …
- Alkylation and Acylation of Benzene.
What are the most common reaction of benzene and its derivatives?
Most common reactions of benzene (aromatic hydrocarbon) and its derivatives are. Electrophilic addition reactions. Electrophilic substitution reactions. Nucleophilic addition reactions.
Does benzene undergo combustion reactions?
As is the case with all hydrocarbons, which are compounds that only contain carbon and hydrogen, benzene’s combustion will lead to the formation of only two products, carbon dioxide, CO2 , and water, H2O .Does benzene undergo addition reaction?
Benzene, C6H6, is a planar molecule containing a ring of six carbon atoms each with a hydrogen atom attached. … The presence of the delocalised electrons makes benzene particularly stable. Benzene resists addition reactions because that would involve breaking the delocalisation and losing that stability.
Does benzene undergo sn1 reaction?Generally benzene group does not undergo nucleophilic substitution due to repulsion between benzene pi electrons and negative charge of incoming nucleophile.
Article first time published onDoes benzene test bromine water?
Benzene does not react with any electrophilic addition reactions hence it does undergo bromine water test, as they have delocalised pi bonds. Therefore, it does not decolourise the bromine water.
What is resonance in benzene?
Resonance in benzene: Benzene ring has three double bonds in it and is expected to be quite reactive. … Due to resonance in benzene, the carbon-carbon bonds in benzene acquire an intermediate character of carbon-carbon single and double bonds.
Why does benzene resist addition reaction?
Benzene is an aromatic compound and is stabilized due to resonance or delocalization of π electrons. When it undergoes addition reactions, it will lose resonance stabilization. Hence it resists electrophilic additions to double bonds in it.
Why does c6h6 not react with HCl?
With HCl, benzene would never react. Because HCl is an inorganic acid, it lacks an electrophile to replace benzene’s H atom. Because Cl is a Lewis base, it does not behave as an electrophile.
What are Electrophiles examples?
Electrophiles are atoms or molecules known to be electron deficient and that carry a partial or fully positive charge and will seek an electron pair to form a covalent bond . An example of an electrophile is a Lewis Acid. Other examples include Br+, Cl+, and CH3+.
What is acylation of benzene?
Hint: Acylation is the substitution of an acyl group into an organic compound. In case of benzene, the acyl group is substituted into the benzene ring. This reaction is also known as Friedel-Crafts acylation of Benzene.
What will be the product of the reaction between benzene and ch3cocl in the presence of alcl3?
When benzene reacts with ch3cocl in the presence of alcl3 Acetophenone(c6h5coch3) will form. This is a electrophilic substitution reaction. It is popularly named as as friedal Craft’s acylation reaction.
What is Electrophile in nitration of benzene?
The electrophile is the “nitronium ion” or the “nitryl cation”, NO+2. This is formed by reaction between the nitric acid and the sulphuric acid.
Is ch3 ortho or meta directing?
1: Thus, the methyl group is an ortho, para directing group.
What is the common name of benzene?
NamesPreferred IUPAC name BenzeneOther names Benzol (historic/German) Cyclohexa-1,3,5-triene; 1,3,5-Cyclohexatriene [6]Annulene (not recommended)IdentifiersCAS Number71-43-2
Which is the most reactive for SE reaction?
−NH2 group is the most activating group which is present in aniline (C6H5−NH2) hence it is the most reactive towards electrophilic substitution reaction.
Which is the characteristic reaction of benzene?
The most characteristic reaction of benzene are the substitution reactions that occur when it reacts with electrophilic reagents in these reactions there is more or less no loss of the resonance energy.
Does benzene undergo oxidation reaction?
Alkyl groups are usually fairly resistant to oxidation. However, when they are attached to a benzene ring, they are easily oxidised by an alkaline solution of potassium manganate(VII) (potassium permanganate). … Interestingly, any alkyl group is oxidised back to a -COOH group on the ring under these conditions.
What is the most common reaction of aromatic compounds?
Electrophilic substitution (SE) is the most frequent reaction type found with aromatic compounds. In the classic case, a hydrogen atom is exchanged for an electrophile.
How does benzene burn air?
When benzene undergoes combustion in air, the carbon and hydrogen atoms combine with the molecules of oxygen present in air to form carbon dioxide and water respectively.
Is benzene a gas?
Benzene is a colorless, flammable liquid with a sweet odor. It evaporates quickly when exposed to air. Benzene is formed from natural processes, such as volcanoes and forest fires, but most exposure to benzene results from human activities. Benzene is among the 20 most widely used chemicals in the United States.
Does benzene give sooty flame?
More percentage of carbon requires more oxygen for combustion. … But benzene is an aromatic compound having relatively more carbon content (carbon to hydrogen ratio). So it does not get completely oxidized during combustion and gives out sooty flames.
Does benzene undergo resonance?
Benzene shows resonance. It is highly inflammable and burns with a sooty flame.
Can benzene rings undergo sn2?
The mechanism of SN2 reaction doesn’t occur due to steric hindrance of the benzene ring. In order to attack the C atom, the nucleophile must approach in line with the C-LG (leaving group) bond from the back, where the benzene ring lies.
What is meant by addition reaction?
addition reaction, any of a class of chemical reactions in which an atom or group of atoms is added to a molecule. … An addition reaction may be visualized as a process by which the double or triple bonds are fully or partially broken in order to accommodate additional atoms or groups of atoms in the molecule.
How does benzene react with bromine?
Benzene reacts with chlorine or bromine in the presence of a catalyst, replacing one of the hydrogen atoms on the ring by a chlorine or bromine atom. … It reacts with some of the chlorine or bromine to form iron(III) chloride, FeCl3, or iron(III) bromide, FeBr3.