What causes electron affinity

Nathan Sanders
Published Mar 20, 2026
The electron affinity is a measure of the attraction between the incoming electron and the nucleus – the stronger the attraction, the more energy is released. The factors which affect this attraction are exactly the same as those relating to ionization energies – nuclear charge, distance and screening.
What affects electron affinity?
There are two factors that can affect electron affinity. These are atomic size and nuclear charge. … The smaller the atom is, the closer the outermost shell is; therefore, it is a stronger attraction between the nucleus and the incoming electron. That means the electron affinity is higher for smaller atoms.
How does electron affinity change in the periodic table?
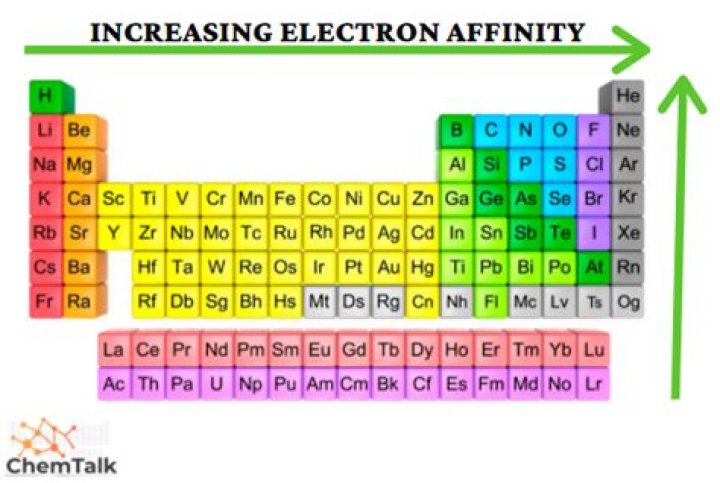
Electron affinity generally increases across a period in the periodic table and sometimes decreases down a group. … The chemical rationale for changes in electron affinity across the periodic table is the increased effective nuclear charge across a period and up a group.
What causes electron affinity trend?
Electron affinity increases from left to right within a period. This is caused by the decrease in atomic radius. Electron affinity decreases from top to bottom within a group. This is caused by the increase in atomic radius.Why does chlorine have the highest electron affinity?
Chlorine has the highest electron affinity among the elements. Its high affinity can be attributed to its large atomic radius, or size. Because chlorine’s outermost orbital is 3p, its electrons have a large amount of space to share with an incoming electron.
What is electron affinity What signs are associated?
Electron Affinity is the energy associated with the addition of an electon to a gaseous atom. Notice the sign on the energy is negative. This is because energy is usually released in this process, as opposed to ionization energy, which requires energy.
Why is electron affinity negative or positive?
Electron affinities are negative numbers because energy is released. The elements of the halogen group (Group 17) gain electrons most readily, as can be seen from their large negative electron affinities. This means that more energy is released in the formation of a halide ion than for the anions of any other elements.
What is the electron affinity of fluorine?
Z9ElementFNameFluorineElectron affinity (eV)3.401 189 8(24)Electron affinity (kJ/mol)328.164 9(3)What is electron affinity and electron gain enthalpy?
Electron affinity is the amount of energy released when a neutral atom or a molecule (in the gaseous phase) gains an electron from the outside. Electron gain enthalpy is the change in enthalpy when a neutral atom or a molecule gains an electron from outside.
Which has maximum electron affinity?Noble gases have maximum electron affinity.
Article first time published onWhy does electron affinity increase across a period and decrease down a group?
This is because as you go down the period table, new valence shells are added increasing the atomic radius. The new orbital is further away from the nucleus, meaning the attraction between the positively charged nucleus and the new electron decrease. Thus, electron affinity decreases.
What element has the smallest electron affinity?
Of the metals, mercury has the lowest electron affinity.
What is electron affinity in simple terms?
Definition of electron affinity 1 : the degree to which an atom or molecule attracts additional electrons. 2 : the minimum energy required to remove an electron from a negative ion to produce a neutral atom or molecule.
Is electron affinity electronegativity?
Electronegativity refers to the ability of the atoms to attract the electrons from the other elements. Electron affinity refers to the amount of energy that is liberated whenever a molecule or a neutral atom tends to acquire an electron from the other elements.
Why do metals have low electron affinity?
Metals have a less likely chance to gain electrons because it is easier to lose their valance electrons and form cations. It is easier to lose their valence electrons because metals’ nuclei do not have a strong pull on their valence electrons. Thus, metals are known to have lower electron affinities.
Why does fluorine not have the highest electron affinity?
Fluorine, though higher than chlorine in the periodic table, has a very small atomic size. This makes the fluoride anion so formed unstable (highly reactive) due to a very high charge/mass ratio. … As a result, fluorine has less electron affinity .
Do noble gases have electron affinity?
Electron affinity of noble gas is near zero. Noble gas has completely filled valence shells and stable octets. It does not accept electrons easily as they have no deficiency, also adding electrons produces repulsion between the electrons.
Why are some electron affinities positive?
Unlike ionization energies, which are always positive for a neutral atom because energy is required to remove an electron, electron affinities can be negative (energy is released when an electron is added), positive (energy must be added to the system to produce an anion), or zero (the process is energetically neutral) …
Which atom has the most exothermic electron affinity?
The trend for electron affinity is as follows: it increases from left to right and decreases down a period in the periodic table. The element that has the most exothermic electron affinity is c) Cl.
Why is electron affinity exothermic?
1st Electron Affinity is usually exothermic as the energy released when the nucleus attracts the the additional electron is larger than the energy absorbed to overcome inter-electronic repulsion.
What are the factors on which electron gain enthalpy depends?
- Browse more Topics under Classification Of Elements And Periodicity In Properties. …
- 1) Atomic Size. …
- 2) Nuclear charge. …
- 3) Electronic configuration. …
- The Electron Gain Enthalpy of Fluorine is Less Negative than that of Chlorine.
Which factors affect electron gain enthalpy?
- Effective Nuclear charge is directly proportional to nuclear charge . …
- Atomic size : Electron gain enthalpy is inversely proportional to atomic size , with increase in atomic size , value of electron gain enthalpy decrease .
Which has highest electron gain enthalpy?
Halogens have the highest electron gain enthalpy.
Why is the electron affinity of nitrogen zero?
Electron affinity of Nitrogen is zero due to its half-filled configuration of 2p-orbital, which gives stability to it. Half filled configuration in Nitrogen has gives stability to it, thus its electron affinity is Zero.
How does electron affinity affect reactivity?
In general, reactivity increases as electron affinity increases. … The greater the tendency to gain electrons, the more reactive an element is.
What is chlorine electron affinity?
Electron Affinity of Chlorine is 349 kJ/mol.
Which family of elements has high electron affinity?
Atoms, such as Group 7 elements, whose anions are more stable than neutral atoms have a higher electron affinity. The electron affinities of the noble gases have not been conclusively measured, so they may or may not have slightly negative values. Chlorine has the highest electron affinity while mercury has the lowest.
Is electron affinity maximum in alkali metals?
Electron affinity is defined as the amount of energy release or absorb when an extra electron is added to the neutral atom or molecule in the gaseous state to form a negative ion. … There is no effect of electron affinity in alkali and and alkaline earth metals.
Which has higher electron affinity fluorine or neon?
In the modern periodic table, electron affinity increases on moving left to right in a period while it decreases on moving down the group. … Thus, noble gases have the least electron affinity in a period. Hence, we can conclude that in a period, fluorine (halogen) has higher electron affinity than neon (noble gas).
Why does electron affinity only apply to gases?
Keep in mind, electron affinity values only apply to gaseous atoms and molecules because the electron energy levels of liquids and solids are altered by interaction with other atoms and molecules.
What is the other name of electron affinity?
Electron gain enthalpy is sometimes also referred to as Electron affinity although there is a minute difference between them. Electron gain enthalpy is defined as the amount of energy released when an electron is added to an isolated gaseous atom.