What do you mean by molecular polarizability

Isabella Wilson
Published May 10, 2026
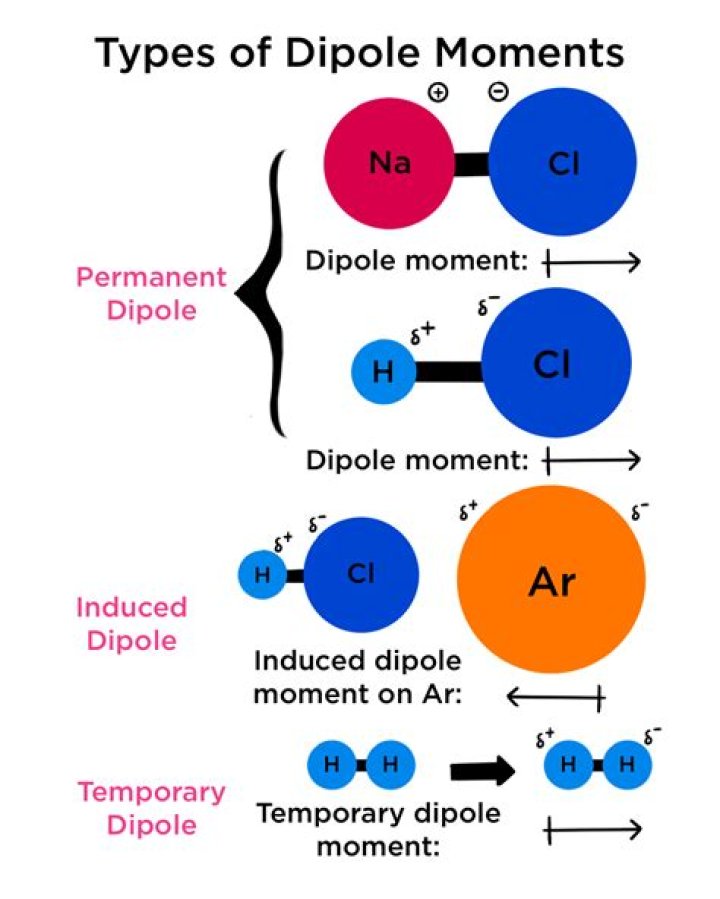
The polarizability of an atom or molecule is defined as the ratio of its induced dipole moment to the local electric field; in a crystalline solid, one considers the dipole moment per unit cell.
How do you determine molecular polarizability?
In their studies, polarizability was simply calculated by adding up the number of valence electrons (NVE) in a molecule: H = 1, C = 4, N = 5, P = 5, O = 6, S = 6 and halogens = 7.
What affects Polarizability?
Molecular orientation, atomic radii, and electron density are the main three factors that influence the Polarizability in the following way: · As the number of electrons increases, the control on distribution of charge by the nuclear charges becomes less, and thus the Polarizability of atom increased.
What is the unit of molecular polarizability?
Nm2C−1.What is Polarizability and polarizing power?
The ability of a cation to distort the electron density of anion towards itself or we can say that ability of cation to pull the electron density of anion towards itself is known as Polarizing power. And the tendency by which the anion undergoes polarization is known as Polarizability.
What is polarizability tensor?
A polarizability tensor of a nanoparticle relates the incident homogeneous electric field with the induced dipolar moment. For some particles, such as spheres or ellipsoids, the polarizability tensor is diagonal (in the frame of reference of the particle axes). … All you need it to choose a suitable frame of reference.
What increases polarizability?
Factors that Influence Polarizability The greater the number of electrons, the less control the nuclear charge has on charge distribution, and thus the increased polarizability of the atom.
Which ion is most Polarising?
Polarizing power depends on cation size and cation charge, so larger cation charge and lower cation size have greater polarising power. Al+3, thus has the highest polarising power.What is the strongest molecular force?
The strongest intermolecular force is hydrogen bonding, which is a particular subset of dipole-dipole interactions that occur when a hydrogen is in close proximity (bound to) a highly electronegative element (namely oxygen, nitrogen, or fluorine).
What is polarizability in organic chemistry?Illustrated Glossary of Organic Chemistry – Polarizability. Polarizability: The ability of a bond or molecule to be polarized by distorting its electron cloud. … When polarization occurs easily, the species is said to be soft. When polarization occurs only with difficulty the species is said to be hard.
Article first time published onWhat is polarization in chemistry BYJU's?
Polarization is defined as the property of electromagnetic radiations where the relationship between the direction and magnitude of the vibrating electric field is explained.
What do you mean by polarization?
polarization, property of certain electromagnetic radiations in which the direction and magnitude of the vibrating electric field are related in a specified way. … Light may be polarized by reflection or by passing it through filters, such as certain crystals, that transmit vibration in one plane but not in others.
What is polarization Class 11?
Distortion of electron cloud of the negatively charged ion by the positively charged ion is called Polarization. The Extent of polarization: Is given by Fazan rule. According to Fazan, Smaller the size of ion, more it can distort the electron cloud of other. Hence, more is the polarization.
What is polarization of ion?
Polarization in chemistry means deformation of symmetric electron charge cloud of anion by cation. … For example, Al3+ ion cause the high polarization of a particular anion than Na+ ion ,due to small size and high charge with respect to Na+ ion.
What is polarization in solid state physics?
Polarization occurs when an electric field distorts the negative cloud of electrons around positive atomic nuclei in a direction opposite the field. This slight separation of charge makes one side of the atom somewhat positive and the opposite side somewhat negative.
What are the 4 types of intermolecular forces?
12.6: Types of Intermolecular Forces- Dispersion, Dipole–Dipole, Hydrogen Bonding, and Ion-Dipole. To describe the intermolecular forces in liquids.
What intermolecular force is weakest?
The dispersion force is the weakest of all IMFs and the force is easily broken. However, the dispersion force can become very strong in a long molecule, even if the molecule is nonpolar.
What are the 5 types of intermolecular forces?
There are five types of intermolecular forces: ion-dipole forces, ion-induced-dipole forces, dipole-dipole forces, dipole-induced dipole forces and induced dipole forces.
How does Polarizability affect solubility?
Increase in solute dipolarity/polarizability increases solubility, whereas an increase in solute excess molar refraction, and especially, volume decrease solubility.
How do you calculate polarizing power?
According to Fajan’s rule, “the higher the charge on the cation, the more is its polarizing power.” The next factor which is considered is the size of the cation, “smaller the size of the cation, higher is its polarizing power.”
What is polarizability of an anion?
Polarizability represents the ability of an anion to become polarized. In order for an anion to become polarized, its electron cloud must be distorted. … This ultimately means that the electron clouds are very easy to distort, which is why larger anions are more polarizable than smaller anions.
What is polarization Class 12?
The light waves which travel only in a single plane are known as polarized light waves. The process of transforming unpolarized light waves to polarized light waves is called the polarisation of light. Complete step by step answer: … So one plane is occupied by an electric field and the other by the magnetic field.
What is polarization Class 8?
What is Polarization? Polarization, in Physics, is defined as a phenomenon caused due to the wave nature of electromagnetic radiation. Sunlight travels through the vacuum to reach the Earth, which is an example of an electromagnetic wave.
What is molecular polarity?
In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment, with a negatively charged end and a positively charged end. Polar molecules must contain one or more polar bonds due to a difference in electronegativity between the bonded atoms.
What is polarization in a bond?
Bond polarity in chemistry is the division of electric charge around a bond, leading to an electric dipole or dipole moment for a molecule or its chemical groups. … There is a permanent dipole that occurs in the bond because of this and it is said that the covalent bond is polarised.