What does all matter consist of according to the kinetic theory

Andrew White
Published May 02, 2026
The Kinetic Theory of Matter states that all matter is made up of atoms and molecules. These atoms and molecules act like tiny particles that are always in motion.
What is part of the kinetic theory of matter?
The kinetic molecular theory of matter states that: Matter is made up of particles that are constantly moving. All particles have energy, but the energy varies depending on the temperature the sample of matter is in. This in turn determines whether the substance exists in the solid, liquid, or gaseous state.
Does all matter have kinetic energy?
The energy of moving matter is called kinetic energy. Scientists think that the particles of all matter are in constant motion. In other words, the particles of matter have kinetic energy. The theory that all matter consists of constantly moving particles is called the kinetic theory of matter.
What are the 3 parts of the kinetic theory of matter?
- Part 1 kinetic theory. All matter is made up of tiny particles.
- Part 2 kinetic theory. These particles are always in motion.
- Part 3 kinetic theory. Particles with more mass move more slowly then particles with less mass.
What is kinetic theory of matter Class 8?
In Physics, the kinetic theory of matter states that matter should be made up of particles that must be in constant motion. All the particles should possess energy, and the energy of the particles depends on the temperature, which determines the existence of the substance in a solid, liquid, or gas state. 4.5 (12)
Which of the 3 main states of matter can be compressed?
Particles can move about within a liquid, but they are packed densely enough that volume is maintained. Gaseous matter is composed of particles packed so loosely that it has neither a defined shape nor a defined volume. A gas can be compressed.
What is matter made of?
At the most fundamental level, matter is composed of elementary particles known as quarks and leptons (the class of elementary particles that includes electrons). Quarks combine into protons and neutrons and, along with electrons, form atoms of the elements of the periodic table, such as hydrogen, oxygen, and iron.
What are the 4 assumptions of the kinetic theory?
The kinetic-molecular theory of gases assumes that ideal gas molecules (1) are constantly moving; (2) have negligible volume; (3) have negligible intermolecular forces; (4) undergo perfectly elastic collisions; and (5) have an average kinetic energy proportional to the ideal gas’s absolute temperature.What does the kinetic molecular theory explain at least 3 things?
Kinetic theory explains macroscopic properties of gases, such as pressure, temperature, viscosity, thermal conductivity, and volume, by considering their molecular composition and motion.
What does the kinetic theory of matter explain quizlet?Kinetic Theory of Matter. the idea that all matter is made up of molecules that are in constant motion. Plasma. A state of matter similar to gas in which a certain portion of the particles is ionized. You just studied 20 terms!
Article first time published onHow are states of matter defined?
In physics, a state of matter is one of the distinct forms in which matter can exist. Four states of matter are observable in everyday life: solid, liquid, gas, and plasma. … Matter in the liquid state maintains a fixed volume, but has a variable shape that adapts to fit its container.
What are matter properties?
Any characteristic that can be measured, such as an object’s density, color, mass, volume, length, malleability, melting point, hardness, odor, temperature, and more, are considered properties of matter.
What is kinetic theory in physics?
Kinetic theory explains the behaviour of gases based on the idea that the gas consists of rapidly moving atoms or molecules. … It gives a molecular interpretation of pressure and temperature of a gas, and is consistent with gas laws and Avogadro’s hypothesis. It correctly explains specific heat capacities of many gases.
What does the kinetic theory state apex?
The Kinetic Theory of gases states that the gases are made of small particles (atoms or molecules) which are in random motion.
What is all matter made up of atoms?
[Explanation] Idea a: All matter is made up of particles called atoms and molecules (as opposed to being continuous or just including particles). On the following page, the idea is stated as one of four concepts in Dalton’s theory: “All matter is composed of tiny, indivisible particles called atoms” (p. 158s).
Is everything made of matter?
Matter is the Stuff Around You Matter is everything around you. Atoms and compounds are all made of very small parts of matter. Those atoms go on to build the things you see and touch every day. Matter is defined as anything that has mass and takes up space (it has volume).
What is matter made of and how do we know?
All matter is made up of tiny particles called atoms. Matter takes on different forms depending on how the atoms are arranged. We call these forms “states of matter”. On Earth, the most common states are solids, liquids, and gases.
What is matter and its three states?
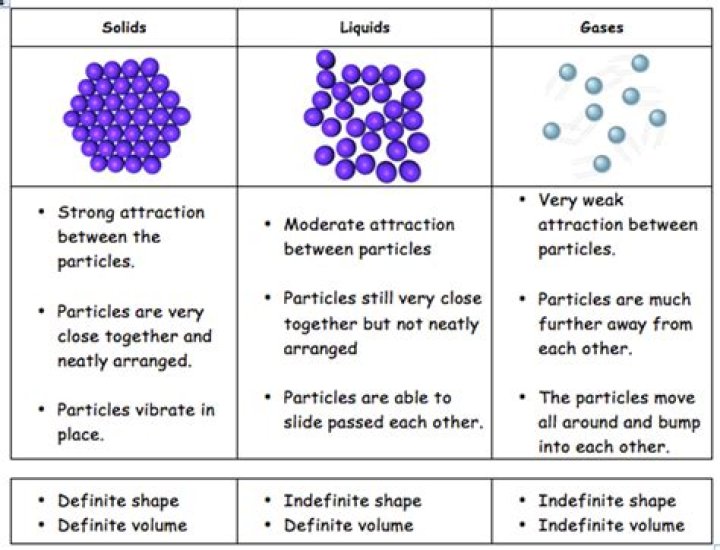
The three states of matter are solid, liquid and gas. The particle model represents particles by small, solid spheres. It describes the arrangement, movement and energy of particles in a substance. The model can be used to explain the physical properties of solids, liquids and gases.
What is matter write the three states of matter?
There are three states of matter: solid; liquid and gas. They have different properties, which can be explained by looking at the arrangement of their particles. This is the theoretical temperature at which particles have the least amount of energy and the slowest movement.
Why do the three states of matter differ?
The different states of matter are due to the variation in the characteristics of their constituent particles. The three states of matter, solid, liquid and gas differ in the way their atoms are arranged, their intermolecular distance and the intermolecular force of attraction between their particles.
How can you relate the kinetic molecular theory with the experiment?
The kinetic molecular theory can be used to explain each of the experimentally determined gas laws. The pressure of a gas results from collisions between the gas particles and the walls of the container. Each time a gas particle hits the wall, it exerts a force on the wall.
What happens to the temperature of a substance while it is changing state?
The temperature of a substance is related to the speed of the substance’s particles. So, when the temperature of a substance changes,the speed of the particles also changes. But the temperature of a substance does not change until the change of state is complete.
What state of matter are all minerals answer?
Crystalline solids, or crystals, are regarded as “true solids.” Minerals are crystalline solids. Common table salt is one example of this kind of solid. In crystalline solids, the atoms, ions or molecules are arranged in an ordered and symmetrical pattern that is repeated over the entire crystal.
What is a matter answer?
A common or traditional definition of matter is “anything that has mass and volume (occupies space)”. For example, a car would be said to be made of matter, as it has mass and volume (occupies space). The observation that matter occupies space goes back to antiquity.
How many phases of matter are there?
The three fundamental phases of matter are solid, liquid, and gas (vapour), but others are considered to exist, including crystalline, colloid, glassy, amorphous, and plasma phases.
What are changes of matter?
There are two types of change in matter: physical change and chemical change. … Many physical changes are reversible (such as heating and cooling), whereas chemical changes are often irreversible or only reversible with an additional chemical change.
What are the 3 basic properties of matter?
1. The three basic properties of matter are volume, mass, and shape. 2. All matter is made up of tiny particles called atoms.
What are the two properties of all matter?
Matter can be defined or described as anything that takes up space, and it is composed of miniscule particles called atoms. It must display the two properties of mass and volume.
Who proposed kinetic theory of matter?
The kinetic theory relates the independent motion of molecules to the mechanical and thermal properties of gases—namely, their pressure, volume, temperature, viscosity, and heat conductivity. Three men—Daniel Bernoulli in 1738, John Herapath in 1820, and John James Waterston in 1845—independently developed the theory.
What is the kinetic theory model?
The model, called the kinetic theory of gases, assumes that the molecules are very small relative to the distance between molecules. The molecules are in constant, random motion and frequently collide with each other and with the walls of any container. … The higher the temperature, the greater the motion.
What are the 5 assumptions of the kinetic molecular theory?
The five main postulates of the KMT are as follows: (1) the particles in a gas are in constant, random motion, (2) the combined volume of the particles is negligible, (3) the particles exert no forces on one another, (4) any collisions between the particles are completely elastic, and (5) the average kinetic energy of …