What does oxygen bind to in the hemoglobin molecule quizlet

Dylan Hughes
Published Mar 27, 2026
Heme-The deep red, nonprotein, iron-containing component of hemoglobin that carries oxygen. Heme is a porphyrin with an iron atom at its center. One of the free valence electrons of the iron atom of heme is bound to the hemoglobin molecule, while the other is available for binding to an oxygen atom.
What does oxygen bind to in the hemoglobin molecule?
Each subunit surrounds a central heme group that contains iron and binds one oxygen molecule, allowing each hemoglobin molecule to bind four oxygen molecules. … Iron associated with the heme binds oxygen. It is the iron in hemoglobin that gives blood its red color.
What molecules can bind to hemoglobin?
Aside from oxygen transport, hemoglobin can bind and transport other molecules like nitric oxide and carbon monoxide. Nitric oxide affects the walls of blood vessels, causing them to relax. This in turn reduces the blood pressure.
Where in the body does oxygen bind to hemoglobin quizlet?
Oxygen is loaded onto hemoglobin molecules in the lungs and unloaded from the hemoglobin molecules in the tissues.How is oxygen transported in the blood quizlet?
How is most oxygen carried in the blood? Hemoglobin transports 98% percent of our oxygen through our blood. … In the pulmonary capillaries, oxygen from the alveoli dissolves in the plasma. Dissolved O2 then diffuses into red blood cells, where it can bind to hemoglobin.
What is the oxygen binding site in hemoglobin and myoglobin?
The site at which oxygen binds to both hemoglobin and myoglobin is the heme shown in the figure below. At the center of the heme is an Fe(II) atom. Four of the six coordination sites around this atom are occupied by nitrogen atoms from a planar porphyrin ring.
How does oxygen binding change the quaternary structure of hemoglobin quizlet?
-Oxygen binding causes a change in the quaternary structure of hemoglobin where hemoglobin changes quaternary structure from the T (tense) state that has a low affinity of oxygen to the R (relaxed) state that has a higher affinity of oxygen.
How is oxygen transported around the body?
Inside the air sacs, oxygen moves across paper-thin walls to tiny blood vessels called capillaries and into your blood. A protein called haemoglobin in the red blood cells then carries the oxygen around your body.Which ion allows hemoglobin readily bind with oxygen quizlet?
–The Fe2+ ion binds to oxygen REVERSIBLY. This allows hemoglobin to pick up oxygen in the lungs and let go of it in the tissues.
How many oxygen molecules can one hemoglobin carry?The hemoglobin molecule has four binding sites for oxygen molecules: the iron atoms in the four heme groups. Thus, each Hb tetramer can bind four oxygen molecules.
Article first time published onWhy is it hard for the first oxygen to bind to hemoglobin?
Hemoglobin is made up of four symmetrical subunits and four heme groups. … It is easier to bind a second and third oxygen molecule to Hb than the first molecule. This is because the hemoglobin molecule changes its shape, or conformation, as oxygen binds. The fourth oxygen is then more difficult to bind.
What is the major way of that oxygen is transported in the blood?
Oxygen is transported in the blood in two ways: A small amount of O 2 (1.5 percent) is carried in the plasma as a dissolved gas. Most oxygen (98.5 percent) carried in the blood is bound to the protein hemoglobin in red blood cells. A fully saturated oxyhemoglobin (HbO 2) has four O 2 molecules attached.
How is oxygen transported in the blood a level biology?
How is oxygen transported? Oxygen is absorbed in the lungs from fresh air which has been breathed in. Red blood cells (erythrocytes) contain the rotein haemoglobin which can reversibly combine with oxygen. In the lungs, where the concentration of oxygen is high, haemoglobin will take up oxygen and form oxyhaemoglobin.
How does oxygen binding change the quaternary structure of hemoglobin?
Oxygenation causes such extensive quaternary structural changes to hemoglobin that oxy- and deoxyhemoglobin have different crystalline forms [7]. The quaternary structural changes preserve the exact 2-fold symmetry of hemoglobin and take place entirely across its α1-β2 (and α2-β1) interface.
When an oxygen atom binds to one of hemoglobin's four binding sites the affinity to oxygen of the three remaining available binding sites increases this example?
Hemoglobin (Hb) exhibits positive cooperativity. When one O2 molecule binds to one of hemoglobin’s four binding sites, the affinity to oxygen of the three remaining available binding sites increases; i.e. oxygen is more likely to bind to a hemoglobin bound to one oxygen than to an unbound hemoglobin.
Which of the following increases the affinity of oxygen to hemoglobin?
Increase the affinity of hemoglobin to oxygenDecrease the affinity of hemoglobin of oxygenb. Decreased temperaturea. Increased BPGd. Decreased carbon dioxide concentrationc. Glycolysisf. Hypothermiae. Decreased pHk. Increased pHg. Increased carbon dioxide concentration
Where are the binding sites for oxygen on a molecule of hemoglobin?
The Oxygen Binding Site of Hemoglobin The heme is located in a hydrophobic pocket of the protein. The iron atom is bonded to a fifth nitrogen atom in the side chain of the deprotonated proximal histidine residue.
What does the binding of oxygen by myoglobin depend on?
Myoglobin is a small oxygen-binding protein found in muscle cells. … Whether myoglobin binds to oxygen depends on the presence of the prosthetic group, heme. When myoglobin is able to bind to oxygen, it serves as the primary oxygen-carrying molecule in muscle tissue.
How does oxygen transfer from hemoglobin to myoglobin?
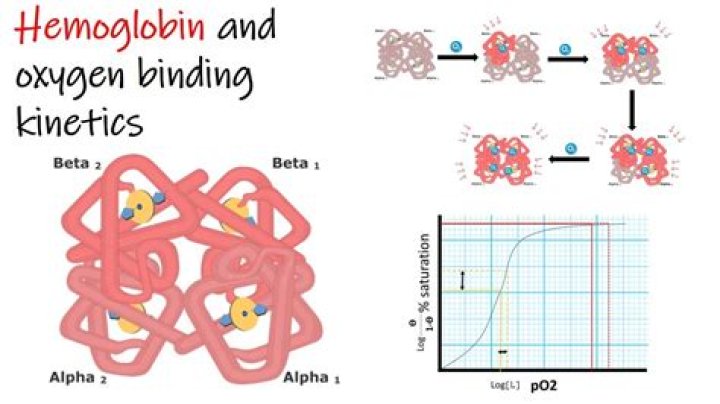
The O2-binding curve of hemoglobin is S shaped (Figure 4.2. … In the tissues, however, where the oxygen pressure is much lower, the decreased oxygen affinity of hemoglobin allows it to release O2, resulting in a net transfer of oxygen to myoglobin. Figure 4.2.
What happens to oxygen binding to hemoglobin when blood pH drops quizlet?
As pH decreases, hemoglobin’s affinity for oxygen decreases. As 2,3-BPG decreases, hemoglobin’s affinity for oxygen increases. Why is more oxygen unloaded from hemoglobin in an actively metabolizing tissue than in a resting tissue, even at the same concentration of O2?
What happens to oxygen binding to hemoglobin when temperature increases group of answer choices?
As it turns out, temperature affects the affinity, or binding strength, of hemoglobin for oxygen. Specifically, increased temperature decreases the affinity of hemoglobin for oxygen. As oxyhemoglobin is exposed to higher temperatures in the metabolizing tissues, affinity decreases and hemoglobin unloads oxygen.
What shape is the oxygen binding curve of hemoglobin?
Hence the curve has a sigmoidal or S-shape. At pressures above about 60 mmHg, the standard dissociation curve is relatively flat, which means that the oxygen content of the blood does not change significantly even with large increases in the oxygen partial pressure.
Which part of the hemoglobin molecule actually binds the oxygen molecule and which part binds carbon dioxide?
The part of the hemoglobin molecule that binds to oxygen is called the heme group. A heme group is a prosthetic group in the hemoglobin protein.
Which can bind o2 molecules?
The correct answer is Red Blood cells. Most oxygen,98.5 percent is bound to a protein called hemoglobin (which found in Red Blood Cells) and carried to the tissues. Hemoglobin(Hb), is a protein molecule found in red blood cells made of four subunits – two alpha subunits and two beta subunits.
How is oxygen transported in the blood and released to the tissues?
Oxygen transport within the human body occurs through both convection and diffusion. Within the pulmonary capillaries, one haemoglobin molecule binds up to four oxygen molecules in a cooperative manner.
Does oxygen bound to hemoglobin contribute to partial pressure?
It absolutely does affect the partial pressure of oxygen. Since the affinity of an oxygen molecule for heme increases as more oxygen is bound, as the partial pressure of oxygen increases a proportionally greater amount of oxygen molecules are bound.
How is oxygen loaded transported and unloaded in the blood?
Hemoglobin is loaded with oxygen as it passes through the pulmonary capillaries and is then transported to the peripheral tissues where the oxygen is unloaded. The primary factor determining whether oxygen is loaded or unloaded onto hemoglobin is the surrounding partial pressure of oxygen.
How does haemoglobin help in transport of oxygen to the cells?
The binding of oxygen to haemoglobin is in such a way that the binding of the first oxygen oxygen molecule binds to the haemoglobin, it increases the affinity for the second molecule of oxygen to bind. Subsequently, haemoglobin attracts more oxygen and this gives the curve a sigmoid shape.
How does haemoglobin load and unload oxygen in the body a level?
The process by which hemoglobin binds oxygen to form oxyhemoglobin is called loading. That’s what happens in the lungs. Once in the metabolizing tissues, oxyhemoglobin is unloaded as oxygen is released and diffuses into the plasma and ultimately our cells.
How does haemoglobin help in the transport of oxygen from lungs to tissues?
“How does haemoglobin help in the transport of oxygen from lung to tissue?” … Haemoglobin combines with oxygen and becomes oxyhaemoglobin in pulmonary capillaries. This oxygenated blood circulates in the body. When it reaches the tissues having low partial pressure of oxygen the oxygen is released into the tissues.
What does the binding of O2 to deoxy hemoglobin do?
Hence, when a single heme group in the hemoglobin protein becomes oxygenated, the whole protein changes its shape. … Thus, the binding of one molecule of O2 to hemoglobin enhances the ability of hemoglobin to bind more O2 molecules. This property of hemoglobin is known as “cooperative binding.”