What factor is thrombin

Ava Hall
Published Apr 28, 2026
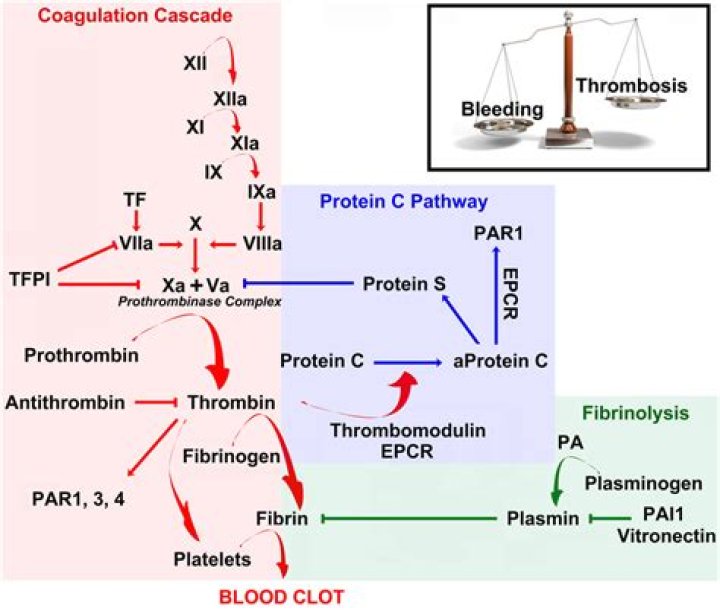
Prothrombin (factor II) is a soluble 72-kDa protein that is produced by the liver. It is activated to thrombin (factor IIa) via enzymatic cleavage of two sites by activated FX (FXa). Activated thrombin leads to cleavage of fibrinogen into fibrin monomers that, upon polymerization, form a fibrin clot.
Is thrombin a factor V?
Activated factor X is another factor upstream from factor V in the coagulation cascade. Thrombin is a downstream product of activated factor V and activated factor X activity and acts in a positive feedback manner to further increase the production of itself.
Is thrombin a tissue factor?
Available structuresPDBshowList of PDB id codes
What factor does thrombin activate?
Thrombin (FIIa) converts fibrinogen to fibrin and additionally activates factor XIII, which is important for cross-linking and stabilizing the fibrin clot (see Section II.What is factor 1 in blood clotting?
The following are coagulation factors and their common names: Factor I – fibrinogen. Factor II – prothrombin. Factor III – tissue thromboplastin (tissue factor)
Does thrombin activate Factor VII?
Autoimmune Inhibitors to Factor VII FVII binds to tissue factor (TF) and is activated by thrombin, FXIa, FXII, and FXa.
What is prothrombin factor?
The protein produced from the F2 gene, prothrombin (also called coagulation factor II), is the precursor to a protein called thrombin that initiates a series of chemical reactions in order to form a blood clot.
Does thrombin activate clotting factors?
Thrombin is an unique molecule that functions both as a procoagulant and anticoagulant. In its procoagulant role it activates platelets through its receptor on the platelets. It regulates its own generation by activating coagulation factors V, VIII and even XI resulting in a burst of thrombin formation.Is thrombin an enzyme?
Thrombin, the key enzyme of blood coagulation, is a Na+-activated allosteric serine protease (Wells and Di Cera 1992; Di Cera 2003; Di Cera et al. … 2000), it is not surprising that thrombin itself retains signatures of its descent from a growth factor.
Is prothrombin an enzyme complex?Prothrombin is activated by a membrane-bound enzyme complex Prothrombinase assembles through reversible interactions between the serine proteinase Xa and the protein cofactor Va on membranes containing phosphatidylserine.
Article first time published onWhat is factor 7 used for?
Factor VII, also called proconvertin, is one such clotting factor produced by the liver. It requires vitamin K for its production. Along with other clotting factors and blood cells, it promotes blood clotting at the site of an injury. It forms normal blood clots and closes the wound to prevent blood loss.
Is tissue factor Factor III?
Coagulation Factor III, also known as Tissue Factor (TF), Thromboplastin, and CD142, is the primary initiator of the extrinsic coagulation pathway. … Coagulation Factor III plays an important role in thrombotic diseases, disseminated intravascular coagulation, and atherosclerosis.
What is Factor 7a?
Factor VIIa is used to treat and prevent bleeding episodes in patients with Hemophilia A or B who have formed antibodies against other clotting proteins (eg, Factor VIII or Factor IX) that help bleeding to stop.
What are the clotting factors in plasma?
The clotting factors are Factor I (fibrinogen), Factor II (prothrombin), Factor III (tissue thromboplastin or tissue factor), Factor IV (ionized calcium), Factor V (labile factor or proaccelerin), Factor VII (stable factor or proconvertin), and Factor VIII (antihemophilic factor).
What is Factor 8 called?
Factor VIII (FVIII) is an essential blood-clotting protein, also known as anti-hemophilic factor (AHF). In humans, factor VIII is encoded by the F8 gene. Defects in this gene result in hemophilia A, a recessive X-linked coagulation disorder.
Where is factor VIII made?
Coagulation factor VIII is made chiefly by cells in the liver. This protein circulates in the bloodstream in an inactive form, bound to another molecule called von Willebrand factor, until an injury that damages blood vessels occurs.
What clotting factor is prothrombin?
Prothrombin (factor II) is a vitamin K–dependent coagulation factor. On activation, prothrombin is proteolytically cleaved to form thrombin, and in turn acts as a serine protease that converts fibrinogen to fibrin. In addition, thrombin catalyzes many other coagulation-related reactions.
What Is factor II in coagulation?
Clotting factor II, or prothrombin, is a vitamin K–dependent proenzyme that functions in the blood coagulation cascade. Factor II deficiency is a rare, inherited or acquired bleeding disorder with an estimated incidence of one case per 2 million population.
What is factor 2 prothrombin mutation?
Prothrombin gene mutation (or Factor II mutation or Prothrombin G20210A) is an inherited condition that increases your predisposition to develop abnormal blood clots in the veins (deep vein thrombosis or DVT) and lungs (pulmonary embolism or PE).
What is Factor VII deficiency?
Factor VII deficiency is a rare bleeding disorder that varies in severity among affected individuals. The signs and symptoms of this condition can begin at any age, although the most severe cases are apparent in infancy. However, up to one-third of people with factor VII deficiency never have any bleeding problems.
What activated factor 7?
Recombinant activated factor VII (rFVIIa) binds to exposed tissue factor at the place of tissue and vascular injury. Thrombin generated by this process activates platelets and the coagulation cascade. It has been used in controlling hemorrhage from surgery, trauma, and other causes with success.
Where is factor VII found?
Factor VII is a protein produced in the liver that plays an important role in helping your blood to clot. It’s one of about 20 clotting factors involved in the complex process of blood clotting.
How many amino acids are in thrombin?
Thrombin is a α/β heterodimer composed of a 36 amino acid A chain and 259 amino acid B chain connected by a disufide bridge between Cys1 and Cys122, in addition to 3 other intrachain disulfide bonds. Its overall fold is similar to trypsin and chymotrypsin and it belongs to the peptidase S1 protease family.
What is thrombin substrate?
Thrombin is arguably the most extensively studied of all human proteases. It is a serine protease with essential functions in blood coagulation and in numerous other regulatory processes. Known natural substrates for thrombin include coagulation factors V, VIII, XI and XIII, protein C and fibrinogen [4].
What is plasma without clotting factors?
Blood serum is blood plasma without fibrinogen or the other clotting factors (that is, whole blood minus both the cells and the clotting factors).
How does thrombin activate fibrinogen?
It is activated to thrombin (factor IIa) via enzymatic cleavage of two sites by activated FX (FXa). Activated thrombin leads to cleavage of fibrinogen into fibrin monomers that, upon polymerization, form a fibrin clot. Therefore, activation of prothrombin is crucial in physiological and pathophysiological coagulation.
Is prothrombin a plasma protein?
Prothrombin (factor II) is a vitamin K–dependent plasma protein synthesized in the liver.
How does thrombin convert fibrinogen to fibrin?
Fibrinogen is a soluble macromolecule, but forms an insoluble clot or gel on conversion to fibrin by the action of the serine protease thrombin, which is activated by a cascade of enzymatic reactions triggered by vessel wall injury, activated blood cells, or a foreign surface (Fig. 13.1).
Which of the following is the IV clotting factor?
FactorNameIFibrinogenIIProthrombinIIITissue factor or thromboplastinIVCalcium
Is factor Xa prothrombin activator?
Prothrombin activators from snake venoms are endopeptidases that function similar to Factor Xa, which provides the catalytic site for prothrombin cleavage.
Is Va factor a cofactor?
Factor Va is a cofactor for Factor Xa-mediated prothrombin activation and enhances the rate of thrombin formation more than 1000-fold and activation proceeds via specific cleavage of peptide bonds at Arg709, Arg1018, and Arg1545 [253].



