What factors affect the rate of diffusion quizlet

Lily Fisher
Published Apr 17, 2026
extent of the concentration gradient. greater the difference in concentration, the more rapid the diffusion. … mass of the molecules diffusing. … temperature. … solvent density. … solubility. … surface area and thickness of the plasma membrane. … distance travelled.
What factors affect the rate of diffusion?
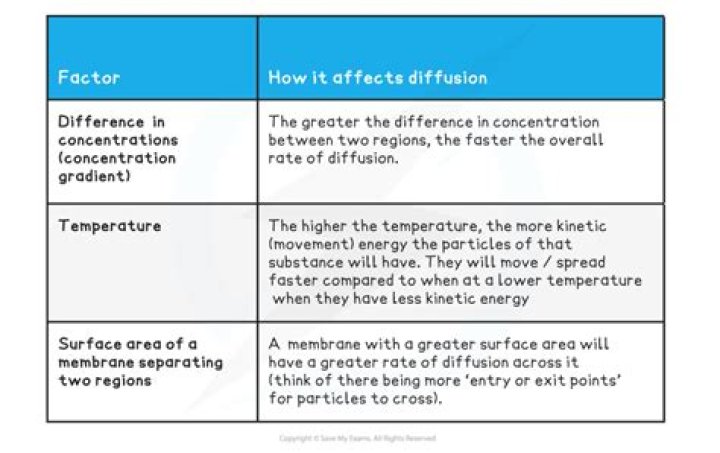
The greater the difference in concentration, the quicker the rate of diffusion. The higher the temperature, the more kinetic energy the particles will have, so they will move and mix more quickly. The greater the surface area, the faster the rate of diffusion.
What factors dont affect the rate of diffusion?
Complete answer: The electrical charges of the diffusion particles are the only factor that does not have an effect on the rate at which diffusion occurs.
Which of the following factors affect the rate of diffusion quizlet?
Terms in this set (7) the greater the difference in concentration, the more rapid th diffusion. The closer the distribution of the material get to equilibrium, the slower the rate of diffusion becomes. Higher temperature increase the energy and therefore the movement of the molecules, increasing the rate of diffusion.What factors increase the rate of diffusion quizlet?
- Size of concentration gradient. Faster at larger gradients.
- Mass of transported molecules. Smaller diffuses faster.
- Membrane surface area. Faster over a larger membrane area.
- Temperature. Increase in temperature and diffusion rate.
What factors affect the rate of osmosis and diffusion?
- The water potential gradient – the higher the water potential gradient, the faster the rate of osmosis. …
- The thickness of the exchange surface – the thinner the exchange surface, the faster the rate of osmosis.
What are the three factors that affect diffusion?
Concentration gradient, size of the particles that are diffusing, and temperature of the system affect the rate of diffusion.
What three factors explored in this lab will affect the rate of diffusion?
A number of factors can affect the rate of diffusion, including temperature, molecular weight, concentration gradient, electrical charge, and distance. Water can also move by the same mechanism.Which factors will affect the rate of diffusion of a gas?
- The partial pressure difference across the diffusion barrier.
- The solubility of the gas.
- The cross-sectional area of the fluid.
- The distance molecules need to diffuse.
- The molecular weight of the gas.
- Concentration gradient.
- Permeability of the membrane.
- Temperature.
- Pressure.
How can distance affect the rate of diffusion?
distance – if the diffusion distance is small, diffusion happens faster because the particles do not have as far to travel. … surface area – the larger the surface area the higher the number of particles that will be able to move in a given time so the faster the rate.
Does polarity affect the rate of diffusion?
Polarity does affect diffusion but only in the case where diffusion is occurring across the cell membrane.
What is the cause of diffusion?
The kinetic energy of the molecules results in random motion, causing diffusion. … it is the random motion of the molecules that causes them to move from an area of high concentration to an area with a lower concentration. Diffusion will continue until the concentration gradient has been eliminated.
What are 3 factors that affect the rate of osmosis?
- Pressure.
- Temperature.
- Surface Area.
- Water Potential.
- Concentration gradient.
What environmental factors affect kinetic energy and diffusion?
What environmental factors affect kinetic energy and diffusion? Temperature, pressure, nature of the material, size of the molecule.
What are the factors that influence the diffusion of materials across the cell membrane?
- Membrane Thickness: For Nutrients to diffuse into a cell they must traverse the cell membrane. …
- Temperature and Pressure: …
- Concentration Gradient: …
- Surface Area:
How does the concentration gradient affect diffusion?
Molecules can move into or out of cells by the process of diffusion . … The bigger the difference, the steeper the concentration gradient and the faster the molecules of a substance will diffuse. The direction of diffusion is said to be ‘down’ or ‘with’ the concentration gradient.
How does time affect diffusion?
If the medium that a given particle has to diffuse through is very dense or viscous, then the particle will have a harder time diffusing through it. So the rate of diffusion will be lower. If the medium is less dense or less viscous, then the particles will be able to move more quickly and will diffuse faster.
What are the factors affecting diffusion Class 9?
The rate of diffusion is affected by the concentration gradient, membrane permeability, temperature, and pressure.
On which factors rate of diffusion depends Class 9?
Particle size, temperature, concentration difference and diffusion distance affect the rate of diffusion.
How does solution density affect the rate of diffusion?
Solvent density: As the density of a solvent increases, the rate of diffusion decreases. The molecules slow down because they have a more difficult time getting through the denser medium. If the medium is less dense, diffusion increases.
How does the shape of molecules affect the rate of diffusion?
Diffusion is like going through a net or sieve. Smaller sizes and shapes pass through more easily than large ones.
Why do nonpolar molecules diffuse faster?
In general, polar molecules diffuse more rapidly through the lipid bilayer part of cell membranes than do nonpolar molecules. … Nonpolar molecules diffuse through more rapidly because much of the bilayer is nonpolar.
What causes diffusion to occur quizlet?
What causes diffusion? Molecules constantly bumping into one another, which causes them to push away from each other and move to an area of lower concentration.
What is the fastest to slowest rate of diffusion?
Neon is the fastest. Chlorine is the slowest.
What does diffusion depend on?
The diffusion rate depends on several factors: the concentration gradient (the increase or decrease in concentration from one point to another); the amount of surface area available for diffusion; and the distance the gas particles must travel.
What 4 things affect the rate of osmosis?
Several factors can affect the rate of osmosis. For example, temperature, particle size, and the size of the concentration gradient can all affect the rate of osmosis.
What factors affect the rate of osmotic movement of water?
The rate of osmosis varies with a number of factors, including temperature, pressure, and the difference in solute concentrations between two solutions separated by a selectively permeable membrane.
How does concentration affect rate of osmosis?
The concentration of a solute affects the rate of osmosis over time, in a way where, the higher the concentration of a solute, the faster the rate of osmosis. This happens because, in a semi-permeable membrane the water is the only through that can move through. … That results in the rate of osmosis being faster.