What happens when acetic acid reacts with calcium hydroxide

Rachel Hunter
Published Mar 02, 2026
> The reaction of acetic acid and calcium hydroxide produces calcium acetate, on dry distillation acetone and calcium carbonate. … So this reaction is a very important reaction industrially.
What is the chemical equation when acetic acid reacts with calcium hydroxide?
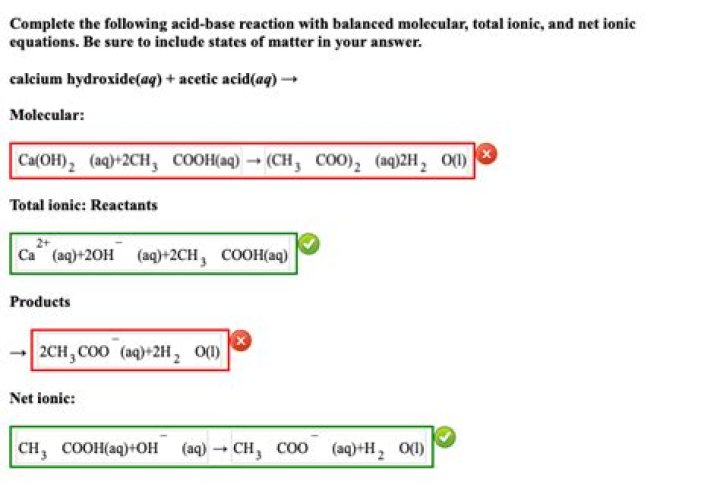
Ethanoic acid is the systematic name for acetic acid (CH3COOH) . The formula for calcium hydroxide is Ca(OH)2 . Balanced word equation: Two moles of ethanoic acid (acetic acid) react with one mole of calcium hydroxide to produce one mole of calcium acetate and two moles of water.
What happens when acetic acid is heated with CaO?
2CH3COOH+CaO—>(CH3COO)²Ca+H2O. Calcium acetate is obtained by the action of calcium oxide on acetic acid.
What happens when calcium hydroxide reacts with hydrochloric acid?
When calcium hydroxide reacts with hydrochloric acid, calcium chloride and water are produced….How does acetic acid react with hydroxide?
In the reaction between acetic acid and sodium hydroxide, the acetic acid donates a proton to the hydroxide ion and acts as an acid. The hydroxide ion accepts a proton and acts as a base. The stoichiometric relationship between acetic acid and sodium hydroxide is 1:1 (from Equation 2).
What is the chemical formula of acetic acid?
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula CH3COOH (also written as CH3CO2H, C2H4O2, or HC2H3O2).
What happens when acetic acid is treated with Ca Oh 2 and heat?
And when the calcium acetate is heated, acetone and calcium carbonate is obtained. This process is a way to produce acetone.
Which of the following is the formula of calcium hydroxide?
Calcium hydroxide (traditionally called slaked lime) is an inorganic compound with the chemical formula Ca(OH)2.What does calcium hydroxide react with to make calcium chloride?
Calcium hydroxide reacts with hydrochloric acid in a neutralization reaction to create water and calcium chloride.
What happens when carboxylic acid reacts with NaOH and CaO?The solid sodium salt of a carboxylic acid is mixed with solid soda lime, and the mixture is heated. The given reaction involves removal of carbon dioxide molecules. … The first reaction occurring between an acid and the soda lime forms sodium benzoate, and then a decarboxylation occurs.
Article first time published onWhat is the reaction of acetic acid with lime water?
Acetic acid reacts with sodium bicarbonate to produce carbon dioxide. On passing CO2 gas through lime water, the lime water turns milky. The milky appearance of lime water is due to the formation of solid calcium carbonate (CaCO3).
What happens when NaOH reacts with acetic acid?
Sodium hydroxide solution is treated with acetic acid to form sodium acetate and water.
Is CH3COOH and NaOH a buffer?
Not a buffer; the NaOH completely neutralizes the acetic acid to give sodium acetate. Buffer; the NaOH neutralizes only half of the acetic acid to give a solution containing equal amounts of acetic acid and sodium acetate.
When NaOH is added to the solution of CH3COOH then?
When a strong base (NaOH) is added to a weak acid (acetic acid,CH3COOH) then dissociation of acetic acid increases.
What does nitric acid and calcium hydroxide make?
When nitric acid and calcium hydroxide are combined, calcium nitrate and water are formed: Molecular Equation: 2HNO3 + Ca (OH)2 –>Ca (NO3)2 + 2H2O (l) HNO3 is a strong acid.
What is slaked lime in chemistry?
Calcium hydroxide, also called slaked lime, Ca(OH)2, is obtained by the action of water on calcium oxide. When mixed with water, a small proportion of it dissolves, forming a solution known as limewater, the rest remaining as a suspension called milk of lime.
What is formed when hydrochloric acid reacts with sodium hydroxide?
Hydrochloric acid + Sodium hydroxide → Sodium chloride + Water.
What reacts with acetic acid?
Reactions with inorganic compounds Acetic acid is mildly corrosive to metals including iron, magnesium, and zinc, forming hydrogen gas and salts called acetates: Mg + 2 CH3COOH → (CH3COO)2Mg + H.
What is C2H4O2 called?
Acetic acid | C2H4O2 | ChemSpider.
What happens when you add NaOH to calcium hydroxide?
When we add aqueous NaOH solution to aqueous CaCl2 solution, a white precipitate is formed. This precipitate is calcium hydroxide (Ca(OH)2). However, if ion concentrations of calcium and hydroxyl ions are very low, calcium hydroxide can be dissolved in water and give colorless solution.
Does CaCl2 and NaOH form a precipitate?
If you have calcium chloride and sodium hydroxide, you can mix saturated solutions of both and a white precipitate should form. The precipitate is calcium hydroxide.
What is the difference between calcium chloride and calcium hydroxide?
05 summary is that the calcium chloride raises calcium, but not much else to too great of a degree. The calcium hydroxide raises calcium, alkalinity, and Ph. If you dose too much at once it can cause dangerous Ph problems.
What is Chuna made of?
Limestone, commonly known as chuna in Hindi, is a type of sedimentary rock. The use of limestone as a building material is profound. It is also a popular ingredient in the chemical industry for the production of lime.
How do you write caoh2?
Calcium hydroxide (traditionally called slaked lime) is an inorganic compound with the chemical formula Ca(OH)2.
What is Raja Chuna for?
Raja Chuna is used in the making supari and paan and lime chutney.
Which of the following gives phenol with CaO and NaOH?
Only salicylic acid (2-Hydroxybenzoic acid) on decarboxylation gives phenol. Complete answer: We must know that salicylic acid will give phenol with \[{\text{CaO}}\] and \[{\text{NaOH}}\].
What does NaOH and CaO do?
A 3:1 mixture of NaOH and CaO is called sodalime. CaO makes NaOH less reactive. NaOH is quite hygroscopic and forms a sodium hydroxide solution when it is exposed to air. Sodalime is not that hygroscopic and hence, stabilises NaOH by avoiding moisture absorption.
When phenol is treated with chcl3 and NaOH the product formed is?
When phenol is treated with CHCl3 and NaOH the product formed is salicylaldehyde, this reaction is known as Reimer-Tiemann reaction.
Does calcium hydroxide react with vinegar?
> The reaction of acetic acid and calcium hydroxide produces calcium acetate, on dry distillation acetone and calcium carbonate.
Is calcium acetate soluble in acetic acid?
NamesSolubility in water37.4 g/100 mL (0 °C) 34.7 g/100 mL (20 °C) 29.7 g/100 mL (100 °C)
Is calcium hydroxide soluble in acetic acid?
-Calcium carbonate and calcium hydroxide are soluble in acetic acid as its hydration energy is greater than lattice energy. … -Since acetic acid is an acid, it undergoes a chemical reaction with oxides, carbonates, and hydroxides and no reaction with oxalates.