What is a block on the periodic table

Andrew Campbell
Published Feb 26, 2026
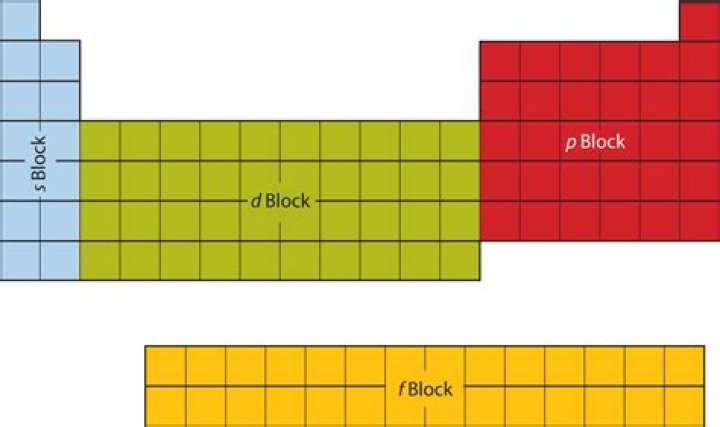
A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. The term appears to have been first used by Charles Janet. Each block is named after its characteristic orbital: s-block, p-block, d-block, and f-block.
What is the block on a periodic table?
A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. The term appears to have been first used by Charles Janet. Each block is named after its characteristic orbital: s-block, p-block, d-block, and f-block.
What's-block elements are called?
The s-block in the periodic table of elements occupies the alkali metals and alkaline earth metals, also known as groups 1 and 2. … Alkali metals are the chemical elements found in Group 1 of the periodic table. The alkali metals include: lithium, sodium, potassium, rubidium, cesium, and francium.
What are the four blocks of the periodic table?
This gives the periodic table block in the standard periodic table in which the element is to be found. The four blocks are s, p, d, and f. Elements in each block are colour coded in the background graphics, the periodic tables, and the element pages themselves.How many blocks in periodic table what are they?
On the basis of electronic configuration, the elements have been categorized into four blocks in the periodic table. Complete step by step answer: On the basis of electronic configurations, the elements in the long form periodic table have been divided into four blocks. These are s, p, d, f-blocks.
How do you read block elements?
Element Boxes The atomic number, the number at the top of each element box, is the number of protons in the nucleus and the number of electrons in the atom. The elemental symbol is an abbreviation of the element name. Some periodic tables do not include the element names, only the symbols.
How do you find the block of an element?
If the element is in the p block, then the number of the group can be determined by the formula: (number of valence electrons + 10). 3. If the element is in the d block, then the number of the group can be determined by the formula: numberofelectronsin(n−1)dsubshell + (number of electrons in (n) s subshell).
Why are they called S block?
The s-block and p-block elements are so called because their valence electrons are in an s orbital or p orbital respectively. They are also called Typical Elements to distinguish them from the transition and inner transition series.Which of the blocks on the periodic table contains the most elements?
The f-block is the largest block on the periodic table. They are the inner transition elements.
How many periods are in the periodic table?The periodic table of the elements contains all of the chemical elements that have been discovered or made; they are arranged, in the order of their atomic numbers, in seven horizontal periods, with the lanthanoids (lanthanum, 57, to lutetium, 71) and the actinoids (actinium, 89, to lawrencium, 103) indicated …
Article first time published onWhy alkali metals belong to S Block in the periodic table?
The alkali metals are so called S block because they form alkalies when they react with water. Alkalies, including sodium hydroxide and potassium hydroxide, are hydroxide compounds of these elements. … As only two electrons can satisfy the s-orbital, two classes (1 & 2) belong to the Periodic Table’s s-block.
Where is the f-block on the periodic table?
The f-block elements,found in the two rows at the bottom of the periodic table, are called inner transition metals and have valence electrons in the f-orbital’s.
How many blocks are in the Mendeleev periodic table?
The periodic table is divided into four blocks (s-block, p-block, d-block and f-block) and the elements in the periodic table are classified on the basis of their electronic configuration.
What are the four blocks of the periodic table based on electron configuration?
An element’s placement in the periodic table is determined by its electron configuration. The chemical properties of various elements are intimately related to their valence electron configurations. The periodic table is divided into four blocks (s, p, d, f) based on which sublevel is in the process of being filled.
What do you mean by blocks in a periodic table and why elements were placed in blocks?
A block of the periodic table of elements is a set of adjacent groups. The respective highest-energy electrons in each element in a block belong to the same atomic orbital type. … Blocks are often called families.
What block is chlorine?
ChlorineAtomic number (Z)17Groupgroup 17 (halogens)Periodperiod 3Blockp-block
How is group period and block of elements predicted?
The period is represented by the principal quantum number of the valence shell. Block is predicted on the basis of sub shell which receives the last electron. Group is predicted on the basis of the number of electrons in the penultimate or the outer most shell.
How do you read a periodic table?
To read the periodic table, start at the top left with the elements with the lowest atomic numbers, which tells you how many protons each atom has. Then, as you move right across the chart, make note that the atomic weight, shown at the bottom of the square, also increases.
What do all the numbers mean on the periodic table?
What do the symbols and numbers mean? … The number above the symbol is the atomic mass (or atomic weight). This is the total number of protons and neutrons in an atom. The number below the symbol is the atomic number and this reflects the number of protons in the nucleus of each element’s atom.
What do periods tell us on the periodic table?
Periods: The horizontal rows in the periodic table that signify the number of electron shells in an element.
How are the blocks indicated?
The block names (s, p, d, f and g) are derived from the spectroscopic notation for the associated atomic orbitals: sharp, principal, diffuse, and fundamental.
Which block contains all the metalloids?
Complete answer: Metalloids are present in the p block of periodic table because their properties lie in between metals and nonmetals.
Why is P block called P block?
Why are they called P block elements? The elements s-block and p-block are so-called because their valence electrons are either in an orbital s or p. These are often called Standard Components, in order to differentiate them from the sequence of transformation and internal transformation.
What are P block and S-block elements?
Representative elements of the periodic table include s are p block elements. s-block elements included the elements of group 1 and group 2 of the periodic table while p – block elements include the elements of group 13, 14, 15, 16, 17 and 18.
Why is the d-block called the d-block?
They’re called “D-block elements” because they have their valence electrons in one or more d-orbitals (but not in any p-orbitals).
Who is the father of periodic table?
Dmitri Mendeleev, Russian in full Dmitry Ivanovich Mendeleyev, (born January 27 (February 8, New Style), 1834, Tobolsk, Siberia, Russian Empire—died January 20 (February 2), 1907, St. Petersburg, Russia), Russian chemist who developed the periodic classification of the elements.
Which is the longest period in the periodic table?
Hence, the sixth period is known as the longest period in the periodic table.
Are there 7 or 9 periods on the periodic table?
From here, there are seven periods on the periodic table, and nine if you count the lanthanides and actinides. Hydrogen starts the first period of the periodic table, while oganesson (element 118 ) ends the seventh period.
What are s-block elements with examples?
The s-block elements include hydrogen (H), helium (He), lithium (Li), beryllium (Be), sodium (Na), magnesium (Mg), potassium (K), calcium (Ca), rubidium (Rb), strontium (Sr), cesium (Cs), barium (Ba), francium (Fr) and radium (Ra).
In which block on the periodic table do you find the alkali metals?
The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr). Together with hydrogen they constitute group 1, which lies in the s-block of the periodic table.
What block is iron Fe?
Iron’s element symbol is Fe, and has an atomic number of twenty six. As a transition metal, iron is located in the d-block, specifically in group 8 and period 4. Iron’s electron configuration is [Ar] 3d6 4s2, and thus it has 2 valence electrons.