What is an analyte in chromatography

Lily Fisher
Published Mar 26, 2026
Chromatography terms. Analyte – the substance to be separated during chromatography. It is also normally what is needed from the mixture. Analytical chromatography – the use of chromatography to determine the existence and possibly also the concentration of analyte(s) in a sample.
What is analyte in chromatography Mcq?
Analyte :- substance for separation. … Eluent :- It is a solvent that used for separation of absorbed material from stationary phase. Eluate :- is a liquid solution that is a result from Elution. Chromatogram :- It is a graphical represention of Chromatography.
How are analytes separated in GC?
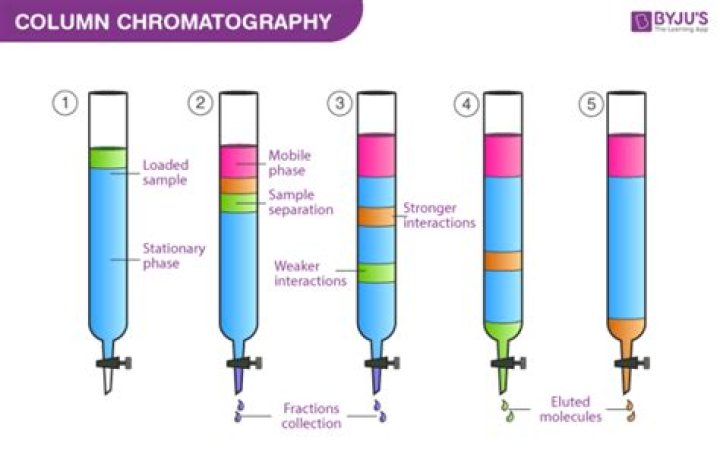
In gas chromatography, the components of a sample are dissolved in a solvent and vaporized in order to separate the analytes by distributing the sample between two phases: a stationary phase and a mobile phase.
What are the components of chromatography?
- Stationary phase: This phase is always composed of a “solid” phase or “a layer of a liquid adsorbed on the surface solid support”.
- Mobile phase: This phase is always composed of “liquid” or a “gaseous component.”
- Separated molecules.
What is an eluent in chromatography?
ELUENT. The eluent is the mobile phase or the solvent that is passed through the column. Molecules in the sample will desorb off the adsorbent and dissolve in the eluent when the polarity of the eluent matches the polarity of the molecules.
Why is ion exchange chromatography used?
Ion exchange chromatography is commonly used to separate charged biological molecules such as proteins, peptides, amino acids, or nucleotides. The amino acids that make up proteins are zwitterionic compounds that contain both positively and negatively charged chemical groups.
What is analyte derivatization?
(a) Derivatization that replaces active (polar) hydrogen atoms in the analyte to decrease its boiling point. The active hydrogens in a chemical compound typically enhance the capability to form hydrogen bonds and increase the compound polarity.
Why do components separate in chromatography?
The different components of the mixture travel through the stationary phase at different speeds, causing them to separate from one another. The nature of the specific mobile and stationary phases determines which substances travel more quickly or slowly, and is how they are separated.What are the 4 types of chromatography?
There are four main types of chromatography. These are Liquid Chromatography, Gas Chromatography, Thin-Layer Chromatography and Paper Chromatography. Liquid Chromatography is used in the world to test water samples to look for pollution in lakes and rivers.
What is solvent front in chromatography?In chromatography, the solvent front is the position on the TLC plate indicating the furthest distance traveled by the developing solvent (or eluent)
Article first time published onWhat is column in gas chromatography?
The column is the heart of the gas chromatograph. It is through interactions between solutes (individual compounds in the sample, also called analytes) and the stationary phase within the column that separation can occur.
Why does the width of the peak WB increase as TR increases?
The trade-off is that the retention time increases proportionally to the column length and a significant peak broadening will be observed as well because of increased longitudinal diffusion inside the column.
What does it mean when a GC peak appears asymmetrical?
A peak is considered asymmetric when the distance from the start of the peak to the centre (A) and from centre to the end (B) of the peak differs (Fig 1). It is best to measure these distances at about 10% of the peak height. Within asymmetric peaks, there are two possibilities that could exist; Fronting and Tailing.
What is elution process?
In analytical and organic chemistry, elution is the process of extracting one material from another by washing with a solvent; as in washing of loaded ion-exchange resins to remove captured ions. … Predicting and controlling the order of elution is a key aspect of column chromatographic methods.
What is elution volume in chromatography?
Elution volume is the amount of elution or the volume of elution required to cause the elution process, which is the removal of materials that are absorbed with a solvent. … Predicting and controlling the order of elution is a key aspect of column chromatography methods.
What is eluent and effluent?
Eluate is the. mixture of solute and solvent exiting. the column. Effluent is the stream. flowing out of a chromatographic col-
What is derivatization method?
Derivatization is a technique used in chemistry which converts a chemical compound into a product (the reaction’s derivate) of similar chemical structure, called a derivative. … Resulting new chemical properties can be used for quantification or separation of the educt.
What is precolumn derivatization?
Pre-column derivatization and Post-column derivatization are well-known as general methods for amino acid HPLC analysis. The pre-column method derivatizes the amino acids prior to separation on the C18 column. … In addition, amino acids which are derivatized using OPA can be detected by UV detector.
What is derivatization reagent?
Derivatization is a technique that usually involves a reaction of the analyte(s) of interest with a particular derivatization reagent. … One of the more commonly used in GC analyses involves a reaction that adds a trimethylsilyl (TMS) functional group to the compound. This is also known as trimethylsilylation.
What are the two types of ion exchange chromatography?
The two types of ion chromatography are anion-exchange and cation-exchange. Cation-exchange chromatography is used when the molecule of interest is positively charged.
What does Ion Chromatography measure?
What is Ion Chromatography? … Ion chromatographs are able to measure concentrations of major anions, such as fluoride, chloride, nitrate, nitrite, and sulfate, as well as major cations such as lithium, sodium, ammonium, potassium, calcium, and magnesium in the parts-per-billion (ppb) range.
What are the 12 types of chromatography?
The twelve types are: (1) Column Chromatography (2) Paper Chromatography (3) Thin Layer Chromatography (4) Gas Chromatography (5) High Performance Liquid Chromatography (6) Fast Protein Liquid Chromatography (7) Supercritical Fluid Chromatography (8) Affinity Chromatography (9) Reversed Phase Chromatography (10) Two …
What are the two main types of chromatography?
There are two main types of chromatography: liquid chromatography (LC) and gas chromatography (GC).
What are the seven types of chromatography?
- Adsorption Chromatography. In the process of adsorption chromatography, different compounds are adsorbed on the adsorbent to different degrees based on the absorptivity of the component. …
- Thin Layer Chromatography. …
- Column Chromatography. …
- Partition chromatography.
Why do colors separate in chromatography?
The reason why the colors separate has to do with the chemicals that make up the color, the water, and the paper. The chemicals that make up the color are called pigments. Some pigments attach to water better than others so they move further through the paper before sticking.
What factors affect separation in chromatography?
- The dimension of the column: The ideal length: diameter ratio is 30: 1 or 20: 1, but to improve in efficiency 100: 1 can be additionally suitable.
- The particle size of the adsorbent: …
- Nature of the solvent: …
- The temperature of the column: …
- Pressure:
Which solvent is best for chromatography?
SolventPolarity (arbitrary scale of 1-5)SuitabilityWater1 – Most polarGoodRubbing alcohol (ethyl type) or denatured alcohol2 – High polarityGoodRubbing alcohol (isopropyl type)3 – Medium polarityGoodVinegar3 – Medium polarityGood
Why do we mark TLC plates with pencil and not with pen?
Why do you use a pencil and not a pen to mark TLC plates? Answer: The pen ink becomes mobile on the plate and travels up the TLC plate with TLC solvent. But the solid particles of graphite in the pencil won’t get dissolved and hence can be used to mark TLC plates.
What is mobile phase in chromatography?
The mobile phase flows through the packed bed or column. … moving fluid stream, called the mobile phase, and a contiguous stationary phase. The mobile phase may be either a liquid or a gas, while the stationary phase is either a solid or a liquid.
What is the difference between packed and capillary columns?
The main difference between packed column and capillary column is that, in a packed column, the stationary phase is packed into the cavity of the column whereas, in a capillary column, the stationary phase coats the inner surface of the cavity of the column.
How many columns are used in gas chromatography?
Two types of columns are used in gas chromatography: packed columns and capillary columns.



