What is an ion formed from a single atom

Dylan Hughes
Published Apr 03, 2026
A monatomic ion is an ion formed from a single atom. In other words, it is a single atom that has a different number of protons and electrons. … If there is an excess of electrons, the charge is negative. Metals typically form cations, while nonmetals usually form anions.
What is an ion in an atom?
ion, any atom or group of atoms that bears one or more positive or negative electrical charges. Positively charged ions are called cations; negatively charged ions, anions.
How is an ion different from the atom it is formed from?
Explanation: An atom is neutral when it has equal numbers of protons and electrons. An ion is an atom that is no longer neutral because it has either lost or gained one or more electrons. When an atom loses one or more electrons, it becomes a positively charged ion, called a cation .
How are ions formed into atoms?
Ions are formed when atoms lose or gain electrons in order to fulfill the octet rule and have full outer valence electron shells. When they lose electrons, they become positively charged and are named cations. When they gain electrons, they are negatively charged and are named anions.How are ions formed quizlet?
Ions are formed when atoms lose or gain electrons in order to fulfill the octet rule and have full outer valence electron shells.
How is an ion different from an atom quizlet?
What is the difference between an atom and an ion? An atom has no electrical charge. Ions have an electrical charge because they have an imbalance of protons and electrons.
How do you know what ions are formed?
- the number of charges on an ion formed by a metal is equal to the group number of the metal.
- the number of charges on an ion formed by a non-metal is equal to the group number minus eight.
- hydrogen forms H + ions.
How do ions form ionic bonds?
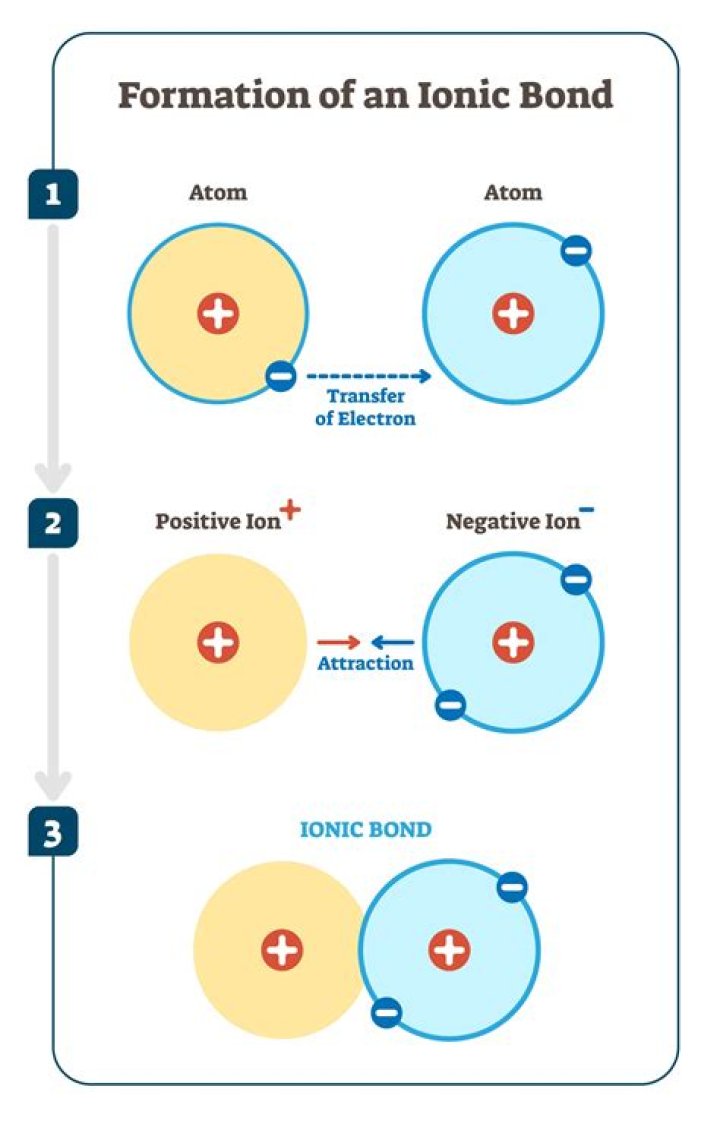
An ionic bond is formed by the complete transfer of some electrons from one atom to another. The atom losing one or more electrons becomes a cation—a positively charged ion. The atom gaining one or more electron becomes an anion—a negatively charged ion.Is an ion a type of atom?
An ion (/ˈaɪɒn, -ən/) is an atom or molecule with a net electrical charge. The charge of an electron is considered negative by convention and this charge is equal and opposite to charge of a proton, which is considered positive by convention.
What changes when an ion is formed from an atom?When an ion is formed, the number of protons does not change. Neutral atoms can be turned into positively charged ions by removing one or more electrons. A neutral sodium atom, for example, contains 11 protons and 11 electrons.
Article first time published onWhat changes when an ion is formed from an atom quizlet?
An atom becomes an ion when its number of protons change. Some ions are positively charged, and some ions have no charge. The formation of an ion results in a full outermost energy level. Ions usually form when electrons are transferred from one atom to another.
How do ions form ionic bonds quizlet?
An ionic bond is the force of attraction that holds together oppositely charged ions. It forms when atoms of a metal transfer electrons to atoms of a nonmetal. When this happens, the atoms become oppositely charged ions. … As a result, ionic compounds are brittle solids with high melting and boiling points.
What are examples of an ion?
Ions with a positive charge are called cations. Ions with a negative charge are called anions. Many normal substances exist in the body as ions. Common examples include sodium, potassium, calcium, chloride, and bicarbonate.
What is ionic charge?
ionic charge The electrical charge of an ion, created by the gain (negative charge) or loss (positive charge) of one or more electrons from an atom or group of atoms.
What ion is formed by a group 6 element?
GroupElementIon charge1Na+2Mg2+6O2-7Cl-
What is ion quizlet?
An ion is a charged atom or molecule . It is charged because the number of electrons does not equal the number of protons in the atom or molecule.
How are atoms and ions different from each other?
An atom has an equal number of protons and electrons. An ion has an imbalance between the number of protons and electrons. In chemistry, the main difference between an atom and an ion is that an atom is a neutral particle, while an ion has a positive or negative electrical charge.
How are ions of the same element different?
An ion is an atom of the same element that has different numbers of electrons. An isotope is an atom of the same element that has different numbers of neutrons. … An anion is a negatively charged ion that has gained more electrons than protons and added them to it’s energy levels.
How do you know if an atom is an ion?
If an atom has the same number of protons and electrons, it is electronically neutral. However, if the total number of electrons does not equal the number of protons, the atom has a net electrical charge. Any atom or molecule with a net charge, either positive or negative, is known as an ion.
How are ions and isotopes formed?
Neutral atoms of an element contain an equal number of protons and electrons. … The number of neutrons can vary to produce isotopes, which are atoms of the same element that have different numbers of neutrons. The number of electrons can also be different in atoms of the same element, thus producing ions (charged atoms).
Which best describe an ion?
An ion is a charged atom or molecule. … When an atom is attracted to another atom because it has an unequal number of electrons and protons, the atom is called an ION. If the atom has more electrons than protons, it is a negative ion, or ANION.
What do ions form after ionic?
Forming an Ionic Bond Once the oppositely charged ions form, they are attracted by their positive and negative charges and form an ionic compound. Ionic bonds are also formed when there is a large electronegativity difference between two atoms.
Why do ions form after ionic?
Ions form when atoms gain or lose electrons. Since electrons are negatively charged, an atom that loses one or more electrons will become positively charged; an atom that gains one or more electrons becomes negatively charged. Ionic bonding is the attraction between positively- and negatively-charged ions.
How and why are positive ions formed?
Positive ions are formed by atoms or molecules suffering an inelastic collision with an energetic electron in which an electron is lost from the atom or molecule (electron impact ionization). … In electron attachment ionization, negative ions are formed by electron attachment in the gas.
What changes an ion?
The number of electrons changes when an atom forms an ion.
Do ions usually form when electrons are transferred?
Some ions are positively charged, and some ions have no charge. … The formation of an ion results in a full outermost energy level. This statement is true. Ions usually form when electrons are transferred from one atom to another.
How are most ions formed ck12?
Most ions form when atoms transfer electrons to or from other atoms or molecules. For example, when a sodium atom loses an electron, it becomes a positive sodium ion.
What is true about the formation of ions?
Ions. Ions are charged substances that have formed through the gain or loss of electrons. Cations form from the loss of electrons and have a positive charge while anions form through the gain of electrons and have a negative charge.
What molecules form ionic bonds?
Ionic bonds are formed between a cation, which is usually a metal, and an anion, which is usually a nonmetal. A covalent bond involves a pair of electrons being shared between atoms.
How does an atom become a positive ion?
The atom that has lost an electron becomes a positively charged ion (called a cation), while the atom that picks up the extra electron becomes a negatively charged ion (called an anion). Opposite charges attract one another while similar charges repel one another.
How do ionic compounds form crystals?
The ions have a regular, repeating arrangement called an ionic lattice . The lattice is formed because the ions attract each other and form a regular pattern with oppositely charged ions next to each other. … This is why solid ionic compounds form crystals with regular shapes.



