What is Cooperativity biochemistry

Dylan Hughes
Published Mar 30, 2026
cooperativity, in enzymology, a phenomenon in which the shape of one subunit of an enzyme consisting of several subunits is altered by the substrate (the substance upon which an enzyme acts to form a product) or some other molecule so as to change the shape of a neighbouring subunit.
What do you mean by cooperativity?
Definition of cooperativity : the quality or state of being cooperative To safeguard the scientific enterprise, cooperativity and humility need to instead become central virtues of science.—
What is cooperative binding in biology?
Abstract. Molecular binding is an interaction between molecules that results in a stable association between those molecules. Cooperative binding occurs if the number of binding sites of a macromolecule that are occupied by a specific type of ligand is a nonlinear function of this ligand’s concentration.
What is an example of cooperativity in biology?
An example of positive cooperativity is the binding of oxygen to hemoglobin. One oxygen molecule can bind to the ferrous iron of a heme molecule in each of the four chains of a hemoglobin molecule. … Heterotropic cooperativity is where a third party substance causes the change in affinity.What is cooperative effect in chemistry?
The cooperative effect describes the ability of the four identical haemoglobin subunits to change their conformation. The cause of this change is the acceptance or release of an O2 molecule by one of the subunits, which increases the ability of the other haemoglobin domains to accept or release oxygen.
What is cooperativity phenomenon in Haemoglobin?
Hemoglobin displays something called positive cooperativity. This means that when deoxyhemoglobin binds a single oxygen, it causes the other heme groups to become much more likely to bind other oxygen molecules.
What is cooperative binding of Haemoglobin?
The way by which hemoglobin binds oxygen is referred to as cooperative binding. The binding of oxygen to hemoglobin makes it easier for more oxygen to bind. … Oxygen binds to hemoglobin in the lungs, which increases the affinity for even more oxygen to bind.
What are the two types of cooperativity?
- Types of Cooperative Binding. Cooperative binding can produce negative cooperativity, positive cooperativity, heterotropic cooperativity, and homotropic cooperativity. …
- Subunit Cooperativity. …
- Entropy and Cooperativity. …
- Kinetics of cooperativity. …
- Hill Equation.
Which of the following is an example of cooperativity?
Which of the following is an example of cooperativity? a substrate molecule binding at one unit of a tetramer allowing faster substrate binding at each of the other three subunits.
Which of the following describes enzyme cooperativity?Which of the following statements describes enzyme cooperativity? A substrate molecule bound to an active site of one subunit promotes substrate binding to the active site of other subunits. … The reaction is faster than the same reaction in the absence of the enzyme.
Article first time published onWhat is cooperative binding in proteins?
A feature of some proteins comprising more than one subunit is that binding of a ligand to its binding site on one subunit, can increase the affinity of a neighbouring subunit for the same ligand, and hence enhance binding. This type of allosteric regulation is called cooperative binding. …
What does non cooperative binding mean?
When the binding of a ligand to the first ligand site directly impacts the affinity of the second ligand site for that ligand, this is considered cooperative binding. If the binding of a ligand doesn’t affect the affinity of the second ligand site for that ligand, this is non-cooperative binding.
What is the physiological significance of the cooperative binding of oxygen by hemoglobin?
What is the physiological significance of the cooperative binding of oxygen by hemoglobin? The cooperativity allows hemoglobin to become saturated in the lungs, where oxygen pressure is high.
What is cooperativity in supramolecular chemistry?
Cooperativity describes how the binding of one ligand can influence a receptor’s affinity toward further binding interactions and is most easily assessed for the stepwise binding of monovalent ligands.
What are the types of cooperatives?
- Producer / Marketing Cooperatives.
- Consumer Cooperatives.
- Worker Cooperatives.
- Housing Cooperatives.
- Financial Cooperatives.
- New Generation Cooperatives.
- Multi-Stakeholder Cooperatives.
- Non-profit Community Service Cooperatives.
What is a binding polynomial?
In biology, a ligand is a substance that binds to a target molecule to serve a given purpose. In this context, the binding polynomial, arises as the denominator of the rational function describing the average number of occupied binding sites as a function of ligand activity. …
What causes cooperativity?
cooperativity, in enzymology, a phenomenon in which the shape of one subunit of an enzyme consisting of several subunits is altered by the substrate (the substance upon which an enzyme acts to form a product) or some other molecule so as to change the shape of a neighbouring subunit.
What is the physiological significance of cooperative binding?
What is the physiological significance of the cooperative binding of oxygen by hemoglobin? The cooperativity allows hemoglobin to become saturated in the lungs where oxygen pressure is high.
Who observed the cooperative binding of oxygen by hemoglobin?
After 1924, Adair and Svedberg independently showed that the molecule contained four hemes, and Adair’s intermediate compound hypothesis, with four binding constants suitably chosen, could formally explain cooperative binding.
Who proposed the Cooperativity?
Created by Ross Firestone.
How many o2 molecules are attached to each hemoglobin?
The hemoglobin molecule has four binding sites for oxygen molecules: the iron atoms in the four heme groups. Thus, each Hb tetramer can bind four oxygen molecules.
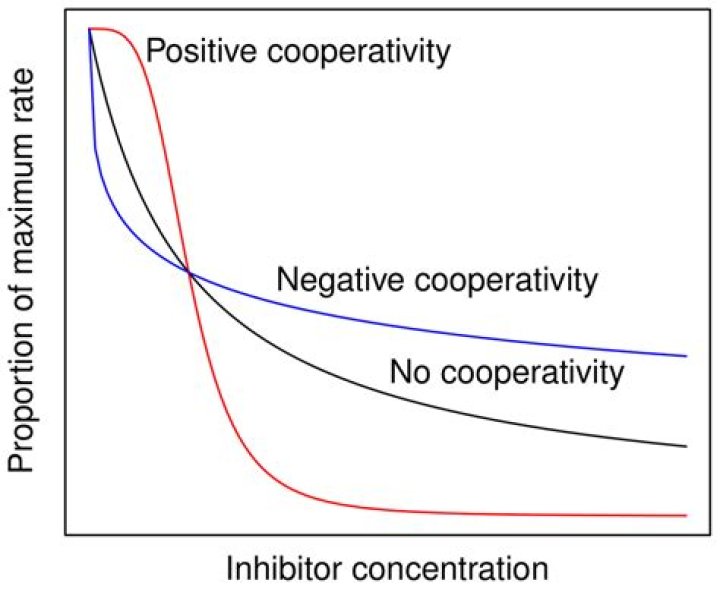
How do you measure Cooperativity?
Cooperativity can be recognized by plotting velocity against substrate concentration. An enzyme that displays positive cooperativity sill be sigmoidal (or S-shaped), while noncooperative enzymes display Michaelis-Menten kinetics and the plots are hyperbolic.
Is cooperativity a type of allosteric regulation?
Positive cooperativity implies allosteric binding – binding of the ligand at one site increases the enzyme’s affinity for another ligand at a site different from the other site. Enzymes that demonstrate cooperativity are defined as allosteric.
What is the difference between cooperativity and Allostery?
The term cooperativity is used to describe folding of macromolecules and the formation of molecular structures and macromolecular ensembles while allostery is often referenced to illustrate ligand-induced conformational transitions that impact the function of a biological molecule.
How does Cooperativity influence metabolic control in the cell?
A significant proportion of enzymes display cooperativity in binding ligand molecules, and such effects have an important impact on metabolic regulation. … Sharp responses to changes in metabolite concentrations can allow organisms to better respond to environmental changes and maintain metabolic homeostasis.
Are all allosteric enzymes cooperative?
Allosteric sites allow effectors to bind to the protein, often resulting in a conformational change involving protein dynamics. … Whereas enzymes without coupled domains/subunits display normal Michaelis-Menten kinetics, most allosteric enzymes have multiple coupled domains/subunits and show cooperative binding.
Which of the following best describes the forward reaction?
The term that best describes the forward reaction in the figure is B) exergonic, ∆G < 0.
What is cooperativity in bioinorganic chemistry?
Cooperativity is the interaction process by which binding of a ligand to one site on a macromolecule (enzyme, receptor, etc.) influences binding at a second site, e.g. between the substrate binding sites of an allosteric enzyme.
Does higher cooperativity mean higher affinity?
Two binding sites, one with low affinity and one with high affinity, produce a cooperative response with the overall affinity being the average of the two; a third high-affinity site pushes the average affinity higher while increasing cooperativity.
Which of the following proteins exhibit positive cooperativity in oxygen binding?
The correct answer choice is “Myoglobin has a higher affinity for oxygen compared to hemoglobin.”
What happens to hemoglobin when oxygen binds?
Binding of oxygen to haem alters oxygen affinity by inducing structural changes in the adjacent globin chains. This molecular ‘co-operativity’ within haemoglobin is responsible for a sigmoidal-shaped oxygen dissociation curve and is influenced by pH, carbon dioxide, and 2,3-diphosphoglycerate.



