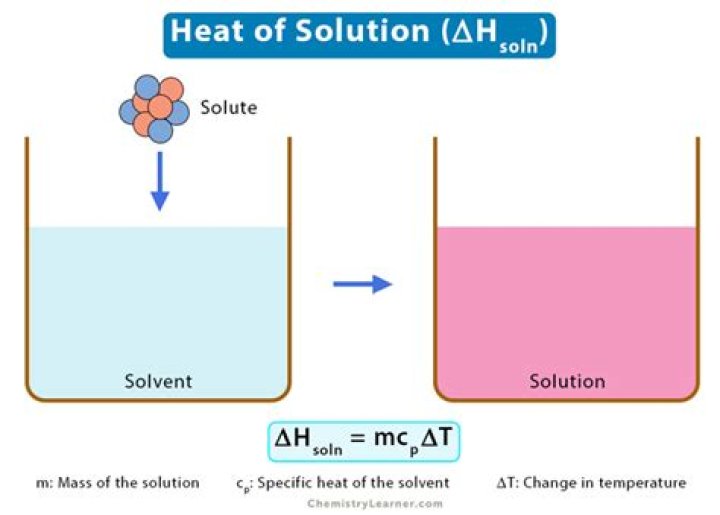
What is heat solvation

Andrew White
Published Mar 11, 2026
Answer Not Found
What does solvent heat mean?
: the heat evolved or absorbed when a substance dissolves specifically : the amount involved when one mole or sometimes one gram dissolves in a large excess of solvent.
Does solvation release heat?
In salts for which the hydration energy is higher than the lattice energy, solvation occurs with a release of energy in the form of heat. For instance, CaCl2 (anhydrous calcium chloride) heats the water when dissolving.
How do you find the heat of solvation?
- Amount of energy released or absorbed is calculated. q = m × Cg × ΔT. q = amount of energy released or absorbed. …
- calculate moles of solute. n = m ÷ M. …
- Amount of energy (heat) released or absorbed per mole of solute is calculated. ΔHsoln = q ÷ n.
What is an example of heat of solution?
Chemical hot packs and cold packs work because of the heats of solution of the chemicals inside them. When the bag is squeezed, an inner pouch bursts, allowing the chemical to dissolve in water. Heat is released in the hot pack and absorbed in the cold pack.
Is solvation exothermic?
As illustrated in (Figure), the formation of a solution may be viewed as a stepwise process in which energy is consumed to overcome solute-solute and solvent-solvent attractions (endothermic processes) and released when solute-solvent attractions are established (an exothermic process referred to as solvation).
What is the difference between Q and Delta H?
Q is the energy transfer due to thermal reactions such as heating water, cooking, etc. anywhere where there is a heat transfer. You can say that Q (Heat) is energy in transit. Enthalpy (Delta H), on the other hand, is the state of the system, the total heat content.
How do you determine the heat of formation?
This equation essentially states that the standard enthalpy change of formation is equal to the sum of the standard enthalpies of formation of the products minus the sum of the standard enthalpies of formation of the reactants. and the standard enthalpy of formation values: ΔH fo[A] = 433 KJ/mol.How do I calculate heat?
Subtract the final and initial temperature to get the change in temperature (ΔT). Multiply the change in temperature with the mass of the sample. Divide the heat supplied/energy with the product. The formula is C = Q / (ΔT ⨉ m) .
How do you calculate heat gained by a solution?Find the solution’s specific heat on a chart or use the specific heat of water, which is 4.186 joules per gram Celsius. Substitute the solution’s mass (m), temperature change (delta T) and specific heat (c) into the equation Q = c x m x delta T, where Q is the heat absorbed by the solution.
Article first time published onWhat happens in solvation process?
In the process of solvation, ions are surrounded by a concentric shell of solvent. Solvation is the process of reorganizing solvent and solute molecules into solvation complexes. Solvation involves bond formation, hydrogen bonding, and van der Waals forces. Solvation of a solute by water is called hydration.
What is the difference between dissociation and solvation?
The key difference between dissociation and solvation is that dissociation is the breakdown of a substance into the atoms or ions from which the substance is made of whereas solvation is the dissolution of a substance in a solvent due to the attraction forces between the solvent molecules and the components of the …
What is heat hydration?
The heat produced by concrete during concrete curing is called heat of hydration. This exothermic reaction occurs when water and cement react. The amount of heat produced during the reaction is mostly related to the composition and fineness of the cement.
Why is heat of solution important?
To form a solution, energy is required to break the bonds between the particles within the solid or liquid. Heat energy is also required to break the bonds in a solvent to insert one of the molecules into the solution.
What is standard heat of solution?
The heat of solution, also referred to the enthalpy of solution or enthalpy of dissolution, is the enthalpy change associated with the dissolution of a solute in a solvent at constant pressure, resulting in infinite dilution.
What does Q MCAT stand for?
Q = Heat energy (in Joules, J) m = Mass of a substance (kg) c = Specific heat (J/kg∙K) ∆T = Change in temperature (Kelvins, K)
What is Q thermochemistry?
Apr 14, 2015. q=mcΔT , in which q is the energy gained or lost, m is mass, c is specific heat capacity, and ΔT is the change in temperature (final temperature – initial temperature).
What is W thermochemistry?
Work Energy used to cause an object that has mass to move. w = F × d • Energy is the ability to do work or transfer heat. … Thermochemistry is the study of chemical reactions and the energy changes that involve heat.
What is solvation in chemistry?
Water chemistry Solvation is the interaction of a solvent with the dissolved solute—in the case of water, solvation is often referred to as hydration. Solvent polarity is the most important factor in determining how well it solvates a particular solute.
Is solvation endothermic?
3: An endothermic solvation process where the endothermic steps absorb more energy than the exothermic releases. Such a process will cool the surroundings.
Which of the following best describes the term solvation?
Solvation is called hydration when the solvent is in water. Solvation involves the interaction between solute molecules and solvent molecules. During solvation, a solute molecule is surrounded by solvent molecules arranged in a particular way.
How do you calculate joules?
In equation form: work (joules) = force (newtons) x distance (meters), where a joule is the unit of work, as defined in the following paragraph. In practical terms, even a small force can do a lot of work if it is exerted over a long distance.
What defines heat?
Heat is the form of energy that is transferred between systems or objects with different temperatures (flowing from the high-temperature system to the low-temperature system). Also referred to as heat energy or thermal energy. Heat is typically measured in Btu, calories or joules.
How do u calculate mass?
Mass is always constant for a body. One way to calculate mass: Mass = volume × density. Weight is the measure of the gravitational force acting on a mass.
What causes heat formation?
Heat energy is the result of the movement of tiny particles called atoms, molecules or ions in solids, liquids and gases. … Heat energy can be transferred from one object to another. The transfer or flow due to the difference in temperature between the two objects is called heat.
What is heat of formation simple?
From Wikipedia, the free encyclopedia. The standard enthalpy of formation or standard heat of formation of a compound is the change of enthalpy during the formation of 1 mole of the substance from its constituent elements, with all substances in their standard states.
How do you find the heat of formation from the heat of combustion?
- Standard heat of combustion: The energy liberated when a substance X undergoes complete combustion, with excess of oxygen at standard conditions (25°C and 1 bar). …
- Example: …
- The heat of combustion of ethanol, ΔHc°(C2H6O, l) = 2*393.51 + 6*142.915 + (-277.6) = 1366.91 kJ/mol.
How do you calculate heat gained by water?
The specific heat capacity of water is 4.18 J/g/°C. We wish to determine the value of Q – the quantity of heat. To do so, we would use the equation Q = m•C•ΔT. The m and the C are known; the ΔT can be determined from the initial and final temperature.
What does a positive Q mean?
When heat is absorbed by the solution, q for the solution has a positive value. This means that the reaction produces heat for the solution to absorb and q for the reaction is negative. … This means that the reaction absorbs heat fron the solution, the reaction is endothermic, and q for the reaction is positive.
What is solvation effect in amines?
In water, the ammonium salts of primary and secondary amines undergo solvation effects (due to hydrogen bonding) to a much greater degree than ammonium salts of tertiary amines. These solvation effects increase the electron density on the amine nitrogen to a greater degree than the inductive effect of alkyl groups.
What factors affect solvation and solvation energy?
There are several factors that effect the rate of solvation. These include: temperature, concentration, surface area of solute, concentration of solvent, and stirring. The general reason for an increase in rate of solvation is that solute molecules come into contact with solvent molecules more often.