What is molecular geometry used for

Andrew White
Published Apr 21, 2026
Molecular geometry or molecular structure is the three-dimensional arrangement of atoms within a molecule. It is important to be able to predict and understand the molecular structure of a molecule because many of the properties of a substance are determined by its geometry.
Why is molecular geometry important give some examples?
Why is molecular geometry important? Cite some examples. The properties of molecules are directly related to their shape. The sensation of taste, immune response, the sense of smell, and many types of drug action all depend on shape-specific interactions between molecules and proteins.
How does molecular geometry affect properties?
What Is Molecular Geometry? Molecular geometry studies the three-dimensional shapes molecules form and how these shapes relate to chemical reactivity and physical characteristics. A molecule’s shape can affect the physical properties of the molecule, such as freezing point, density, and other properties.
Why is molecular shape important for us?
Molecular shapes are important in determining macroscopic properties such as melting and boiling points, and in predicting the ways in which one molecule can react with another.Why is molecular shape important in biology?
Each molecule has a characteristic size and shape that determines its function in the living cell. The shapes of molecules are determined by the positions of the atoms’ orbitals. … Molecular shape is crucial in biology because of the way it determines how most molecules recognize and respond to each other.
What is the molecular geometry of a molecule?
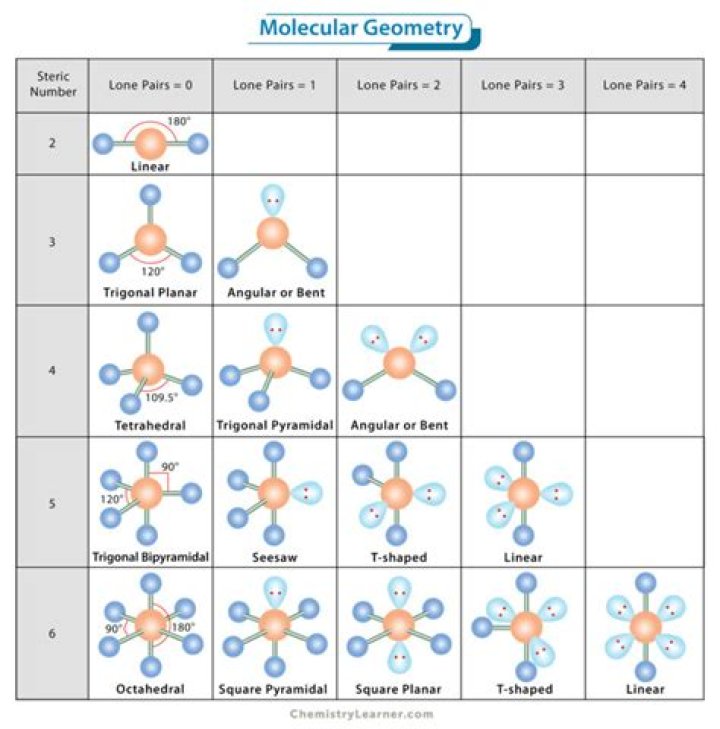
Molecular geometry is the 3-dimensional shape that a molecule occupies in space. It is determined by the central atom and the surrounding atoms and electron pairs. … Some of the most common shapes that can be taken are linear, trigonal planar, tetrahedral, pyramidal, and angular (or bent).
How does molecular geometry affect reactivity?
Another property determined by molecular geometry is reactivity. The reactivity is affected in many ways. One example is the type of bonds the molecule makes based on polarity. For example, some polar molecule can react in hydrogen bonds while non polar molecules react in london dispersion forces.
What would cause the shape of a molecule to be tetrahedral?
Explanation: If there are 4 electron pairs around a central atom, the shape, the geometry that results in LEAST electrostatic interaction is the tetrahedron. For methane, CH4 , the 8 electrons that comprise the 4 C−H bonds demonstrably tend to adopt a shape that minimizes electrostatic repulsion.Why is molecular geometry necessary for determining molecular polarity?
Because molecular polarity results from the VECTOR sum of the individual bond dipoles. Vectors have magnitude and direction, so polarity is in part a function of geometry.
How does molecular geometry affect melting point?The molecular geometry determines how stable the compound is in its solid form. … More stable crystal structures will require higher temperatures to melt in order to overcome the thermodynamic stability in its structure (ie: more heat needed to break apart the intermolecular forces).
Article first time published onWhat factors affect molecular geometry?
Many factors lead to variations from the ideal bond angles of a molecular shape. Size of the atoms involved, presence of lone pairs, multiple bonds, large groups attached to the central atom, and the environment that the molecule is found in are all common factors to take into consideration.
Why is molecular shape important in medicine design?
Shape is a fundamentally important molecular feature that often determines the fate of a compound in terms of molecular interactions with preferred and non-preferred biological targets.
How does the molecular geometry of the atoms affect the polarity of molecules?
Think of each polar bond in a molecule as a little arrow pointing from positive to negative. … So, the geometry of the molecule determines the direction that the bond dipole vectors point. The polarity of the molecule results from adding up all these individual bond dipoles.
What is molecular geometry vs electron geometry?
The molecular geometry definition in chemistry is the arrangement of atoms in relation to a central atom in three-dimensional space. Electron geometry is the arrangement of electron groups.
What molecular geometries can you considered as polar?
Formula3D Structure Shape Polarity1.CH4tetrahedral nonpolar2.NH3trigonal pyramidal polar3.H2Obent polar4.H3O+trigonal pyramidal charged
What are the different molecular geometry?
Molecular Geometries. The VSEPR theory describes five main shapes of simple molecules: linear, trigonal planar, tetrahedral, trigonal bipyramidal, and octahedral.
How do you find the molecular geometry?
- Draw the Lewis Structure.
- Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons. …
- Name the electron-group geometry. …
- Looking at the positions of other atomic nuclei around the central determine the molecular geometry.
Why are a Molecules atoms as far away?
According to VSEPR, the valence electron pairs surrounding an atom mutually repel each other; they adopt an arrangement that minimizes this repulsion, thus determining the molecular geometry. This means that the bonding (and non-bonding) electrons will repel each other as far away as geometrically possible.
What is octahedral molecular geometry in chemistry?
In chemistry, octahedral molecular geometry describes the shape of compounds with six atoms or groups of atoms or ligands symmetrically arranged around a central atom, defining the vertices of an octahedron. The octahedron has eight faces, hence the prefix octa.
Does molecular geometry affect boiling point?
Large molecules have more electrons and nuclei that create van der Waals attractive forces, so their compounds usually have higher boiling points than similar compounds made up of smaller molecules. … Molecular shape is also important, as the second group of compounds illustrate.
How does molecular structure affect physical properties?
The properties of organic molecules depend on their structure being made of simple molecules . … The intermolecular forces between molecules are weaker. The intermolecular forces vary between molecules, so different organic compounds have different melting points and boiling points.
How are molecular geometry and dipole moments related to physical properties such as melting point and boiling point?
Physical properties like boiling point and melting point depend upon the existence and magnitude of the dipole moment of a molecule. In general, molecules that have substantial dipole moments are likely to exhibit greater intermolecular interactions, resulting in higher melting points and boiling points.
What is ideal molecular geometry?
The five ideal shapes are: linear, trigonal planar, tetrahedral, trigonal bipyramidal and octahedral. One important point to note about molecular shape is that all diatomic (compounds with two atoms) compounds are linear. So H2, HCl and Cl2 are all linear.
What two theories can be used to predict molecular geometry?
The theories that affect the shape of a molecule are VSEPR and hybridization. Hybridization is the formation of hybrid orbitals from atomic orbitals which influences molecular geometry and bonding properties.
What techniques are used in drug designing?
SBDD includes approaches such as molecular docking, virtual screening (VS), structure-based pharmacophore modeling, and de novo drug design, whereas LBDD approaches include similarity-based screening, quantitative structure–activity relationship (QSAR) modeling, ligand-based pharmacophore modeling, and scaffold hopping …
Why is shape so important in drugs and medicinal effects?
3.2 Chirality and the Shape of Molecules Stereochemistry is of critical importance to drug action because the shape of a drug molecule is an important factor in determining how it interacts with the various biological molecules (enzymes, receptors, etc.) that it encounters in the body.
How computers are used in drug designing?
Computer-aided drug design uses computational approaches to discover, develop, and analyze drugs and similar biologically active molecules. The ligand-based computer-aided drug discovery (LB-CADD) approach involves the analysis of ligands known to interact with a target of interest.
What is the molecular geometry around an atom in a molecule or ion which is surrounded by three lone pairs of electrons and two single bonds?
If these are all bond pairs the molecular geometry is tetrahedral (e.g. CH4). If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3). If there are two bond pairs and two lone pairs of electrons the molecular geometry is angular or bent (e.g. H2O).
What will be the molecular geometry for a molecule with a central atom that has five regions of electron density if one of the regions of electron density is a lone pair?
The electron geometry about a central atom that has five regions of electron density is trigonal bipyramidal. This is one of the common geometries…
What is the molecular geometry of a molecule with 4 outer atoms and 1 lone pair on the central atom?
# of bonding groups/domains on ‘central’ atom# of lone pair electrons on ‘central’ atomMolecular Geometry40tetrahedral31trigonal pyramidal22bent50trigonal bipyramidal
Under what circumstances is the molecular geometry around a single central atom the same as the electron group geometry around the central atom?
The molecular geometry is the geometrical arrangement of the atoms around the central atoms. The electron geometry and the molecular geometry are the same when every electron group bonds two atoms together. The presence of unbonded lone pair electrons gives a different molecular geometry and electron geometry.