What is Q MC

Rachel Hunter
Published Apr 02, 2026
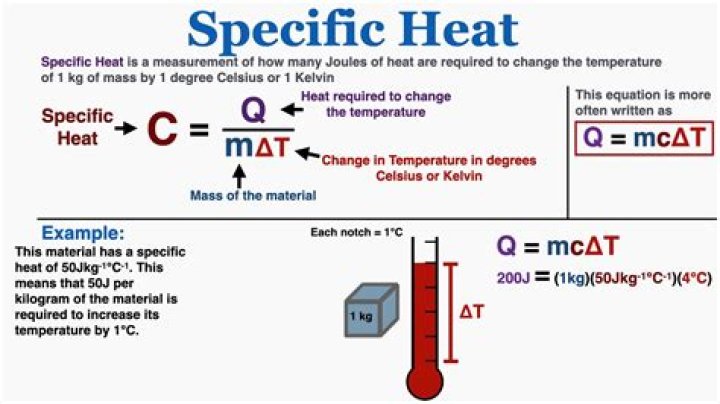
Q=mcΔT Q = mc Δ T , where Q is the symbol for heat transfer, m is the mass of the substance, and ΔT is the change in temperature. The symbol c stands for specific heat and depends on the material and phase. The specific heat is the amount of heat necessary to change the temperature of 1.00 kg of mass by 1.00ºC.
What does Q MC mean?
Q = mc∆T. Q = heat energy (Joules, J) m = mass of a substance (kg) c = specific heat (units J/kg∙K) ∆ is a symbol meaning “the change in”
How do you calculate Q MCAT?
In the equation Q=mcΔt: Q= the heat energy (Joules), m= the mass of the object/substance being heated (in this case it’s water; also important in this situation to remember that 1ml of water is equal to 1g of water), c= the specific heat of the object/substance being heated (again…
What equation is Q MC ∆ T?
The amount of heat gained or lost by a sample (q) can be calculated using the equation q = mcΔT, where m is the mass of the sample, c is the specific heat, and ΔT is the temperature change.What is Q MCAT used for?
Q=mcΔT Q = mc Δ T , where Q is the symbol for heat transfer, m is the mass of the substance, and ΔT is the change in temperature. The symbol c stands for specific heat and depends on the material and phase. The specific heat is the amount of heat necessary to change the temperature of 1.00 kg of mass by 1.00ºC.
What is MCP Delta?
Q = m * Cp * dT heat = mass flow * specific heat capacity * temperature difference kW = kg/s * kJ/kg/°C * °C.
What is Q mCpdT?
Q = mCpdT shows the heat required to raise the temperature of a material through a finite temperature difference (dT). … Q = mCpdT shows the heat required to raise the temperature of a material through a finite temperature difference (dT).
Is Delta H the same as Q?
Q is the energy transfer due to thermal reactions such as heating water, cooking, etc. anywhere where there is a heat transfer. You can say that Q (Heat) is energy in transit. Enthalpy (Delta H), on the other hand, is the state of the system, the total heat content.How do you find q in chemistry?
The equation for Q, for a general reaction between chemicals A, B, C and D of the form: Is given by: So essentially it’s the products multiplied together divided by the reactants multiplied together, each raised to a power equal to their stoichiometric constants (i.e. the numbers of each component in the reaction).
What does Deltat equal?The equation of delta t is: ΔT = T2 – T1.
Article first time published onIs Q MCT in kJ or J?
You want your q to be in units of Joules or kJ. If you used the q=mC(delta T) with your given C, your q would be in units of (grams)(kJ).
How do you find Q surroundings?
We wish to determine the value of Q – the quantity of heat. To do so, we would use the equation Q = m•C•ΔT. The m and the C are known; the ΔT can be determined from the initial and final temperature.
How do you find Q heat?
To calculate the amount of heat released in a chemical reaction, use the equation Q = mc ΔT, where Q is the heat energy transferred (in joules), m is the mass of the liquid being heated (in kilograms), c is the specific heat capacity of the liquid (joule per kilogram degrees Celsius), and ΔT is the change in …
What is the C in Q MCT?
Q = Heat energy (in Joules, J) m = Mass of a substance (kg) c = Specific heat (J/kg∙K)
What units are Q?
So, the units for Q are Joules (energy) divided by area (square meters) and time (seconds). Joules/(m^2∙sec). Since power is defined as energy divided by time and 1 Watt is equal to 1 Joule/second, Q can also be expressed as Watts/m^2 .
What is Q of a substance?
q = (mass of the substance)(specific heat of the substance)(DT) The sign associated with q will depend on DT. When the final temperature is higher than the initial temperature, the value of DT will have a positive sign (+).
What are calorimeters used to measure?
calorimeter, device for measuring the heat developed during a mechanical, electrical, or chemical reaction, and for calculating the heat capacity of materials.
What is C water?
Water has a specific heat capacity of 4.186 J/g°C, meaning that it requires 4.186 J of energy (1 calorie) to heat a gram by one degree.
What is Q heat transfer?
The letter Q represents the amount of heat transferred in a time t, k is the thermal conductivity constant for the material, A is the cross sectional area of the material transferring heat, Δ T \Delta T ΔT is the difference in temperature between one side of the material and the other, and d is the thickness of the …
What is Q UA Delta T?
q = UA∆Tm where ∆Tm is the mean temperature difference. … In other words, the rate of heat transfer can be calculated using the heat transfer coefficient, the total area, and the log mean temperature difference.
What does Q stand for in chemistry?
The reaction quotient Q is a measure of the relative amounts of products and reactants present in a reaction at a given time.
What is Q in Nernst equation?
Determine Q, the reaction quotient. Deternine n, the number of electrons transferred in the reaction “n”. Determine Ecell, the cell potential at the non-standard state conditions using the Nernst equation.
What is KP in chemistry?
Equilibrium constant expression in terms of partial pressure is designated as Kp. Equilibrium constant Kp is equal to the partial pressure of products divided by partial pressure of reactants and the partial pressure are raised with some power which is equal to the coefficient of the substance in balanced equation.
What is the difference between H and Q?
Where H is the enthalpy, E is the internal energy , P is the equilibrium pressure of a sytem whose volume is V. q is defined as the heat transferred between the system and its surroundings during some thermodynamic process and is therefore a property of the process, not the system.
What is W thermochemistry?
Work Energy used to cause an object that has mass to move. w = F × d • Energy is the ability to do work or transfer heat. … Thermochemistry is the study of chemical reactions and the energy changes that involve heat.
Is negative Q exothermic?
We know that if we have an exothermic reaction the system loses heat and the sign of q is negative. If we have an endothermic reaction heat is gained by the system and the sign of q is positive.
What should the delta t be on a boiler?
A typical boiler supply Delta T is around 12 degrees.
What is chiller Deltat?
“Delta T” is the most common use of the word delta in the HVAC industry, meaning temperature difference. If the temperature before a cooling coil is 75F and the temperature after the cooling coil is 55F, subtract 55F from a 75F to find a delta t of 20F.
What does triangle T stand for?
ΔT (timekeeping) the difference between two time scales, Universal Time and Terrestrial Time, which results from a drift in the length of a day. The interval of time used in determining velocity. The increment between successive nerve impulses.
What is Deltat?
Delta T, or the difference between return air temperature and supply air temperature, is one of the most commonly used measurements in the HVAC industry – and for good reason. … Calculating Delta T is simple: just subtract the return air temperature from the supply air temperature.
What is bomb in bomb calorimeter?
The bomb calorimeter is used to determine the calorific values of solid and liquid fuels. It consists of a strong steel shell known as a bomb. It consists of a base which supports the platinum crucible and is screwed to the body of the bomb.



