What is scFv antibody

Isabella Wilson
Published Feb 23, 2026
To date, generation of single-chain fragment variable (scFv) has become an established technique used to produce a completely functional antigen-binding fragment in bacterial systems. The advances in antibody engineering have now facilitated a more efficient and generally applicable method to produce Fv fragments.
What is scFv used for?
To date, generation of single-chain fragment variable (scFv) has become an established technique used to produce a completely functional antigen-binding fragment in bacterial systems. The advances in antibody engineering have now facilitated a more efficient and generally applicable method to produce Fv fragments.
How are scFv produced?
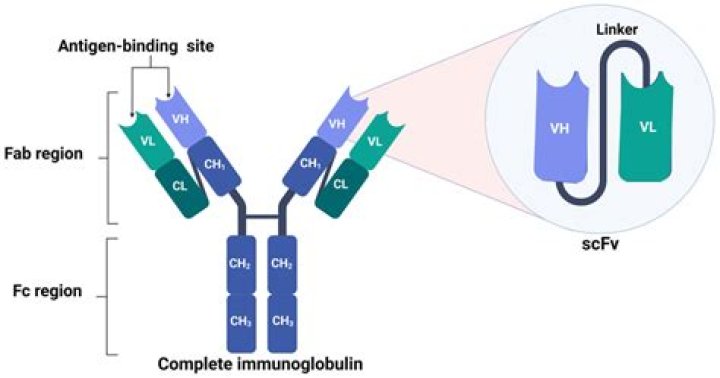
Single-chain variable fragment (scFv) is a class of engineered antibodies generated by the fusion of the heavy (VH) and light chains (VL) of immunoglobulins through a short polypeptide linker.
What is scFv composed of?
Single-chain variable fragments (scFvs) are small-sized artificial constructs composed of the immunoglobulin heavy and light chain variable regions connected by a peptide linker.What is an scFv What is a benefit of scFvs compared to traditional antibodies?
ScFv fragments retain the binding specificity of the parent antibody and offer several advantages compared to full-length mAbs. They display improved pharmacokinetic properties, such as better tissue penetration and rapid blood clearance, which may be beneficial in radiotherapy and diagnostic applications.
How long is an scFv?
In a single chain variable fragment (scFv), VH and VL domains are linked together by a short peptide linker that is usually 15–20 amino acids long, while in diabodies two scFv are assembled together.
Is scFv glycosylated?
In humans, the antibody synthesized is usually glycosylated in the Fc region, which stabilizes the antibody and is necessary for the antibody-dependent immune responses. … The immunogenicity generated by the Fc portion of the antibody is absent in the conventional scFv molecule.
What is a bispecific drug?
Bispecific antibodies are artificial proteins that have promising applications in the field of cancer immunotherapy. They are comprised of two monoclonal antibodies held together by a flexible peptide linker. As the name suggests, this makes them able to bind to two different antigens.What is scFv FC?
The scFv-Fc format allows for rapid characterization of candidate scFvs isolated from phage display libraries before conversion into a full-length IgG. This format offers several advantages over the phage display-derived scFv, including bivalent binding, longer half-life, and Fc-mediated effector functions.
What is antibody CDR?Complementarity-determining regions (CDRs) are part of the variable chains in immunoglobulins (antibodies) and T cell receptors, generated by B-cells and T-cells respectively, where these molecules bind to their specific antigen. A set of CDRs constitutes a paratope.
Article first time published onHow do I get ScFv?
In order to get scFv antibody fragments, mRNA is first isolated from hybridoma (or spleen, lymph cells, and bone morrow) followed by reverse transcribed into cDNA to serve as a template for antibody genes amplification (PCR).
Where does protein bind to IgG?
Protein A antibody binding It has been shown via crystallographic refinement that the primary binding site for protein A is on the Fc region, between the CH2 and CH3 domains. In addition, protein A has been shown to bind human IgG molecules containing IgG F(ab’)2 fragments from the human VH3 gene family.
What is the domain of an antibody?
Antibodies all have the same basic structure consisting of two heavy and two light chains forming two Fab arms containing identical domains at either end attached by a flexible hinge region to the stem of the antibody, the Fc domain, giving the classical ‘Y’ shape.
What is a single chain protein?
Single-chain antigen-binding proteins are novel recombinant polypeptides, composed of an antibody variable light-chain amino acid sequence (VL) tethered to a variable heavy-chain sequence (VH) by a designed peptide that links the carboxyl terminus of the VL sequence to the amino terminus of the VH sequence.
What advantage does an antibody fragment offer over a full size antibody?
One advantage of fragments over full-size antibodies is that antibody fragments are smaller than conventional antibodies and generally lack glycosylation, allowing their production in prokaryotic expression systems, which provide time and cost savings.
How big is a nanobody?
A VHH antibody corresponds to the variable region of a heavy chain of a camelid antibody and has a very small size of around 15 kDa – hence the name “nanobody”.
What is phage display technology?
Phage display technology is an in vitro screening technique for identifying ligands for proteins and other macromolecules. At the crux of phage display technology is the ability to express peptide or protein sequences as fusions to the coat proteins of a bacteriophage.
Which design format of antibody would be most commonly manufactured today?
The building block that is most frequently used to create novel antibody formats is the single-chain variable (V)-domain antibody fragment (scFv), which comprises V domains from the heavy and light chain (VH and VL domain) joined by a peptide linker of ~15 amino acid residues.
How do Nanobodies work?
Nanobodies for photothermal therapy. Nanobodies, which are able to bind tumor antigens like HER2, are coupled to branched gold nanoparticles that absorb light energy and create heat in order to kill cancer cells.
What is panning in phage display?
It involves conjugating the phage library to the desired target. This procedure is termed panning. It utilizes the binding interactions so that only specific peptides presented by bacteriophage are bound to the target. For example, selecting antibody presented by bacteriophage with coated antigen in microtiter plates.
Are bispecific antibodies immunotherapy?
Following the clinical success of immune checkpoint antibodies targeting CTLA-4, PD-1 or PD-L1 in cancer treatment, bispecific antibodies are now emerging as a growing class of immunotherapies with potential to further improve clinical efficacy and safety.
Are bispecific antibodies natural?
The bispecific antibodies that occur naturally in vivo may play a special role in the immune responses associated with human diseases. Natural bispecific antibodies are functionally monovalent and therefore cannot crosslink antigens and trigger pathophysiological effects associated with antigen aggregation.
How many bispecific antibodies are FDA approved?
So far, the FDA has approved three bispecific antibodies while over a hundred more are in development.
How do you identify CDR in antibodies?
Exact identification of complementarity determining regions (CDRs) is crucial for understanding and manipulating antigenic interactions. One way to do this is by marking residues on the antibody that interact with B cell epitopes on the antigen.
Which CDR is most important?
All six antibody CDRs can be involved in antigen recognition (3, 13), though the CDRH3 loop is often the most important (14, 15). The structural complementarity between the binding sites of the antigen receptor and their cognate antigen governs the binding interactions.
What is CDR H3?
CDR H3 is the most diverse region in antibodies due to its position relative to the V(D)J recombination sites, junctional diversification at these sites, and somatic hypermutation. Accordingly, the CDR H3 loop often plays a central role in antigen recognition and is a major contributor to binding strength.
What type of protein binds to antibody?
Antibody binding proteins such as Protein A and Protein G are often used in antibody purification and in applications such as immunoprecipitation (IP) and chromatin immunoprecipitation (ChIP). Each antibody binding protein varies in its ability to bind to antibodies of different subtypes and species.
What is IgG made of?
Immunoglobulin G (IgG) is the major class of immunoglobulins. About three-quarters of all serum immunoglobulins belong to this class. IgG molecules consist of two heavy γ and two light chains (2γ + 2L). Normally each molecule of IgG has two identical antigen combining sites.
How do you purify antibodies?
Antibody fragments can be purified if they contain the region that interacts with the ligand attached to the matrix. scFv, Fab, and dAb can all be purified using affinity chromatography. Affinity chromatography can be used as the only purification step for applications that do not require the highest purity.
What happens when an antigen foreign invader enters the body and how the body's immune system fights off this invasion?
When an antigen enters the body, the immune system produces antibodies against it. Antibodies are always Y-shaped. It is like a battle with the army (antibody) fighting off the invader (antigen).
Are antibodies tertiary or quaternary?
For antibodies, each polypeptide chain has a tertiary structure composed of different domains, in which the basic structural unit of each domain is a barrel-shaped structure formed from two anti-parallel β-sheets. This unique barrel-shaped fold of the antibody is also known as the immunoglobulin fold.



