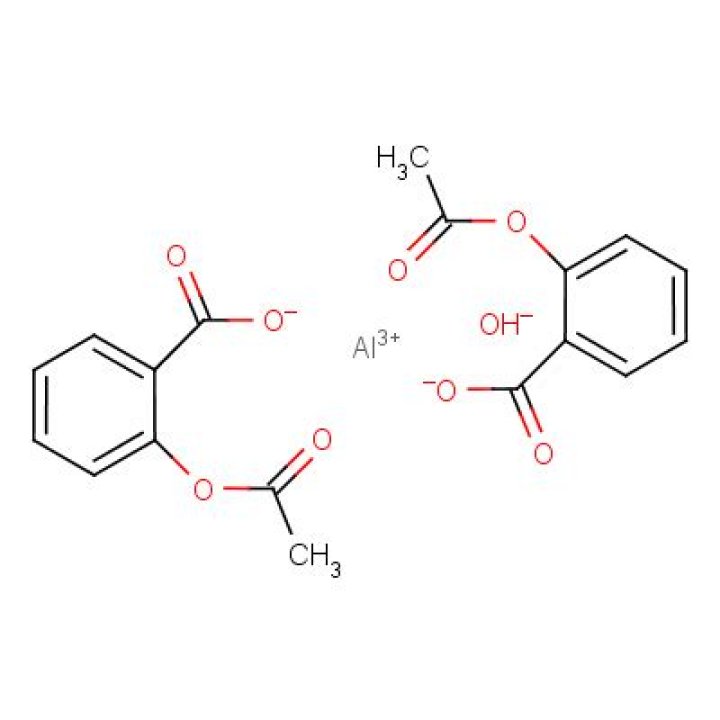
What is sodium Acetylsalicylate

Andrew White
Published Apr 05, 2026
ABSTRACT: Acetylsalicylic acid (trade name aspirin) is a well-known drug with antipyretic and analgesic effects. Mixtures that benefit from better solubility of its sodium salt have been sold for almost 90 years.
Is sodium salicylate harmful?
Harmful if swallowed. Skin Harmful if absorbed through skin. Causes skin irritation. Eyes Causes eye irritation.
What is the use of sodium salicylate?
Uses. It is used in medicine as an analgesic and antipyretic. Sodium salicylate also acts as non-steroidal anti-inflammatory drug (NSAID), and induces apoptosis in cancer cells and also necrosis. It is also a potential replacement for aspirin for people sensitive to it.
Is sodium Acetylsalicylate aspirin?
PHARMACONTROL SODIUM ACETYLSALICYLATE CLASSIFICATION AS “SUBSET OF ASPIRIN” should be established by FDA, qualifying the drug for Category I OTC status, the company said in a recent letter to the agency.How is sodium Acetylsalicylate formed?
This is an acid-base reaction in which the acetylsalicylic acid reacts with the base sodium hydroxide to produce the salt sodium acetylsalicylate and water (acid + base → salt + water).
How much salicylate is toxic?
Salicylate levels greater than 100 mg/dL are considered severe toxicity and occur 12 to 24 hours after ingestion. Damage to the basement membranes will cause cerebral and pulmonary edema. Patients may become obtunded and develop seizures.
Is sodium salicylate safe for skin?
Like all acids, SA can produce somatosensory and visible irritation to the skin and as such may be unsuitable for subjects with sensitive skin. Aims: To provide evidence that sodium salicylate (SS) obtained from neutralization of 1% SA by sodium hydroxide can deliver significant anti-aging benefits.
What is the common chemical name of aspirin?
The Chemistry of Aspirin (acetylsalicylic acid) Aspirin is prepared by chemical synthesis from salicylic acid, through acetylation with acetic anhydride.What happens when you add HCl to sodium Acetylsalicylate?
If 6M HCl is added to the solution of sodium acetylsalicylate, it forms aspirin.
Why are salts of aspirin more soluble?Aspirin contains polar functional groups which can form hydrogen bonds with polar water molecules. … Ionic salts of aspirin, such as sodium acetylsalicylate, are more soluble in water since they form stronger ion-dipole interactions with water.
Article first time published onWhat is the difference between aspirin and sodium salicylate?
Sodium salicylate is considered somewhat less effective in reducing pain when compared with aspirin. However, some patients who are hypersensitive to aspirin may tolerate sodium salicylate. The dose of sodium salicylate is the same as for aspirin—325 mg to 650 mg every 4 hours, as needed.
Can you take sodium salicylate and Tylenol together?
A few reports have suggested that acetaminophen and salicylates used together may cause kidney damage or cancer of the kidney or urinary bladder. This may occur if large amounts of both medicines are taken together for a very long time.
Does sodium salicylate raise blood pressure?
Consistently, our results demonstrate that sodium salicylate lowers blood pressure in SHR but not in WKY, further supporting that inhibiting RhoA/Rho kinase is the major mechanism for the unique blood pressure effect of salicylates.
Where can you find the acid ch3cooh at your home?
Vinegar is a solution of acetic acid in water and contains between 5% to 8% ethanoic acid by volume.
Is aspirin flammable?
Not flammable or combustible.
Is aspirin soluble in water?
Aspirin is only slightly soluble in water so if you add it to the reaction mixture the product would rather clump together than be in the water phase. This causes the solid to begin to form or ‘precipitate’.
Is sodium salicylate preservative?
Sodium Salicylate is used as a denaturant and preservative (0.09% to 2%). The TEA salt of Salicylic Acid is used as an ultraviolet (UV) light absorber (0.0001% to 0.75%).
Does sodium salicylate exfoliate?
Sodium Salicylate | Paula’s Choice. Salt form of salicylic acid (BHA). Because it is not the acid form of salicylate (i.e., salicylic acid), it does not have exfoliating properties.
What is salicylate in skincare?
Salicylate is a chemical found naturally in plants and is the main ingredient of the oral pain-relieving tablet, aspirin. It is also found in many fruits and vegetables as well as in many toiletry and cosmetic products.
What organ is damaged by taking too much aspirin?
Symptoms of Aspirin Poisoning Severe disease can cause acute kidney injury… read more ), kidney failure. Kidney failure is the inability of the kidneys to adequately filter metabolic waste products from the blood. Kidney… read more , and difficulty breathing.
Is ibuprofen a salicylate?
Currently available oral OTC analgesics include acetaminophen, nonsteroidal anti-inflammatory drugs (NSAIDs [ibuprofen and naproxen]), and salicylates (aspirin, magnesium salicylate, and sodium salicylate). Criteria in selecting an appropriate analgesic include a thorough assessment of the patient’s medical, medication …
What are the signs and symptoms of salicylate toxicity?
Salicylate poisoning can cause vomiting, tinnitus, confusion, hyperthermia, respiratory alkalosis, metabolic acidosis, and multiple organ failure. Diagnosis is clinical, supplemented by measurement of the anion gap, arterial blood gases, and serum salicylate levels.
Is aspirin soluble in NaOH?
Aspirin would be slightly soluble in NaOH because the carboxylic acid group would be deprotonated making it negatively charged.
How do you know how much aspirin is in a tablet?
6. Calculate the mass of aspirin in each tablet according to the equation below. (MW of aspirin = 180 g/mole) Moles of aspirin × Molecular Weight of aspirin (in g/mole) = Mass of aspirin (in grams) 7. Record the mass of aspirin in each tablet (as claimed on the bottle) in Table 3.
What is the reason for boiling the NaOH and aspirin mixture?
This involves reacting the aspirin solution with a measured amount of sodium hydroxide; an amount that will exceed the amount of aspirin present. Because the hydrolysis reaction occurs at a very low rate at room temperature it will be heated to increase the reaction rate.
Why is aspirin banned?
NEW DELHI: The Delhi government on Tuesday banned without-prescription sale of non-steroidal anti-inflammatory drugs (NSAIDs) such as aspirin, disprin, brufen and voveran because of the danger these drugs pose to dengue patients.
Is Ibuprofen A aspirin?
No. The pain reliever in Advil is ibuprofen, however both are part of the class of drugs known as NSAIDs (nonsteroidal anti-inflammatory drug).
Is aspirin a paracetamol?
It reduces or completely prevents the production of prostaglandins – a pain and inflammation-causing chemical find throughout the body. However, paracetamol targets the prostaglandins found in the brain. Aspirin, acetylsalicylic acid, is a non-steroidal anti-inflammatory drug.
Is aspirin soluble in vinegar?
Similar to what happened in the vinegar, an uncoated aspirin would quickly dissolve in the stomach. For some people with sensitive stomachs, the quickly-dissolved aspirin could cause discomfort. The enteric-coated tablet did not react to the vinegar, so it would not impact the person taking it.
Why does aspirin smell like vinegar?
When acetylsalicylic acid ages, it may decompose and return to salicylic acid and acetic acid. If you have a very old bottle of aspirin around the house, open it and take a sniff. It may smell like vinegar, because vinegar is dilute acetic acid.
What improves aspirin solubility?
The solubility increases with increasing environmental pH and the solubility of aspirin at starting pH above 3.5 is limited by the solution buffer capacity. It has been suggested that aspirin solubility can exceed 200 mg/ml (i.e., 100 g/500 ml) if the solution ending pH can be kept above 5 (Fahmy et al.