What is the Bohr model of fluorine

Lily Fisher
Published Feb 21, 2026
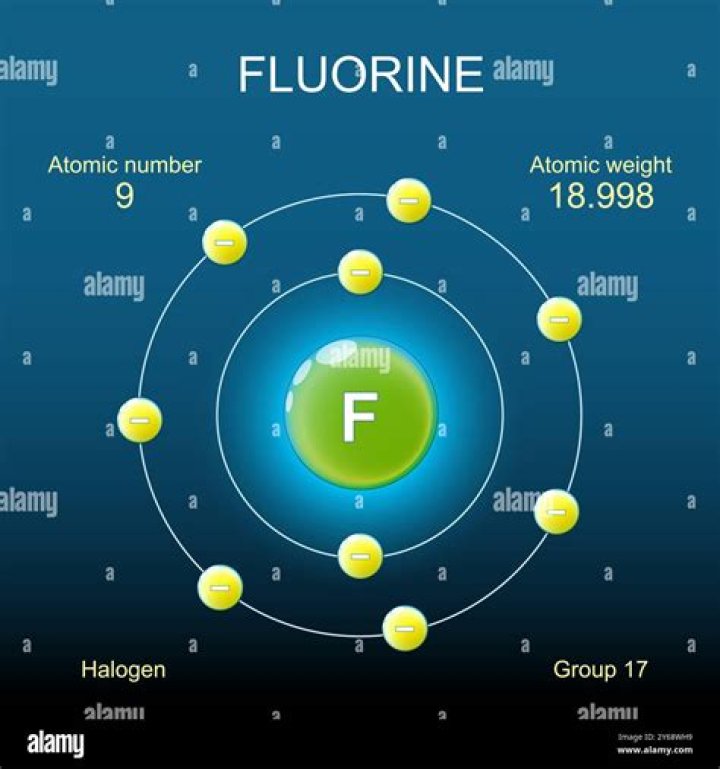
The Bohr model of Fluorine is drawn with only two electron shells, the first shell contains 2 electrons and the second shell contains 7 electrons. Fluorine is neutral and its atomic number is 9, hence, the number of protons and electrons available for its Bohr diagram is also 9.
What is the Bohr model of fluorine? - Google Search
The model states that electrons in atoms move in circular orbits around a central nucleus and can only orbit stably in certain fixed circular orbits at a discrete set of distances from the nucleus. These orbits are associated with definite energies and are also called energy shells or energy levels.
What is the isotope symbol for fluorine?
IsotopeAtomic Mass (uncertainty) [u]Abundance (uncertainty)19F18.998 403 163(6)1
How do you find the Bohr model of an element?
- Find the number of protons, neutrons and electrons for the atom. The number of protons is the atomic number. …
- Set up the diagram. To set up the diagram, you will need a circle in the middle. …
- Add in orbitals and electrons.
What is F in chemistry periodic table?
Fluorine – Element information, properties and uses | Periodic Table.
When drawing Bohr models What is the first thing that you should draw?
You must fill one level before going on to draw the next level! In order to draw Bohr models of these elements, you must first determine the number of protons, neutrons, and electrons. Once you have found this information, follow the directions to draw your model.
Is Bohr model correct?
This model was proposed by Niels Bohr in 1915; it is not completely correct, but it has many features that are approximately correct and it is sufficient for much of our discussion.
How fluorine got its name?
Where did fluorine get its name? The name fluorine is derived from the mineral fluorite which comes from the Latin word “fluere” meaning “to flow.” The name was suggested by English chemist Sir Humphry Davy. Fluorine has one stable isotope, fluorine-19.What is the atomic name of fluorine?
Fluorine is a chemical element with the symbol F and atomic number 9. It is the lightest halogen and exists at standard conditions as a highly toxic, pale yellow diatomic gas. As the most electronegative element, it is extremely reactive, as it reacts with all other elements, except for argon, neon, and helium.
Who discovered fluorine 18?The noble gases, or Group 18 (VIIIA), also known as the inert gases, generally do not react with other elements. Fluorine was discovered in 1886 by French chemist Henri Moissan (1852-1907). Moissan collected the gas by passing an electric current through one of its compounds, hydrogen fluoride (H 2 F 2 ).
Article first time published onWhat is the electronegativity of F?
The concept of electronegativity was introduced by Linus Pauling in 1932; on the Pauling scale, fluorine is assigned an electronegativity of 3.98, and the other elements are scaled relative to that value.
How do you make fluorine?
Fluorine is produced commercially by electrolyzing anhydrous hydrogen fluoride containing dissolved potassium fluoride to achieve adequate conductivity (Jaccaud and Faron 1988; Shia 1994). Potassium fluoride and hydrogen fluoride form potassium bifluoride (KHF2 or KF·HF).
Where do you find fluorine?
Fluorine occurs naturally in the earth’s crust where it can be found in rocks, coal and clay. Fluorides are released into the air in wind-blown soil. Fluorine is the 13th most aboundant element in the Earth’s crust: 950 ppm are contanined in it.
What is James Chadwick model?
This atomic model is known as the quantum mechanical model of the atom. … In 1932, James Chadwick bombarded beryllium atoms with alpha particles. An unknown radiation was produced. Chadwick interpreted this radiation as being composed of particles with a neutral electrical charge and the approximate mass of a proton.
What is James Chadwick atomic theory?
Chadwick is best known for his discovery of the neutron in 1932. A neutron is a particle with no electric charge that, along with positively charged protons, makes up an atom’s nucleus. … In this way, Chadwick’s findings were pivotal to the discovery of nuclear fission, and ultimately the development of the atomic bomb.
Why Niels Bohr proposed this model?
Bohr Atomic Model : In 1913 Bohr proposed his quantized shell model of the atom to explain how electrons can have stable orbits around the nucleus. … To remedy the stability problem, Bohr modified the Rutherford model by requiring that the electrons move in orbits of fixed size and energy.
What are the four principles of Bohr's model?
Main Points of the Bohr Model Electrons orbit the nucleus in orbits that have a set size and energy. The energy of the orbit is related to its size. The lowest energy is found in the smallest orbit. Radiation is absorbed or emitted when an electron moves from one orbit to another.
What is electron configuration and Bohr model?
The Bohr model shows the atom as a central nucleus containing protons and neutrons with the electrons in circular orbitals at specific distances from the nucleus. These orbits form electron shells or energy levels, which are a way of visualizing the number of electrons in the various shells.
How many electrons do F have?
Fluorine is the ninth element with a total of 9 electrons. In writing the electron configuration for fluorine the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for F go in the 2s orbital. The remaining five electrons will go in the 2p orbital.
What information is the same in a Bohr model electron configuration and a full configuration?
nl202s
Is fluorine and fluoride the same?
Fluoride is chemically related to fluorine, but they are not the same. Fluoride is a different chemical compound. Fluoride is created from salts that form when fluorine combines with minerals in soil or rocks. Fluoride is usually very stable and relatively unreactive, unlike its chemical relative fluorine.
What is the Latin name of fluorine?
Fluere is the Latin word for flow and provides the root for the name of the element we know as fluorine. One of the common naturally occurring sources of fluorine is the mineral calcium fluoride, also known as fluorspar.
What is the atomic structure of fluorine?
Atomic Number9Number of Neutrons10Shell structure (Electrons per energy level)[2, 7]Electron Configuration[He] 2s2 2p5Valence Electrons2s2 2p5
Who explained fluorine in 1771?
Andreas Sigismund Marggraf made the first definite preparation of hydrofluoric acid in 1764 when he heated fluorite with sulfuric acid in glass, which was greatly corroded by the product. In 1771, Swedish chemist Carl Wilhelm Scheele repeated this reaction.
How many rings does fluorine have?
ZElementNo. of electrons/shell8Oxygen2, 69Fluorine2, 710Neon2, 811Sodium2, 8, 1
What is the difference between fluorine-18 and fluorine 19?
Only fluorine-19 is stable and naturally occurring; therefore, fluorine is a monoisotopic and mononuclidic element, and only artificially produced fluorine isotopes have mass numbers other than 19. The longest-lived radioisotope is 18F; it has a half-life of 109.739 minutes.
Which has higher electronegativity F or Cl?
F is the most electronegative element on the periodic table. Followed by O, then N and Cl. In general the electronegativity of an element increases as one goes up a family.
Which has more electronegativity F or Cl?
F is the more electronegative than Cl. The reasons is that it has very small size and Electronegativity depends inversely on size of element.