What is the catalyst in the Ostwald process

Andrew Campbell
Published May 12, 2026
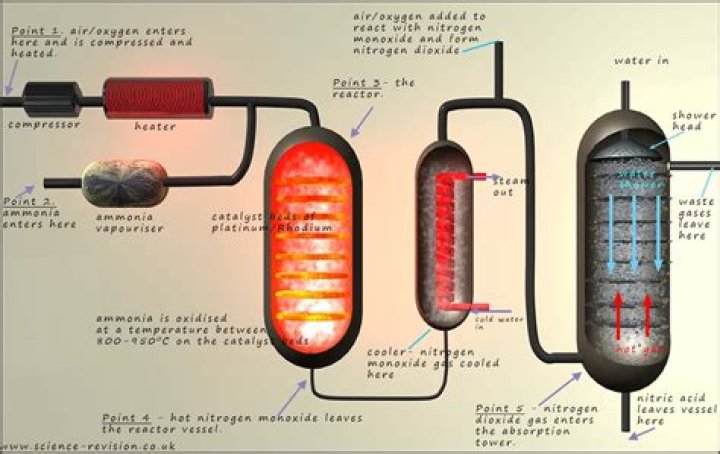
The Ostwald process is used to produce nitric acid. Ammonia is the feedstock for this reaction. It is reacted with oxygen and water using a platinum catalyst to produce nitric acid.
What are the reactants in the Ostwald process?
Reactants required for Ostwald process are ammonia and oxygen of air.
What is formed in Ostwald process?
Nitric acid is most commonly manufactured by Ostwald process. The Ostwald process converts ammonia to nitric acid and involves two steps. In step 1, ammonia is oxidized to form nitric oxide and also nitrogen dioxide. Then in step 2, the nitrogen dioxide that was formed is absorbed in water.
Why is a platinum catalyst used in the Ostwald process?
Most catalytic oxidations of ammonia follow the Ostwald process. In this process, platinum is used as the catalyst. … This gas is adsorbed by the water, which allows the reaction to occur to reduce some of it back into nitric oxide and the rest into the desired product of nitric acid.What is the catalyst used in Haber process?
Why is Iron Used as a Catalyst in the Haber Process? Iron can be used in the Haber process as a low-cost catalyst.
Why are two different catalysts used in the production of nitric acid?
Higher catalyst temperatures increase reaction selectivity toward the production of nitric oxide (NO), while lower catalyst temperatures tend to be more selective toward the production of nitrogen (N2) and nitrous oxide (N2O).
Which catalyst is used in oxidizing nh3 in Ostwald process?
Catalyst used in Ostwald process is Platinum. During ostwald process ammonia is converted to nitric acid in 2 stages.
Which catalyst is used in Deacon process?
In 1874, Henry Deacon had derived a process to reduce HCl emissions as mandated by the Alkali Act. In this process, hydrogen chloride is oxidized by oxygen over a copper chloride catalyst, resulting in the production of chlorine.What is platinum catalyst?
Platinum catalyst is the best-known precious metal catalyst. It is active against almost all of chemical reactions such as hydrogenration, dehydrogenation and oxidation. Even in reactions which are not active with other catalysts, platinum catalyst is often active.
Why is platinum used as a catalyst in nitric acid?In large-scale nitric acid manufacturing, knitted platinum-rhodium gauze is necessary to catalyze chemical reactions for maximum efficiency. … This oxidation reaction creates nitric oxide. In order to convert as much of the ammonia as possible, the process is carried out under pressure with a platinum-rhodium catalyst.
Article first time published onWhat is a catalyst in science?
A catalyst is a substance that speeds up a chemical reaction, or lowers the temperature or pressure needed to start one, without itself being consumed during the reaction. Catalysis is the process of adding a catalyst to facilitate a reaction.
What is Ostwald method of nitric acid?
Ostwald’s process is a chemical process to convert ammonia into nitric acid in two stages. In stage1 ammonia is oxidized to form nitric oxide and nitric dioxide and in the second step the nitrogen dioxide is allowed to dissolve in water and finally nitric acid is formed.
What is gauge catalyst?
it is a type of catalyst, contains platinum and rhodium, used in the Ostwald’s process of manufacture of nitric acid – HNO3.
What is observed during catalytic oxidation of ammonia?
▶The colorless ,pungent smelling gas nitric oxide emits which on further oxidation gives a reddish brown gas which is nitrogen dioxide. ▶Platinum continues to glow even when heating is discontinued as the reaction done is an exothermic reaction. ▶Water droplets are seen at the inner surfaces of glass tube.
Which of the following is obtained by catalytic oxidation of ammonia?
Nitric oxide (NO) is obtained by catalytic oxidation of ammonia.
What catalyst is used in making nitric acid?
In order to achieve a high conversion efficiency, this is normally carried out at pressure over a platinum-rhodium catalyst. The nitric oxide is cooled and further oxidised to form nitrogen dioxide, which is then absorbed in water to nitric acid.
What is catalytic hydrogenation reaction?
Catalytic hydrogenation is treatment with hydrogen in the presence of a catalyst such as nickel, palladium or platinum. Catalysts are required for the reaction to be usable; non-catalytic hydrogenation takes place only at very high temperatures. Hydrogenation reduces double and triple bonds in hydrocarbons.
Why are catalysts heterogeneous?
Heterogeneous catalysis is very important because it enables faster, large-scale production and the selective product formation. Approximately 35% of the world’s GDP is influenced by catalysis. The production of 90% of chemicals (by volume) is assisted by solid catalysts.
Why is nickel a good catalyst?
Nickel-based catalysts are the most frequently used in reforming reactions due to C–C bond rupture capability. Nickel has been generally supported onto alumina because of its ability to withstand reaction conditions. … To avoid these inconveniences, different promoters have been added to nickel-based catalysts.
What is platinum rhodium catalyst?
Rhodium-platinum oxide (Rh–Pt oxide), or Nishimura’s catalyst, is an inorganic compound used as a hydrogenation catalyst. …
What is platinum catalytic converter worth?
The price of the platinum metal varies, but there are websites that can give you accurate pricing. However, the average price, according to Business Insider, is $1097 per troy ounce.
Which of the following catalyst is used in manufacture of H2SO4 in contact process?
V2O5 (vanadium pentaoxide) is used as a catalyst in the manufacture of H2SO4 by contact process since it is not easily poisoned.
How is chlorine prepared by Decans process?
In 1868, Deacon developed a process by which chlorine is produced by oxidation of gaseous HCl with O2 in the presence of a Cucl2catalyst. … HCl(g)+14O2Cucl2,723K→12H2O(g)+12Cl2(g). This an overall exothermic process and also fast.
How is chlorine prepared by electrolytic method?
Chlorine can be manufactured by the electrolysis of a sodium chloride solution (brine), which is known as the Chloralkali process. The production of chlorine results in the co-products caustic soda (sodium hydroxide, NaOH) and hydrogen gas (H 2 ). Chlorine is obtained as a byproduct during manufacture of NaOH.
Why are metals used as catalysts?
Transition metals are good metal catalysts because they easily lend and take electrons from other molecules. A catalyst is a chemical substance that, when added to a chemical reaction, does not affect the thermodynamics of a reaction but increases the rate of reaction.
What is the chemical formula of platinum?
PubChem CID23939Chemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaPtSynonyms7440-06-4 Platinum Pt Platinum Black Platin More…Molecular Weight195.08
What is catalyst answer?
catalyst, in chemistry, any substance that increases the rate of a reaction without itself being consumed.
What is catalyst with example?
A catalyst is substance i.e a element or a compound that increases the rate of chemical reaction. Examples: 1) Nickel, Ni is used in hydrogenation of palm oil into margarine. 2) Iron, Fe is used in Haber process. ( Manufacturing of ammonia)
What is meant by catalyst and catalysis?
A catalyst is a substance that initiates or accelerates the rate of a particular chemical reaction without itself being chemically affected. … The process or action by which a catalyst increases the reaction rate is called catalysis.
Which catalyst is used in the Haber process for the manufacture of nh3?
The catalyst used in Haber’s process is Fe. It increases the rate of reaction between nitrogen and hydrogen.
How is nitric acid prepared by catalytic oxidation of ammonia?
When ammonia reacts with oxygen in presence of platinum catalyst it forms nitrogen oxide. In the second step, the nitrogen oxide gas converts into nitrogen dioxide, and this nitrogen dioxide gas reacts with water to form nitric acid.