What is the charge of a copper atom

Sarah Rodriguez
Published Feb 18, 2026
Copper (I) ions have a 1+ charge. This happens when copper atoms lose one electron. Its formula is Cu+ .
Why does copper have a 2+ charge?
Because the charge field density of the copper atom’s electron cloud moves a 4s into 3d. In zinc it’ll add a second electron to 4s. Welcome to chemistry. Thus, copper certainly does have 2 valence electrons it can lose, giving it an oxidized state of 2+.
Why the valency of copper is 1 or 2?
Copper has valency ‘1‘ and ‘2’ that is the number of electrons lost or gained is 1 or 2 in different compounds.. For CuCl the valency is one as it needs one chlorine atom for this compound, hence its name is cuprous chloride.
Is a copper atom positive or negative?
Copper atoms in a metal have no charge, but copper ions in solution as copper chloride have a positive charge.Is copper a insulator?
Insulators oppose electrical current and make poor conductors. Some common conductors are copper, aluminum, gold, and silver. Some common insulators are glass, air, plastic, rubber, and wood. Insulators that can be polarized by an electric field are called dielectrics.
How do you make copper positively charged?
Copper-silver ionization is brought about by electrolysis. An electric current is created through copper-silver, causing positively charged copper and silver ions to form.
Why is copper positive and zinc negative?
Zinc is closer to the halogens than copper, but copper is slightly more electronegative than zinc. To be filled, the 4s subshell of copper requires one more electron. That is a simple explanation of why copper is more electronegative than zinc.
What is copper valency?
ElementAtomic NumberValencyIron262,3,4,6Cobalt272,3,4Nickel283,2Copper292,1How do you find Cu valency?
Answer: The Valency of copper like other transition metals is not immediately obvious. For example if it is Copper(I)chloride, CuCl, oxidation state is +1 and the solid is green and little soluble in water. … Thus the generally shown valency of copper atom is either +1 or +2.
Does copper have 1 or 2 valence electrons?Copper (Cu) has two valences Cu I (cuprous) has one valence electron and Cu II (cupric) has two valence electrons.
Article first time published onIs gold a conductor?
Gold is used as a contact metal in the electronics industry as it is a good conductor of both electricity and heat. … Gold wire Gold is ductile: it can be drawn out into the thinnest wire. © AMNH / Craig Chesek. Gold conducts heat and electricity.
Can a human body conduct electricity?
Electricity is everywhere, even in the human body. Our cells are specialized to conduct electrical currents. … This is due to a slight imbalance between positive and negative ions inside and outside the cell. Cells can achieve this charge separation by allowing charged ions to flow in and out through the membrane.
Is pencil A conductor or insulator?
Conductor. It a mixture of graphite and clay. It is a pretty good conductor. The harder the pencil lead like 6H – the worse a conductor/higher vresistance it will have.
Why is copper a cathode?
Current flows. Cu is deposited as a metal solid on the electrode surface, whatever it is made out of. Copper metal is, as it always is, conductive. As a result of steps 2 and 3, there is now a copper metal cathode instead of a cathode made of another material.
Why is electricity called juice?
Since the 1600s, juice has been slang for sexual fluids and alcohol. By the early 1900s, juice was slang for electrical power. By 1924, juice specifically referred to battery power.
Why is E value of copper positive?
The E° (M2+/M) value for copper is positive (+ 0.34 V). … Copper has high atomisation ΔaH° and low hydration energy Δhyd H°. Due to which the E° value is positive.
Is copper a cathode or anode?
Zinc behaves as the anode (supplying electrons) of the galvanic cell and the copper as the cathode (consuming electrons).
Why is copper the anode?
The high purity is needed because most copper is used to make electrical equipment, and small amounts of impurity metals in copper can seriously reduce its ability to conduct electricity. … To purify copper electrolytically, the impure copper metal is made the anode (the positive electrode) in an electrolytic cell.
How do you make copper anode?
A copper anode is made from pulverizing a copper ore called chalcopyrite, CuFeS2, and mixing it with water, pine oil and amyl xanthate, a salt used to make the mixture water-repelling. After the mixture is made, compressed air is passed through it to allow further purification.
What color is CuCl?
Copper(I) chlorideOther namesCuprous chlorideMolecular formulaCuClMolar mass98.99 g/molAppearancewhite powder, slightly green from oxidation
What is valency of Cu in CuCl2?
Being a transition metal, copper exhibits variable valencies of 1 and 2. Copper forms two types of chlorides – cuprous chloride, Cu2Cl2, and cupric chloride, CuCl2. In the Cu2Cl2, the metal and chlorine atoms are 1:1 ratio and in CuCl2 the atomic ratio is 1:2. Therefore, the valencies of copper are 1 and 2.
What is Silver's valency?
The valency of the element represents the number of bonds that an atom can form as part of a compound. The most common valency of silver metal is 1 and 3.
What is copper formula?
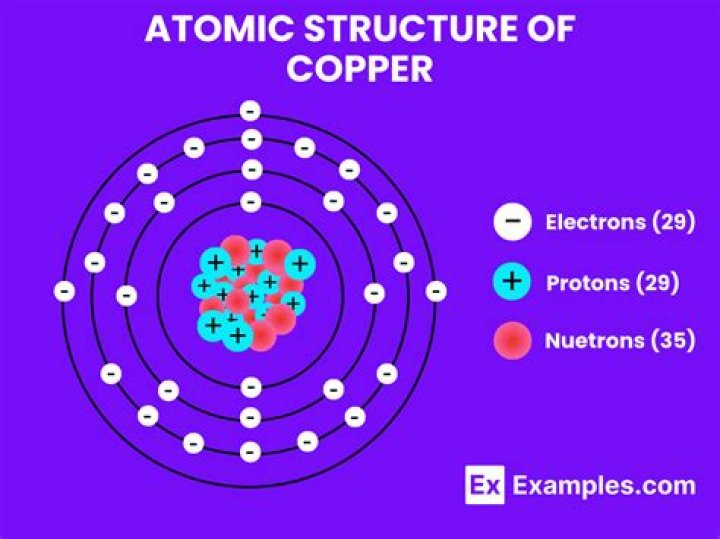
What is Copper? … In Chemistry, the copper chemical formula is denoted as Cu. as copper belongs to the transition metal group in the periodic table that has a d subshell and is denoted as (n – 1) d transition. It has an atomic number 29, thus the copper chemical formula is alternatively written as Cu29 in general.
What is valency class 9th?
The combining capacity of an atom of an element to form chemical bond is called its valency. The valency of an element is. (1) Equal to the number of valence electrons. (2) Equal to the number of electrons required to complete eight electrons in valence shell.
Can copper have a charge of 1?
Explanation: Copper (I) ions have a 1+ charge. This happens when copper atoms lose one electron. … This happpens when copper atoms lose two electrons.
What is the neutrons of copper?
In natural copper, the atoms are of two kinds. One has 29 protons and 34 neutrons in the nucleus; the other has 29 protons and 36 neutrons (Figure 4). The two different kinds of atom are called isotopes of copper.
Does copper have a full valence shell?
Yes, copper only has 1 valence electron. Remember: valence electrons only include the electrons in the highest energy (n) shell.
What can break gold?
‘Aqua Regia’ is a mix of hydrochloric acid and nitric acid, most commonly in a 3:1 ratio. This is believed to be the only solution known that can “dissolve” gold.
Who discovered gold?
Gold! On January 24, 1848, James W. Marshall discovered gold on the property of Johann A. Sutter near Coloma, California.
Can gold be scratched?
Scratching of gold is always a concern for many, particularly when the gold jewellery has more pure gold than other metal alloys. If you wear your gold jewellery pieces daily, like gold rings, gold chains, gold bangles or gold earrings; there is a high chance of them getting scratched due to day-to-day work.
Can the human body power a light bulb?
Each neuron generates a small amount of electricity. When we add all these neurons together, the electricity generated in the brain can power a small light bulb. That’s about 10-25 Watts of power. When you think about it, it’s really impressive.