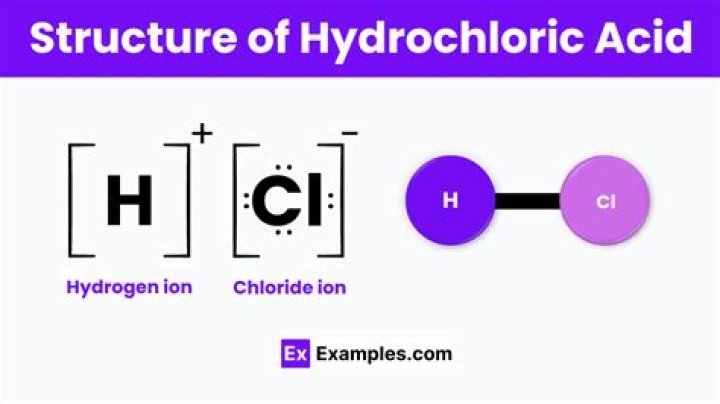
What is the chemical equation of hydrochloric acid

Andrew Campbell
Published Mar 02, 2026
NamesChemical formulaHCl(aq)AppearanceColorless, transparent liquid, fumes in air if concentratedOdorPungent characteristic
What is the chemical equation for hydrochloric acid?
NamesChemical formulaHCl(aq)AppearanceColorless, transparent liquid, fumes in air if concentratedOdorPungent characteristic
Can I drink hypochlorous acid?
One solution is a sanitizer that at 50 parts per million is more effective than chlorine bleach at 200 parts per million, yet it’s so non-toxic you can drink it.
What is the chemical equation of sodium hydrochloric acid?
Sodium hydroxide and hydrochloric acid react as shown in this equation. NaOH(aq)+HCl(aq)→NaCl(aq)+H2O(l)Is it HOCl or HClO?
Hypochlorous acid (HOCl or HClO) is a weak acid that forms when chlorine dissolves in water, and itself partially dissociates, forming hypochlorite, ClO−. HClO and ClO− are oxidizers, and the primary disinfection agents of chlorine solutions.
How do you find the formula for an acid?
- H2SO4 = Sulfuric acid.
- HNO3 = Nitric Acid.
- HC2H3O2 = acetic acid.
What is the chemical formula of acid?
Acid NameFormulahydrochloric acidHClnitric acidHNO3phosphoric acidH3PO4sulfuric acidH2SO4
What type of reaction is Na2CO3 and HCl?
When solutions of sodium carbonate and hydrochloric acid are mixed, the equation for the hypothetical double displacement reaction is: Na2CO3 + 2 HCl → 2 NaCl + H2CO3 Bubbles of a colorless gas are evolved when these solutions are mixed.What is NaOH HCl?
HCl + NaOH = NaCl + H₂O + Q. Hydrochloric acid and sodium hydroxide interact, resulting in salt and a release of heat. As we can see, the result of the interaction of two highly aggressive compounds is table salt and water – compounds that are ultimately harmless, even beneficial, to human beings.
What is the formula for a chlorine molecule?Formula and structure: the chemical structure of chlorine gas is Cl2 and its molecular weight is 70 g/mol. Its structure is Cl-Cl, which is also called the element form of the chlorine element, consist in 2 atoms of chlorine joined by a covalent bond.
Article first time published onWhy is HCl an acid?
HCl is a strong acid because it dissociates almost completely. By contrast, a weak acid like acetic acid (CH3COOH) does not dissociate well in water – many H+ ions remain bound-up within the molecule. In summary: the stronger the acid the more free H+ ions are released into solution.
What does hypochlorous acid smell like?
Killing germs Yet bleach is highly irritating to the eyes, skin, and lungs-and inhalation over long periods could be carcinogenic. In contrast, HOCl has a temporary and mild chlorine smell that dissipates quickly, it is non-irritating, and it does not have poisonous side effects.
What is hypochlorous spray?
Ways to use hypochlorous acid It’s used as a disinfectant and as a irrigant in surgery—meaning, as a solution to clean cuts, burns, or open wounds. Because of its Covid-19 disinfecting properties, it is being used by health care workers, especially when working around the mouth and nose, says Dr. Madfes.
Can you use hypochlorous acid on food?
Hypochlorous acid is usually used to sanitize food products such as vegetables, fruits, and meat. This acid is widely used in the food industry as the disinfection agents of food processing equipment. However, hypochlorous acid readily reacts with proteins to form an aggregate and oxidizes specific amino acids.
How do you find the name for HC2H3O2?
The OOH, represents one oxygen double bonded to the same carbon where the OH is attached. This arrangement is indicated in nomenclature as — oic acid. Combine the two parts to give the name ethanoic acid. the chemical name of HC2H3O2 is Ethanoic Acid(acetic acid) commonly called vinegar.
Is HClO ionic or covalent?
For hypochlorous acid (HClO), we have: O = -2; H = +1; and therefore Cl = +1. Hypochlorous acid, covalent bond (dative). The chlorine present in hypochlorous acid has electronic doublets.
How do you write chemical formulas?
- In a chemical equation, the reactants are written on the left, and the products are written on the right.
- The coefficients next to the symbols of entities indicate the number of moles of a substance produced or used in the chemical reaction.
What are the formulas for hydrochloric acid and nitric acid?
CompoundFormulaConc. Reagent MolarityHydrochloric acidHCl11.6Nitric acidHNO316.0Perchloric acidHClO411.6Phosphoric acidH3PO418.1
How do you name chemical formulas?
The first element in the formula is simply listed using the name of the element. The second element is named by taking the stem of the element name and adding the suffix -ide. A system of numerical prefixes is used to specify the number of atoms in a molecule.
What is the chemical formula of acetic acid?
Acetic acid , systematically named ethanoic acid , is an acidic, colourless liquid and organic compound with the chemical formula CH3COOH (also written as CH3CO2H, C2H4O2, or HC2H3O2).
What is nh3 HCl?
This is an acid-base reaction (neutralization): NH 3 is a base, HCl is an acid. NH 3.
What is NaCl name?
Sodium chloride , commonly known as salt (although sea salt also contains other chemical salts), is an ionic compound with the chemical formula NaCl, representing a 1:1 ratio of sodium and chloride ions.
What happens when HCl is added to baking soda?
Combining an acid with a base typically results in water and salt. … Mixing baking soda, or NaHCO3, with hydrochloric acid, or HCl, results in table salt, NaCl, as well as water, H2O, and carbon dioxide, CO2.
What happens when HCl reacts with NaHCO3?
A saturated solution of sodium bicarbonate is reacted with hydrochloric acid to form carbon dioxide gas.