What is the electronic geometry around the nitrogen in ammonia nh3

Dylan Hughes
Published Feb 14, 2026
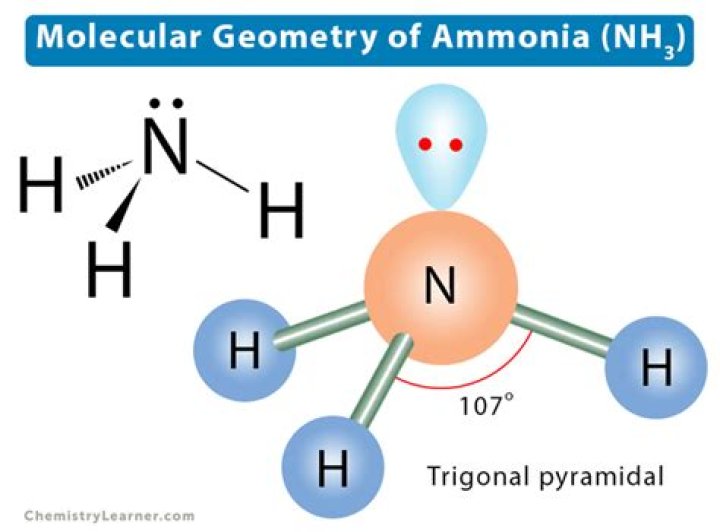
Ammonia also has four electron pairs and the coordination geometry of nitrogen is based upon a tetrahedral arrangement of electron pairs. There are just three bonded groups, therefore there is one lone pair. However since the lone pairs are ‘invisible’, the shape of ammonia is pyramidal. The geometry of ammonia, NH3.
What is the electronic geometry of nitrogen?
In the given molecule, the nitrogen atom has three atoms around it (1 H and 2 C atoms) and one lone pair. The total number of electron pairs around the nitrogen atom is 4 (AX4). The electron pair geometry of the nitrogen atom is tetrahedral.
What molecular shape does ammonia have?
The ammonia molecule has a trigonal pyramidal shape with the three hydrogen atoms and an unshared pair of electrons attached to the nitrogen atom.
What is the electronic configuration of NH3?
The atomic number of the nitrogen is seven, which makes its electronic configuration 1s2 2s2 2p3. As the p shell needs to accommodate a maximum of six electrons, there is a scarcity of three electrons. It makes a single nitrogen atom to have five valence electrons.How many electron pairs are in N2?
After determining the center atom and sketch of N2 molecule, we can start to mark lone pairs on atoms. Remember that, there are total of five electron pairs. There is already one bond in the drawn sketch.
Why does ammonia have a tetrahedral electron geometry but a trigonal pyramidal molecular geometry?
Three of these electron pairs are used as bond pairs which leaves one lone pair of electrons. The lone pair repel more strongly than bond pairs giving tetrahedral arrangement. Since the lone pairs are invincible the shape of ammonia is trigonal pyramidal.
How does the electronic configuration of nitrogen affect the formation structure and shape of ammonia?
Ammonia, NH Each of the 3 hydrogens is adding another electron to the nitrogen’s outer level, making a total of 8 electrons in 4 pairs. Because the nitrogen is only forming 3 bonds, one of the pairs must be a lone pair. The electron pairs arrange themselves in a tetrahedral fashion as in methane.
Is N2 dinitrogen or nitrogen?
ChEBI NamedinitrogenDefinitionAn elemental molecule consisting of two trivalently-bonded nitrogen atoms.Which structure best shows the bond angle and molecular shape of ammonia NH3?
NH3 Ammonia The resulting molecular shape is trigonal pyramidal with H-N-H angles of 106.7°.
Why nitrogen molecule is linear in shape?Steric number: It is the summation number of atoms bonded to the central atom via single bond and the number of lone pairs of electrons present on the atom. So, the molecular geometry of the molecule is linear. As there is appreciable distance between the lone pair of electrons on each atom.
Article first time published onWhat is electron domain geometry?
Electron domain geometry – The arrangement of electron domains surrounding the central atom of a molecule or ion. … Bond angles (BA) – The angle between two adjacent bonds in the same atom. The bond angles are affected by all electron domains, but they only describe the angle between bonding electrons.
Why has ammonia got trigonal pyramidal geometry though nitrogen shows sp3 hybridization?
It is trigonal pyramidal because of the lone pair of electrons associated with the central nitrogen atom.
What is the molecular geometry of bef2bef2?
The molecular geometry of BeF2 , i.e., beryllium difluoride, is linear. This molecule has a beryllium atom located between two fluorine…
Why is NH3 a trigonal pyramidal?
For example; four electron pairs are distributed in a tetrahedral shape. If these are all bond pairs the molecular geometry is tetrahedral (e.g. CH4). If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3).
Which type of hybridization is present in ammonia molecule write geometry and bond angle in it?
Name of the MoleculeAmmoniaMolecular FormulaNH3Hybridization Typesp3Bond Angle107oGeometryPyramidal or Distorted Tetrahedral
What is the shape and bond angle of ammonia?
In the case of ammonia there are 4 charge clouds (3 bonding pairs and 1 lone pair) so the basic shape of ammonia is tetrahedral and the bond angle is 109.5 degrees.
What is the shape of H2O NH3 draw the geometry?
Answer: H2O has v shaped or bent structure. ..it is doue to presence of 2 lone pairs on it that it do not have a straight structure with 180 degree angle. NH3 has pyramidal structure . it’s 3 hydrogen atoms are at 3 base vertices of pyramid while a lone pair forms the apex of it.
Why is nitrogen written as N2?
It forms diatomic molecules, which means that there are two nitrogen atoms per molecule in nitrogen gas (N2). In this configuration nitrogen is very inert, meaning that it doesn’t typically react with other compounds. Nitrogen becomes a liquid at -210.00 degrees C.
What type of compound is nitrogen?
Nitrogen forms many thousands of organic compounds. Most of the known varieties may be regarded as derived from ammonia, hydrogen cyanide, cyanogen, and nitrous or nitric acid. The amines, amino acids, and amides, for example, are derived from or closely related to ammonia.
What Colour is nitrogen?
ElementColor NameRGB ValuesNitrogenlight blue[143,143,255]Sulfuryellow[255,200,50]Phosphorusorange[255,165,0]Chlorinegreen[0,255,0]
How are the electrons arranged around the nucleus of nitrogen?
Nitrogen is the seventh element with a total of 7 electrons. In writing the electron configuration for nitrogen the first two electrons will go in the 1s orbital. Since 1s can only hold two electrons the next 2 electrons for N goes in the 2s orbital. The remaining three electrons will go in the 2p orbital.
What is the molecular geometry of nitrogen in the following molecule?
Therefore, the molecular geometry for this molecule will be linear, with a bond angle of about 180∘ .
What is the hybridization electronic arrangement and molecular geometry of the nitrogen atom 2 in the following molecule?
Since nitrogen has one lone pair, the molecular geometry around the indicated nitrogen atom is bent. For nitrogen, the hybridization is sp2 and the geometry is bent.
How do you find the molecular geometry?
- Draw the Lewis Structure.
- Count the number of electron groups and identify them as bond pairs of electron groups or lone pairs of electrons. …
- Name the electron-group geometry. …
- Looking at the positions of other atomic nuclei around the central determine the molecular geometry.
How many electron domains does nh3?
The University of SheffieldDepartment of ChemistryVSEPR
How do you find the electron domain in geometry?
Count the total number of electron domains. Use the angular arrangement of the chemical bonds between the atoms to determine the molecular geometry. Keep in mind, multiple bonds (i.e., double bonds, triple bonds) count as one electron domain. In other words, a double bond is one domain, not two.
Why is the geometry of ammonia distorted?
Among them, three take point in covalent bond formation with three hydrogen atoms and the other two remain unshared as lone pairs. Lone pairs can’t decide the shape of molecules. So, according to VSEPR theory, the shape of the ammonia molecule is distorted tetrahedral as there is no same type of four bonds.
Why geometry of water is V shaped and that of ammonia is pyramidal?
NH3 has pyramidal shape and H2O hasbent shape. … O in H2O is the central atom of the molecule and have 2 lone pairs. Due to the presence of 2 lone pairs the lone pair-lone pair repulsion occurs due to which the bond angle between the H atoms decreases. Thus bond angle in NH3 is greater than bond angle in H2O.
How would you explain the shape of ammonia molecule on the basis of hybridisation?
- Explanation:Hybridization is a processs of mixing and recasting of atomic orbitals having same energies and forming new orbitals all having same energy, shape.
- in ammonia (NH3)
- ground state / =N=7 = 1s^2 2s^2 2p^3.
- Three new hybridired orbitals will be formed having sp^3 hybridization.
What is the electron pair geometry around the central atom in h2o?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.
What is the electron pair geometry for be in bebr2?
BeBr2 – Beryllium Dibromide: The molecular geometry of BeBr2 is linear with symmetric charge distribution on the central atom.



