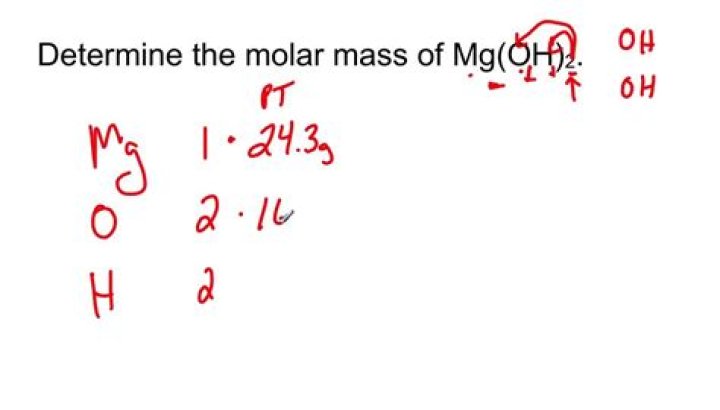
What is the formula weight of Mg OH 2

Andrew White
Published Mar 10, 2026
The molar mass of Mg(OH)2 is 58.33 g/mol. The molar mass of magnesium is 24.31 g/mol. The molar mass of oxygen is 16.00 g/mol, but since there are…
What is the correct formula weight of Mg OH 2?
The molar mass of Mg(OH)2 is 58.33 g/mol. The molar mass of magnesium is 24.31 g/mol. The molar mass of oxygen is 16.00 g/mol, but since there are…
What is the formula weight of magnesium?
The relative masses of each element can be found on the periodic table. For example, one atom of magnesium weighs 24.31 amu (atomic mass units). However, one mole of magnesium weighs 24.31 g.
What is the formula of Mg and oh?
Formula and structure: The chemical formula of magnesium hydroxide is Mg(OH)2, and its molar mass is 58.32 g/mol. The chemical structure is shown below, where the central Mg is bonded to the two hydroxyl groups. The compound is ionic, existing mainly as Mg2+ and OH- ions.What is the percent of Mg OH 2?
The composition of Mg(OH)2 is 41.67 % Mg, 54.87 % O, 3.457 % H .
Is Mg OH 2 a strong or weak base?
Because Mg(OH)2 is listed in Table 15.6. 1, it is a strong base.
What is the use of Mg OH 2?
Uses of Magnesium hydroxide (Mg(OH)2) Magnesium hydroxide is an excellent thermal conductor and poor electrical conductor. It is used in suspensions as a laxative or antacid. Used as a food additive. It is widely used in waste-water treatment.
What is the shape of co NH2 2?
Urea (CO(NH2)2) is a solid product in the shape of pearls or grains; its main characteristic is N in the form of an amide (NH2). The compound is commercially synthesized by a reaction of ammonia (NH3) and carbon dioxide (CO2) under conditions that are dependent on the technology employed in the industrial plant.What is the name of co NH2 2?
UREA [CO(NH2)2]
What is the molecular mass of NH2 co NH2?Property NameProperty ValueReferenceMonoisotopic Mass60.032362755Computed by PubChem 2.1 (PubChem release 2021.05.07)
Article first time published onWhat is the formula equation for magnesium?
StepResult1magnesium + oxygen → magnesium oxide2Mg + O 2 → MgO3reactants: 1 × Mg, 2 × O products: 1 × Mg, 1 × O Not balanced.4Mg + O 2 → 2MgO
How do you find the molecular weight of magnesium?
The molar mass of an element can be described as atomic weight (relative atomic mass) on the periodic table in g/mol. Atomic weight of magnesium is 24.305. Hence molar mass of Magnesium is24. 305 g/mol.
What is the percent by mass of Mg in MgO?
The percent composition of magnesium in magnesium oxide is 39.70 %.
What is the percent composition of magnesium Mg in MgO apex?
The percent composition of magnesium in magnesium oxide with a chemical formula of MgO is 60.30 percent.
Is Mg OH 2 ionic or covalent?
When TWO hydroxide anions find a magnesium dication, they get together and form TWO ionic bonds, between the magnesium dication and the two hydroxide anions. Thereby they form Mg(OH)2, which is an ionic compound.
Can you take magnesium hydroxide daily?
Do not use magnesium hydroxide for longer than 7 days without medical advice.
What are the side effects of magnesium hydroxide?
- severe nausea, vomiting, or diarrhea;
- no bowel movement after using the medicine as a laxative;
- rectal bleeding; or.
- worsening symptoms.
Is magnesium hydroxide bad?
Extended use or overuse of this medication for constipation may result in dependence on laxatives and ongoing constipation. Overuse may also cause persistent diarrhea, loss of too much body water (dehydration), and mineral imbalances (e.g., high magnesium). Tell your doctor if your condition persists or worsens.
Is Mg OH 2 acidic basic or neutral?
Because Mg(OH) 2 is listed in Table 12.2 “Strong Acids and Bases”, it is a strong base.
What is the nature of Mg OH 2?
Magnesium hydroxide is the inorganic compound with the chemical formula Mg(OH)2. It occurs in nature as the mineral brucite. It is a white solid with low solubility in water (Ksp = 5.61×10−12). Magnesium hydroxide is a common component of antacids, such as milk of magnesia.
Why is Mg OH 2 considered a strong base?
Mg(OH)2 is the common component of antacid i.e.it acts as a base but its solubility is very low. Due to the low solubility it is a weak base. But it is completely ionized in aqueous solution. Therefore, it is considered as a strong base.
Is OH and HO the same?
HO is a molecule containing positive Hydrogen Oxygen atoms i.e it is a cation. … OH is a molecule containing negative Hydrogen Oxygen atoms i.e it is an anion. Both are ions, HO+ is Hydroxide, OH- is Oxyhydride. Both are acceptable and the only difference is about the charge on oxygen.
How is hydroxide made?
Hydroxide is made of hydrogen and oxygen. … The hydroxide ion is a negatively charged molecule made up of one oxygen bonded to one hydrogen. When dissolved in water, the hydroxide ion is an incredibly strong base.
Is co NH2 2 an electrolyte?
What mass of urea, CO(NH2)2, a non-volatile non-electrolyte, must be dissolved in 250.0g of water in order to lower its vapor pressure by 10.0%?
What is the structure of NH2?
If we look at the Lewis Structure of NH2- ion, we can see that we have four electron-rich regions around our central nitrogen atom. There are two bonded electron pairs around N to form single covalent bonds each with a Hydrogen atom on either side.
What is the molecular mass of MG 3 and 2?
PubChem CID3673438StructureFind Similar StructuresMolecular FormulaMg3N2Molecular Weight100.93DatesModify 2021-11-20 Create 2005-09-10
How do you calculate molecular mass?
Molecular Mass Problem Find the atomic mass for each element by using the mass given in the Periodic Table. Multiply the subscript (number of atoms) times the atomic mass of that element and add the masses of all of the elements in the molecule to get the molecular mass.
What is the coefficient for MG?
So, to balance a chemical equation we make the number of each element the same on both sides. Thus the equation is balanced and the coefficient of Mg is 1, which is understood so it is not written in the final equation.
What type of reaction is 2Mg O2 → 2MgO?
2Mg+O2=2MgO is a type Of Combination reaction.
Why is magnesium oxide heavier than magnesium?
The increased mass is because of oxygen. Magnesium oxide has the formula of mgo according to the ratio between the moles of magnesium and oxygen in the final product.