What is the IRB responsible for

Nathan Sanders
Published Mar 19, 2026
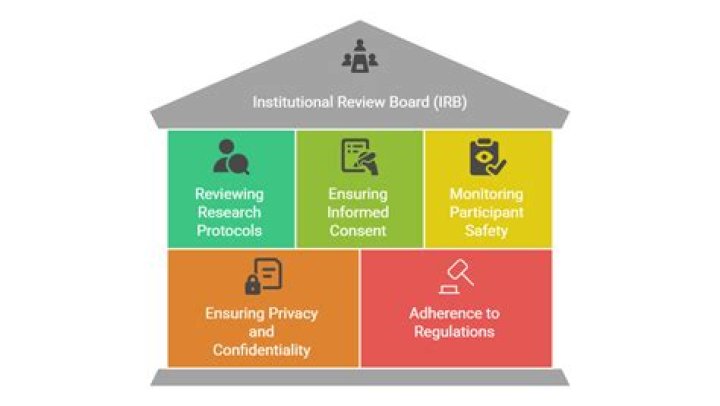
Under FDA regulations, an Institutional Review Board is group that has been formally designated to review and monitor biomedical research involving human subjects. In accordance with FDA regulations, an IRB has the authority to approve, require modifications in (to secure approval), or disapprove research.
What is an IRB and why is it important?
The IRB serves as an objective third party, an oversight committee, governed by Federal Regulations with the purpose of protecting and managing risk to human participants involved in research.
What are the primary responsibilities and priorities of the IRB?
The IRB is concerned with protecting the welfare, rights, and privacy of human subjects. The IRB has the authority to approve, disapprove, monitor, and require modifications in all research activities that fall within its jurisdiction as specified by both the federal regulations and institutional policy.
What is the major responsibility of an institutional review board quizlet?
Institutional review boards (IRBs) are tasked with reviewing all studies involving human subjects to protect their rights and welfare.How does IRB handle ethical cases?
The purpose of the IRB is to ensure that the investigator complies with the protocol and to demonstrate that the trial is necessary and that the risk-benefit ratio is acceptable by reviewing key trial documents to ensure that the subjects’ rights and well-being are protected.
Which of the following studies would need IRB approval *?
Which of the following studies need IRB approval? Studies collecting data about living individuals.
What is the IRB and what purpose does it serve quizlet?
Institutional review board (IRB) definition: … The Institutional Review Board (IRB) means any board, committee, or other group formally designated by an institution to review, to approve the initiation of, and to conduct periodic review of, biomedical research involving human subjects.
What is the purpose of a research review committee quizlet?
-a committee that has been formally designated to approve, monitor, and review biomedical and behavioral research involving humans with the aim to protect the rights and welfare of the research subjects.What information should be provided to an IRB for review at the initiation of a study?
The study protocol (and amendments), the information to be given to the subject (informed consent, advertisements), the Investigator Brochure (or drug label), any other relevant safety information, and an outline of the qualifications of the investigator.
What are the 4 important ethical issues IRB guidelines address?- Respect for persons: respect for patient autonomy.
- Beneficence: maximize benefits and minimize harm.
- Justice: Equitable distribution of research burdens and benefits.
What are IRB protocols?
Under FDA regulations, an Institutional Review Board is group that has been formally designated to review and monitor biomedical research involving human subjects. In accordance with FDA regulations, an IRB has the authority to approve, require modifications in (to secure approval), or disapprove research.
What is justice in the Belmont Report?
Justice: This principle advocates fair treatment for all and a fair distribution of the risks and benefits of the research. It forbids exploitation of vulnerable people (for instance, economically disadvantaged or those with limited cognitive capacity) or those who are easily manipulated as a result of their situation.
Who serves on an IRB?
Who Serves on an IRB? An IRB consists of at least five members of varying backgrounds. IRB members should have the professional experience to provide appropriate scientific and ethical review. An IRB must have at least one scientist member and at least one member whose primary concerns are nonscientific.
What is IRB approval in research?
What is an Institutional Review Board (IRB)? … In accordance with FDA regulations, an IRB has the authority to approve, require modifications in (to secure approval), or disapprove research. This group review serves an important role in the protection of the rights and welfare of human research subjects.
What happens if you do research without IRB approval?
Under 45 CFR 46.103, the IRB must review and approve all non-exempt human subject research. Failure to obtain IRB approval before conducting research could result in the researcher being unable to use data and other information collected through the research process.
What does IRB stand for?
IRB stands for “Institutional Review Board”. The IRB is a committee established to review and approve applications for research projects involving human subjects. The primary purpose of the IRB is to protect the rights and welfare of the human subjects.
What purpose does the IRB review the informed consent?
The primary function or role of the IRB is to safeguard human subjects by training researchers in research ethics and best practices and reviewing research proposals. In reviewing a research protocol the IRB must balance the research’s risk to the human subjects with the benefits to society.
Which statement best describes what an IRB is responsible for reviewing?
Terms in this set (7) – FDA states that IRB has the authority to approve, require modifications in (to secure approval), or disapprove research. This group review serves an important role in the protection of the rights and welfare of human research subjects.
Which IRB should be used for behavioral research?
The Behavioral/NonMedical IRB (IRB02) is responsible for reviewing and monitoring a subset of the research with human subjects conducted at the University of Florida.
What is an IRB quizlet?
An IRB consists of a group of scholars from various departments who are mandated to review proposals to ensure that they comply with ethical guidelines and write a summary of proposed risks to subjects. … You just studied 2 terms!
Why might a researcher debrief his participants?
You submit a study for approval by the institutional review board (IRB), and they tell you that written informed consent is required. … Why might a researcher debrief his participants even if his study didn’t include any deceptive elements? To ensure that his participants had a good research experience. Dr.
Why do researchers need to consider the philosophical worldview assumptions they bring to their research?
Because worldviews shape approaches to research, the questions asked and the methods used. Why do researchers need to consider the philosophical worldview assumptions they bring to their research? … To ensure your proposed research is ethical and protects human subjects or animals.
What are the 3 basic principles of the Belmont Report?
Three basic principles, among those generally accepted in our cultural tradition, are particularly relevant to the ethics of research involving human subjects: the principles of respect of persons, beneficence and justice.
How does the IRB protect human subjects?
At UNH, the primary purpose of the Institutional Review Board for the Protection of Human Subjects in Research (IRB) is to protect the rights and welfare of human research subjects by ensuring that physical, psychological, legal, and/or social risks to subjects are minimized, and when present, justified by the …
What does the IRB do to protect a research participant from harm?
The IRB is a primary mechanism for institutional protection of human subjects. … Under both DHHS and FDA regulations, the IRB has responsibility to approve or disapprove all covered research activity, requiring for instance that subjects are given enough information to be able to provide informed consent.
What are the consequences of violating human subjects responsibilities as dictated by the IRB?
If human subjects protection regulations are willfully violated, the department secretary or agency head may bar the organization or individual from receiving funding from any federal source. [39] Such debarment must be for a specified length of time and, in some extreme cases, may be permanent.
What are the two ethical convictions of Belmont Report?
The Belmont Report states that “respect for persons incorporates at least two ethical convictions: first, that individuals should be treated as autonomous agents, and second, that persons with diminished autonomy are entitled to protection.
Who does the Belmont Report protect?
Its primary purpose is to protect subjects and participants in clinical trials or research studies. This report consists of 3 principles: beneficence, justice, and respect for persons. This article reviews the Belmont Report and these 3 principles as well as its importance to nurse researchers.
What role did the Tuskegee syphilis experiment play in the development of the Belmont Report?
The Belmont Report was written in response to the infamous Tuskegee Syphilis Study, in which African Americans with syphilis were lied to and denied treatment for more than 40 years. Many people died as a result, infected others with the disease, and passed congenital syphilis onto their children.
Do IRB members get paid?
Affiliated IRB Committee members do not receive any direct monetary compensation for participation on the board. Unaffiliated IRB Committee members will be reimbursed at an amount not to exceed $60 per month to pay for internet access and parking. Reimbursement payments will be issued quarterly.
Does IRB protect animals?
The IRB reviews research that involves human participants. Research involving animals must be reviewed by the Institutional Animal Care and Use Committee (IACUC) before it begins.