What is the Lewis dot structure for pbr3

Rachel Hunter
Published Feb 12, 2026
The answer is A. trigonal pyramidal. The central atom for the compound is P. Looking at the central atom, it will have 3 bonds involving the 3 Br…
What is the molecular geometry molecular structure of pbr3?
The answer is A. trigonal pyramidal. The central atom for the compound is P. Looking at the central atom, it will have 3 bonds involving the 3 Br…
How many valence electrons does pbr3?
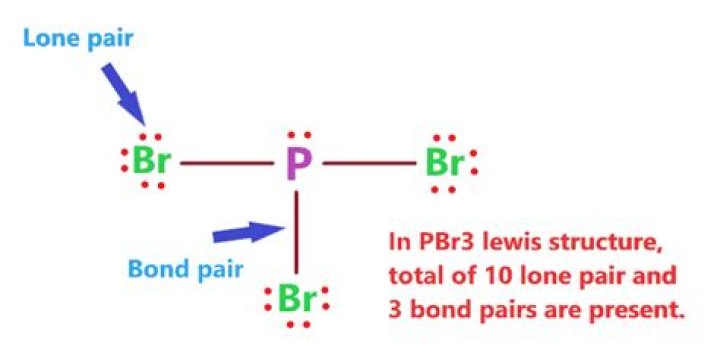
PBr3 has 3×7+5=26 valence electrons of which the Br atoms can consume 8 each (total 24) in a bonding and 3 lone pairs.
How many electrons are available for the Lewis structure of pbr3?
In the Lewis structure for PBr3 there are a total of 26 valence electrons. Three pairs will be used in the chemical bonds between the P and Br.What is the shape of a molecule of PBr three?
The trigonal pyramidal shape of a PBr 3 molecule (top view).
What is the correct Iupac name for PBr3?
PubChem CID24614StructureFind Similar StructuresChemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaPBr3 or Br3PSynonymsPHOSPHORUS TRIBROMIDE 7789-60-8 Phosphorous tribromide Tribromophosphine Phosphorus(III) bromide More…
What is the bond angle of pi3?
SiH4, an 8 valence electron system, is AB4; it has 109.5o bond angles. PI3, a 26 valence electron system, is AB3E; its bond angles are slightly less than 109.5o. BBr3, a 24 valence electron system, is AB3; it has 120o bond angles.
What is the central atom for PBr3?
Answer and Explanation: Phosphorous Tribromide (PBr3 P B r 3 ), the central atom is bonded to 3 Br atoms and one lone pair on the P atom. so, In PBr3 P B r 3 the hybridization is sp3 s p 3 .How many electrons should helium he have around its Lewis dot diagram?
octet rule: Atoms try to achieve the electronic configuration of the noble gas nearest to them in the periodic table by achieving a full valence level with eight electrons. exceptions to the octet rule: Hydrogen (H) and helium (He) only need two electrons to have a full valence level.
How do you determine valence electrons?Valence electrons can be found by determining the electronic configurations of elements. Thereafter the number of electrons in the outermost shell gives the total number of valence electrons in that element.
Article first time published onHow do you write a Lewis dot structure for an element?
- Step 1: Find the Total Number of Valence Electrons. …
- Step 2: Find the Number of Electrons Needed to Make the Atoms “Happy” …
- Step 3: Determine the Number of Bonds in the Molecule. …
- Step 4: Choose a Central Atom. …
- Step 5: Draw a Skeletal Structure. …
- Step 6: Place Electrons Around Outside Atoms.
How do you draw a Lewis dot structure?
- Step 1: Find the number of valence electrons.
- Step 2: Find the number of octet electrons.
- Step 3: Find the number of bonding electrons.
- Step 4: Find the number of bonds.
- Step 5: Draw it!
- Step 6: Add lone pairs.
- Step 7: Figure out which atom is charged.
- Step 1: Count the valence electrons.
How do you write a Lewis dot structure for a compound?
Add up the total number of valence electrons found in the entire compound. Don’t forget to include any positive or negative charges when determining this. Draw the simple structure (skeleton structure) of the compound by connecting everything with single bonds only. Add electrons to all the noncentral atoms.
What is the molecular shape for HBR?
NamesStructureMolecular shapeLinearDipole moment820 mDThermochemistry
What is the shape of SiS2?
ABSiS2 (3-D model)SiS2 – VSEPR shapelinear triatomicSiS2 – hybridizationspSiS2 – bond angle180 degrees
Why is PBr3 polar?
PBr3 is polar. Phosphorus has 5 electrons in its outer shell. It forms three single bonds with Br atoms, so PBr3 has three bonding pairs and one lone pair of electrons, the shape of the bonds is therefore trigonal pyramidal. Bromine is more electronegative than phosphorus so the P-Br bond is polar.
Which of the following is a correct Iupac name?
So the correct IUPAC name is 3-Ethyl-4,4-dimethylheptane. The structure of 3-Ethyl-4,4-dimethylheptane is, Options (B), (C) and (D) are incorrect as the naming is not done as per the IUPAC nomenclature of substituted alkanes. Therefore, the option (A) is correct.
What is the name of the compound with the formula pi3?
PubChem CID83485Chemical SafetyLaboratory Chemical Safety Summary (LCSS) DatasheetMolecular FormulaPI3 or I3PSynonymsPhosphorus triiodide 13455-01-1 Phosphorous triiodide Phosphorus(III) iodide triiodophosphane More…Molecular Weight411.6872
How do you name TiCO3?
- Titanium(II) Carbonate.
- Formula: TiCO3.
- Molar Mass: 107.8759.
What are the 7 valence electrons?
Any element in the halogen group will have seven valence electrons. These elements include fluorine, chlorine, bromine, iodine, and astatine.
How do you find the valence of an element?
valence = number of electrons in valence shell of free atom – number of non-bonding electrons on atom in molecule, valence = number of bonds + formal charge.